Playlist
Show Playlist
Hide Playlist
Infective Endocarditis (IE): Management
-
Slides InfectiveEndocarditis InfectiousDiseases.pdf
-
Reference List Infectious Diseases.pdf
-
Download Lecture Overview
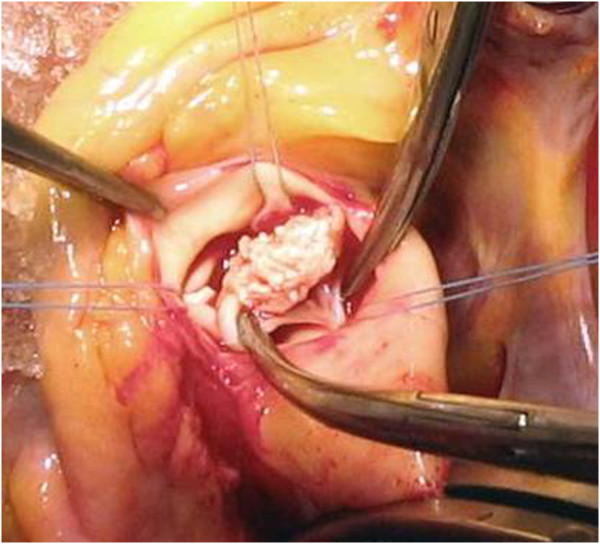
00:00 What’s new and an important approach to the management of infective endocarditis is an endocarditis team. 00:10 That team should consist of a cardiologist, a cardiac surgeon, the ID physician, a microbiologist and an intensivist. 00:21 A lot of these patients are going to be in the intensive care unit. So this should be the endocarditis team. This should be the team that’s involved in every patient that you diagnose with endocarditis. Now, why? We’re going to discuss each patient weekly It’s actually, in my view, malpractice to call in a cardiac surgeon after you’ve been treating somebody It’s actually, in my view, malpractice to call in a cardiac surgeon after you’ve been treating somebody for endocarditis in the hospital for two weeks. A cardiac surgeon should know about a patient who’s in the hospital who’s got endocarditis because things can go bad in a hurry. You don’t want to have a surprise for the cardiac surgeon. Oh, by the way, Mr. Jones has gone into heart failure because his treatment for endocarditis wasn’t good. So anyway, they make these decisions about the surgical priorities and especially for follow up. After their discharge, they would be followed by a cardiologist and ID doc ideally on the same day at one, three, six, and twelve months. When this strategy of the endocarditis team was employed, a study showed that the one year mortality went from 18.5% all the way down to 8.2%. 02:05 So, we can’t argue with those kinds of data. Every hospital should have at the ready an endocarditis team. 02:22 Now, what about starting antibiotics? Well, there are some patients that we want to start antibiotics immediately after we get our cultures cooking. We can then adjust the antibiotics based on the microscopy, culture and sensitivity of the organism. So, who are the high-risk patients? Well, certainly somebody who is in congestive heart failure, who has a stroke, recurrent embolus, septic shock. They’ve had fever for seven to ten days, or they’ve got a large, friable or enlarging vegetation, or if there’s obvious evidence that they’ve got more than just the valve involved, they’ve got part of their valve annulus, that the infection is extending. They’ve got, for example an annular abscess, or somebody who has endocarditis that’s got heart block. That is a relative emergency. Somebody who’s got very severe left-sided regurgitation and that would include aortic regurgitation or mitral regurgitation because one of the most important killers of infective endocarditis is congestive heart failure. 03:42 If their prosthetic valve is just terribly malfunctioning, we know it’s got to come out. 03:49 We know we’ve got to get treatment started fast. They have a low left ventricular ejection fraction or, and this is important, pathogens other than viridans strep especially Staph aureus which can destroy valves quickly; fungi, having fungal endocarditis is almost an immediate indication for removal of the valve because it’s so hard to treat medically; ditto gram-negative bacilli or enterococcal endocarditis; and those who come in in acute renal failure. 04:29 So those are the high-riskpatients that we need to act on fast.
About the Lecture
The lecture Infective Endocarditis (IE): Management by John Fisher, MD is from the course Cardiovascular Infections.
Included Quiz Questions
When is the most appropriate time to consult the cardiac surgeon for a patient with infective endocarditis?
- At the time of diagnosis
- If blood cultures remain positive after 2 weeks of therapy
- Only if it is prosthetic valve endocarditis
- When cultures return negative
- If the patient is in septic shock
Which of the following increases the mortality risk in patients with infective endocarditis?
- Stroke
- Younger age
- Infection of the mitral valve rather than the aortic valve
- Native valve endocarditis rather than prosthetic valve endocarditis
- Absence of intracardiac devices
Which of the following is NOT required during the management of infective endocarditis?
- Antiplatelet therapy
- Removal of any infected implanted devices
- Prompt diagnosis and institution of effective antimicrobial therapy
- Follow-up and prevention of recurrent infective endocarditis
- Establishing an endocarditis team
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |