Playlist
Show Playlist
Hide Playlist
Immune Tolerance and Autoimmune Regulator (AIRE)
-
Slides Autoimmune Disease.pdf
-
Reference List Immune System.pdf
-
Download Lecture Overview
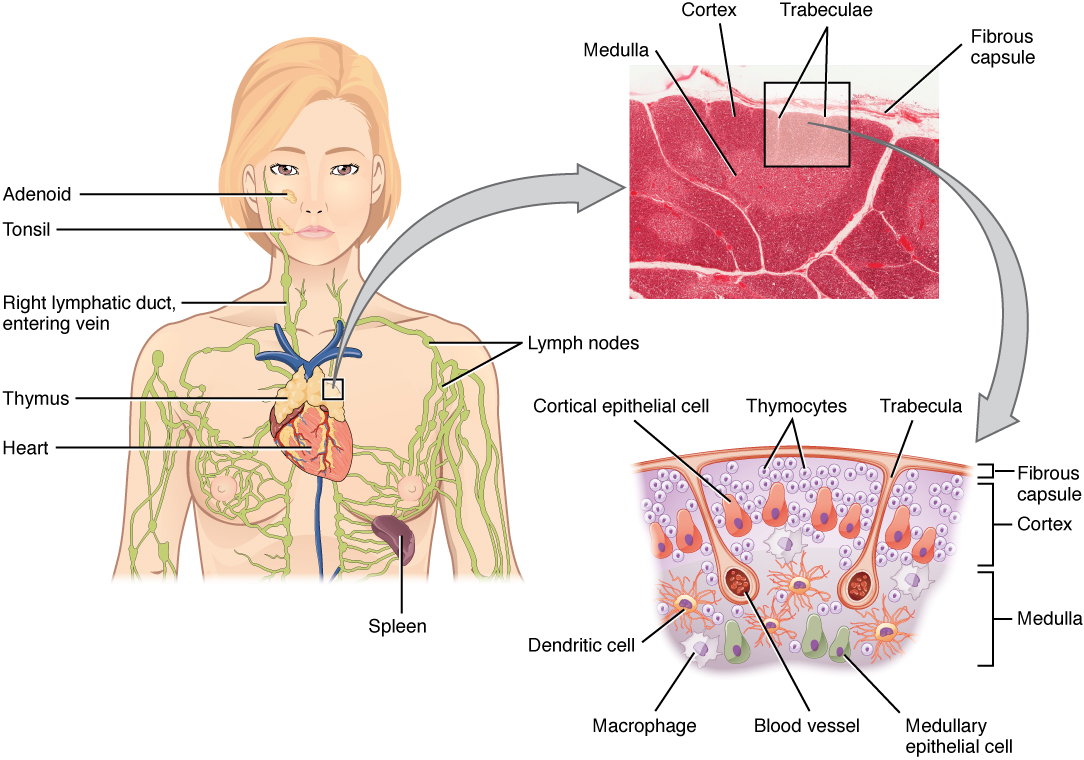
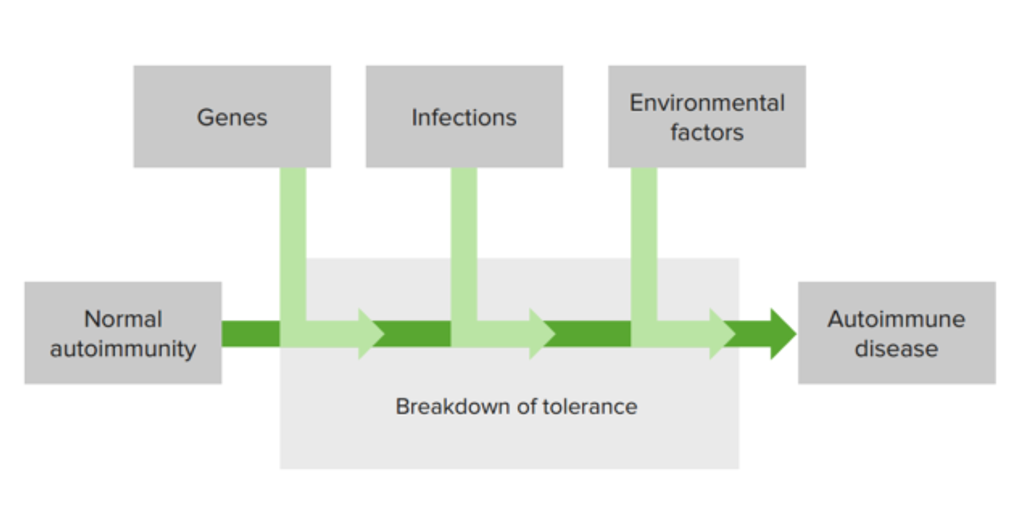
00:01 At the heart of the development of autoimmune disease is a breakdown in immunological tolerance. 00:06 In fact, the immunological tolerance mechanisms have evolved to prevent harmful autoimmunity. 00:14 Whilst T-cells are developing in the thymus and B-cells are developing in the bone marrow, they can undergo central tolerance. Recognition of self antigen in these primary lymphoid organs can lead to apoptotic cell death, with deletion of those particular cells. 00:34 Or in the case of B-cells, there is also the possibility for the B-cell to change the specificity of its antigen receptor. 00:43 This is referred to as receptor editing. 00:47 And regulatory lymphocytes can develop that can suppress unwanted autoimmune responses. 00:54 Central tolerance is not 100% efficient, so it is backed up with a number of peripheral tolerance mechanisms. 01:05 These can involve the induction of anergy, non-responsiveness in lymphocytes when antigen is seen in the absence of co-stimulation. 01:16 Usually during an infection, co-stimulatory molecules such as CD80 and CD86 are up-regulated on dendritic cells. 01:25 But when autoantigens are being shown to lymphocytes by dendritic cells, there is no co-stimulation and this results in anergy. 01:36 Apoptosis can also occur as a mechanism of peripheral tolerance. 01:41 And finally suppressor cells can suppress these responses in the periphery. 01:47 An important discovery a few years ago was that in the thymus, there is a transcription factor called the autoimmune regulator. 01:59 The function of AIRE, the autoimmune regulator is to cause expression of what are normally tissue-restricted antigens so that these antigens are also expressed in the thymus to facilitate tolerance. 02:17 There will be negative selection of the self-reactive T-cells and apoptotic cell death leading to deletion of those autoreactive T-cells. 02:29 In the absence of AIRE which can occur due to mutations, individuals will fail to delete tissue specific T-cells resulting in autoimmunity and this can lead to pathology - autoimmune disease. 02:52 In fact, autoimmunity is not in itself necessarily harmful. 02:59 If I was to take some blood from you and use a sensitive enough assay, I would find autoantibodies in your circulation. 03:05 We all have some level of autoimmunity. 03:09 But in most of us it is not pathological, it doesn’t cause disease. 03:14 So a low level of autoimmunity seems to be the norm. 03:18 Now whether that’s a kind of just noise in the system if you like, or whether there is actually some physiological function of autoantibodies in getting rid of dead and dying cells is still a matter of debate. 03:31 But just beware that autoimmunity does not equal autoimmune disease. 03:37 Clearly you have to have autoimmunity in order to develop autoimmune disease, but 95% of people don’t develop autoimmune disease. 03:46 But virtually all of us have some level of autoimmunity. 03:50 So why is it that in around about 5% of individuals, this normal autoimmunity becomes pathogenic? Well it’s a mixture of genetic factors, environmental factors such as infections. 04:05 And other environmental factors such as diet and stress and so forth, that lead to a breakdown of tolerance which affects the T-cells and B-cells that are potentially pathogenic. 04:19 And that breakdown in tolerance affecting those particular clones of T and B-cells can precipitate the development of autoimmune disease.
About the Lecture
The lecture Immune Tolerance and Autoimmune Regulator (AIRE) by Peter Delves, PhD is from the course Hypersensitivity and Autoimmune Disease. It contains the following chapters:
- Immunological Tolerance to Self Antigens
- The Development of Autoimmune Disease
Included Quiz Questions
Receptor editing mainly occurs during the maturation of which of the following types of immune cells?
- B cells
- Helper T cells
- Cytotoxic T cells
- Regulatory T cells
- Plasma cells
Which of the following is the main function of autoimmune regulator (AIRE)?
- Negative selection of T cells
- Creating T-cell anergy
- Apoptosis of regulatory B cells
- Creating B-cell anergy
- Negative selection of B cells
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
2 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
I needed a short, sweet, simple explanation of immune tolerance, and that is exactly what this is. Thank you!
Very good explained and very comprehensible for students, I'm actualy studying pathology and this kind of resources is exactly what I was looking for