Playlist
Show Playlist
Hide Playlist
Hydrolysis – Carboxylic Acids and Their Derivatives
-
Slides 08 Chemistry Advanced Le Gresley.pdf
-
Download Lecture Overview
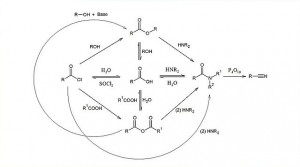
00:00 Right. So, let us move on to hydrolysis reactions. So, this is how you break them apart. So, we talked about hydro… reactivity with nucleophiles, water, to all intents and purposes, also serves as a nucleophile. And so, in this particular scenario, we have to consider the reactivity with water is much the same as reacting with any other. So, in this particular scenario, when it comes to hydrolysis as we have already said, amides, which are almost impossible to form without a coupling reagent, cannot easily be hydrolysed in the presence of water. 00:33 In fairness, it is possible refluxing conditions in concentrated sulfuric acid to achieve this, but again, this is not gentle and can disrupt other parts of the molecule that you may wish to protect. 00:45 Carboxylic esters are relatively easy to hydrolyse and indeed, there are a number of different mechanisms, acid catalysed and base catalysed, for their hydrolysis and indeed, that is how fats are broken down into glycerol and also, fatty acids used in soap manufacture in a process known as saponification. 01:06 Anhydrides are relatively easy to hydrolyse. However, they are not nearly as reactive with water as acid chlorides and sometimes, acetylation reactions with acid anhydrides can actually proceed in the presence of water where there is a better nucleophile such as an amine group. 01:23 In both cases, acid chlorides and anhydrides can be substituted in the carbonyl position to generate carboxylic acids. 01:32 In the case shown in green here, acid catalysts or base catalysts, in the case of carboxylic acid, result either in the formation of the conjugate base, carboxylate or indeed, in the acid itself, the carboxylic acid. But, these things are, especially in the case of the amides, relatively difficult to achieve. 01:52 Right. Now, I’d like to talk to you about the way in which esters can be formed using acid chlorides. You can see on the top left hand side that we have got an acetyl chloride here and the alcohol we are reacting it with is a cyclohexanol. The reaction works in the same way that all reactions with these carboxylic acid derivatives does, which is to say addition elimination. Lone pair on the oxygen on the alcohol attacks the carbonyl carbon, opens up the carbon-oxygen double bond which then reforms and kicks off the chloride. This generates, in this case, a cyclohexylethanoate. 02:28 You can achieve the same effect by reacting a cyclohexanol with an acid anhydride. Note on the right hand side, we are using acetic anhydride. In this case, the reaction proceeds in the same way: attack by the lone pair on the oxygens on the one of the carbonyl carbons, shown here in black, or that could be the other one, doesn’t matter, opens up that carbon-oxygen double bond which then reforms and kicks off an acetic acid group, shown in red there beneath the arrow. In both cases, the same compound, the same ester is formed, which is shown in the green box here. 03:05 Now, let’s also look at amide formation, much the same way you can use acid chlorides and acid anhydrides, shown left and right respectively, and they will react to give you an amide. The way in which this functions is that you got your amine which is one of the reactants in this particular… in this particular reaction and the lone pair on the nitrogen attacks the carbonyl carbon as before, opens up the carbon-oxygen double bond which then reforms and kicks off a chloride ion and that is shown underneath the arrow forming that amide in the center. 03:43 The same applies if you choose the correct acid anhydride which is shown in the bottom right. Same thing applies: lone pair on the nitrogen attacks the carbonyl carbon, in this case shown in black, but it doesn’t matter because if you look both sides have similar substitution patterns, opens up that carbonyl carbon which then reforms and kicks off the carboxylate and that’s how we form amides.
About the Lecture
The lecture Hydrolysis – Carboxylic Acids and Their Derivatives by Adam Le Gresley, PhD is from the course Organic Chemistry.
Included Quiz Questions
Which of the following is NOT related to coupling agent promoted peptide coupling or synthesis?
- Deoxyribonucleic acid (DNA)
- N,N'-Dicyclohexylcarbodiimide (DCC)
- Diisopropylcarbodiimide (DIC)
- N-Ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline (EEDQ)
- 2-propanephosphonic acid anhydride (T3P)
Saponification processes involve which of the following?
- Formation of soaps from carboxylic esters
- Formation of soaps from carboxylic acid amides
- Formation of soaps from acid anhydrides
- Formation of soaps from acyl chlorides
Which of the following represents a CORRECT trend for the reactivity of carboxylic acid derivatives towards hydrolysis?
- RCOCl > RCOOCOR > RCOOR > RCONH2
- RCOOCOR > RCOOR > RCONH2 > RCOCl
- RCOOR > RCONH2 > RCOCl > RCOOCOR
- RCONH2 > RCOOR > RCOOCOR > RCOCl
- RCOOR > RCOOCOR > RCOCl > RCONH2
Which of the following is an INCORRECT statement?
- Carboxylic acid derivates do not undergo hydrolysis reactions.
- Acyl chlorides and carboxylic acid anhydrides readily undergo hydrolysis to yield carboxylic acids.
- The hydrolysis of acyl chlorides, acid anhydrides or carboxylic esters requires an acid or a base as a catalyst.
- Hydrolysis of amides requires stronger acid or base catalysts at elevated reaction temperatures
- Hydrolysis reaction of carboxylic acid derivatives follows addition-elimination reaction mechanisms
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |