Playlist
Show Playlist
Hide Playlist
Coagulation Process and Regulation
-
Slides Thrombus Formation Coagulation Cascade.pdf
-
Reference List Pathology.pdf
-
Download Lecture Overview
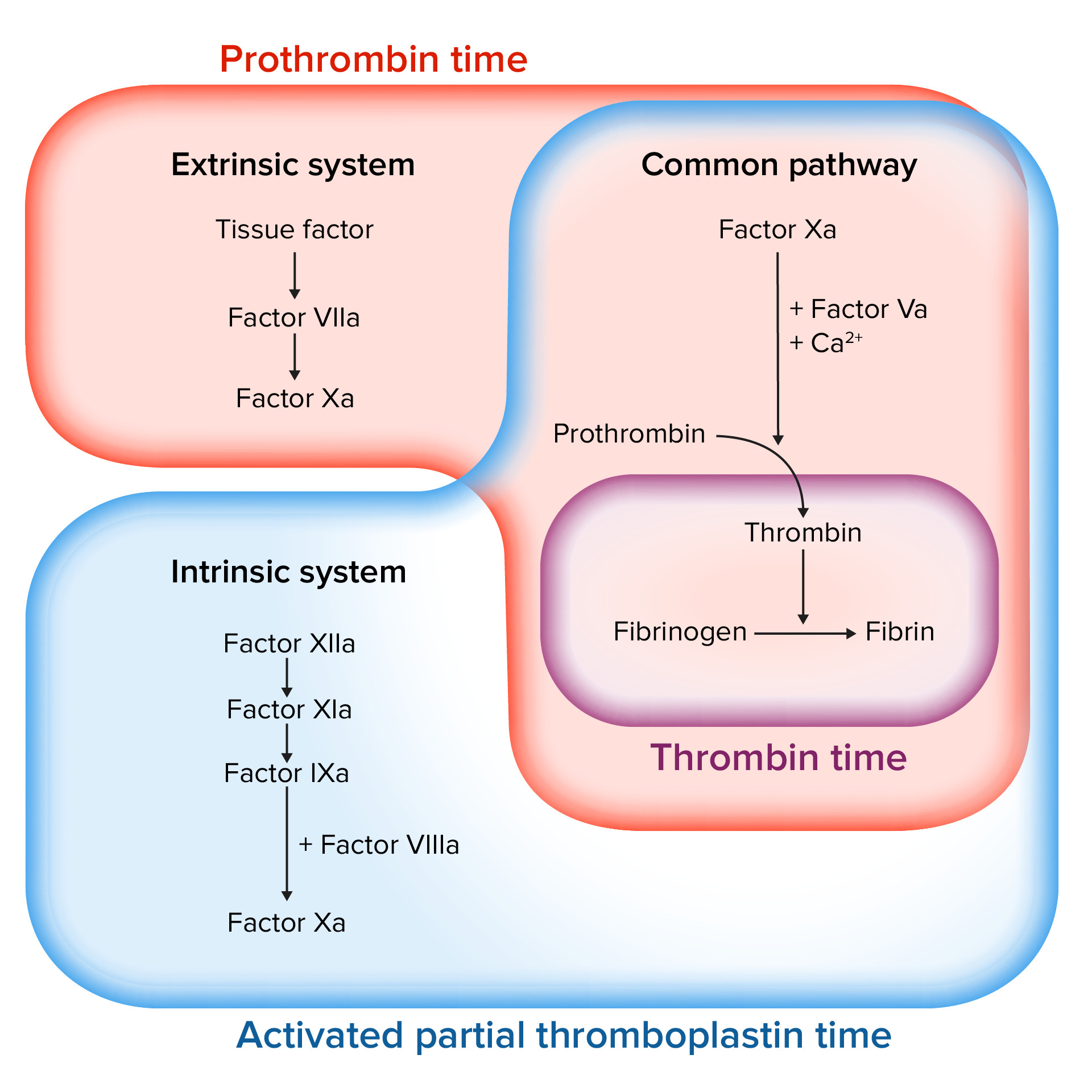
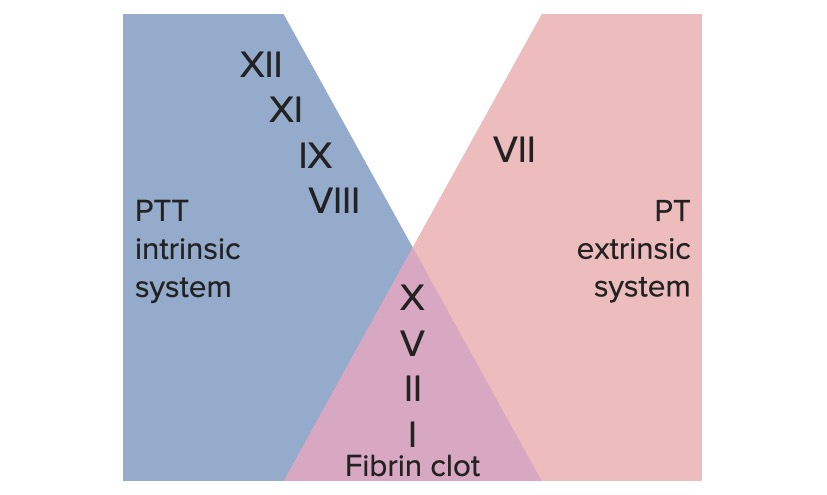
00:00 So with all of these different things, we got the intrinsic, the extrinsic pathway, what's really the major pathway if I have vascular injury, I've cut my finger and it needs to stop bleeding. It's usually mainly the extrinsic cascade that drives that real true coagulation and it's the exposure to tissue factor that occurs when I have injury. So it's the extrinsic pathway and vascular damage. So that vascular damage exposes thromboplastin, otherwise known as factor III otherwise known as tissue factor. And that tissue factor interacts with factor VII, make it a complex now with activated factor VIIa and tissue factor and we get to the common pathway and then we get into the activation of prothrombin to thrombin fibrinogen being crosslinked and there you have it. That's the extrinsic pathway. Now, why then do we have all those other things on the left hand side? Why do we have the intrinsic pathway? Well, it turns out that they get brought into the game through the activity of prothrombin or thrombin. Okay, so the tissue factor, factor VII, the red box there drives extrinsic pathway to activate thrombin but the same factors are also feeding back. So we have VIIa again in our extrinsic pathway that crosses over into the intrinsic pathway to activate IX to IXa and get us into the common pathway so we can get an amplification. Notably, you see here that VIIa activating IX to IXa completely bypasses the original involvement of XII. And it turns out that you can be completely totally deficient in factor XII and have completely normal coagulation that's because of this kind of bypass pathway that gets us from extrinsic to intrinsic. Okay, remember I said that thrombin is also acting back on all those pathways. In fact, not only is it driving fibrinogen to be cleaved to become fibrin and giving us a clot, at the same time it's feeding back. And you see these dotted red lines, these are inhibitory pathways where thrombin is going to be acting on these important cofactors or other pathways to cause other proteolytic cleavages that will inactivate. So thrombin, major linchpin, it drives both coagulation and also helps to regulate that coagulation so we don't clot off the entire arm when we cut our finger. 02:45 So, thrombin, also known as activated factor II, cleaves factors V, VIII, XI, and XIII. Some of that is procoagulant, some of that is anticoagulant. So thrombin is much more than just the step before fibrin. The other thing that it does is that it activates a whole variety of other pathways and then we're going to bring those up now. So thrombin will interact with a specific receptor. It's called PAR1, stands for the proteolysis activated receptor 1. It's on a variety of cells, we'll see what cells in a minute. But interestingly, it's not just thrombin binding to that receptor. It actually binds and cleaves that receptor so that we now get an activated molecule that autostimulates itself. Wow, that's going to be incredibly a problem because it really doesn't go away very much. Okay, so thrombin now has cleaved that. It turns on itself, binds, and activates. And what is it activating? So here we have thrombin acting on platelets. It will drive platelet aggregation and the release of granules. So we produced thromboxane A2 so we can drive improved platelet clotting. It will also act on endothelial cells. Those endothelial cells are going to be making a variety of things such as nitric oxide, prostaglandin II otherwise known as prostacyclin and tPA. And these are going to tend to be antithrombotic. They're going to tend to start the feedback in addition of a whole thrombotic cascade, and we'll talk more about those in a subsequent session. 04:34 Thrombin will also activate lymphocytes. Makes sense actually. You really want in an area of injury to have the immune system also called in to do their thing. It will drive endothelial cells to monocytes to make platelet-derived growth factor. Although it's called platelet- derived growth factor, we now know that it's made by a variety of cell types including in this case endothelium and monocytes. That PDGF synthesized by those cells driven by thrombin will act on smooth muscle cells. Also makes sense because the smooth muscle cells around the vessel will also probably had been injured in the process and we need to drive their proliferation to provide healing for the blood vessel. Thrombin will also induce increased neutrophils adhesion. Again makes sense because in an area of injury we want to bring in the innate immune response. So thrombin is doing a whole lot more than just driving fibrinogen to become fibrin. It is doing a whole ride of activities including counterregulating the aggregation thrombosis response. And with that, we conclude the coagulation factors.
About the Lecture
The lecture Coagulation Process and Regulation by Richard Mitchell, MD, PhD is from the course Hemostasis.
Included Quiz Questions
What principally drives the coagulation cascade after an injury?
- Extrinsic pathway
- Intrinsic pathway
- Common pathway
- Phospholipid and calcium ions
- Thrombin activation
Thrombin cleaves which of the following factors?
- V, VIII, XIII
- VI, VII, XII, XIII
- I, III, V, IX
- VII, VIII, IX, XIII
- IV, V, VI, VII
Thrombin is necessary for which of the following events?
- Endothelial activation
- Lymphocyte inhibition
- Smooth muscle contraction
- Activation of factor X
- Exposure of tissue factor
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |