Playlist
Show Playlist
Hide Playlist
Carboxylic Acids and Their Derivatives: Introduction
-
Slides 08 Chemistry Advanced Le Gresley.pdf
-
Download Lecture Overview
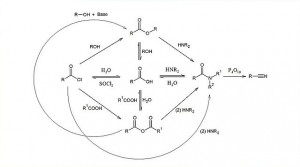
00:02 Related to the carbonyl functional group we discussed in the last lecture, I’d now like to introduce you to carboxylic acids and their derivatives; the derivates being, again, increasingly more useful from a synthetic perspective, as we will see. 00:19 Carboxylic acids, as you can see on the board, have the general structure shown here where you have an R group which can be alkyl or aryl attached to a carbonyl and on that same carbon, that carbonyl carbon you have an OH group attached. This imparts an acidic quality as we will see and hence, the term as carboxylic acids. 00:41 In this particular case, when you are using the… considering the nomenclature from an IUPAC perspective, what you do is you take away the “-e” at the end of the corresponding alkane and then you replace it with “-oic acid”. So, for example, in the case of a 2 carbon system ethane, we remove the “–e” and it is ethanoic acid. In the case of a 3 carbon system, including the carbonyl carbon such as propane, we would then call propanoic acid and butanoic, pentanoic and so on and so forth. 01:16 Examples as I said are ethanoic acid which is otherwise known as acetic acid and here, the substitution nomenclature is also the same. Note that the carbonyl carbon has the priority and therefore, when you count back 1, 2, 3, 4, 5 to where the two substituents are you will call this 5,5-dimethylhexanoic acid. Always remember that the carbonyl carbon counts as that carbon. So, when you are looking at the chain, it is not a pentanoic acid, rather it is a hexanoic acid. 01:54 So, we talked about acidity and acitum bases back in Module II and I’d briefly discussed the idea of induction and how this can improve or increase or decrease the acidity accordingly. 02:09 And here, we have the equilibrium shown for the disproportionation in this case of a carboxylic acid in water to give the conjugate base which is known as a carboxylate and of course, the corresponding hydronium ion, which is, as you should be able to remember, H3O+. 02:28 The acidity constant or the acidity equilibrium is given here as Ka, which equals the product concentration, carboxylate multiplied by hydronium ion divided by the concentration of the carboxylic acid. Now, some of you who may be paying attention will realise that why have I not included the water in that equilibrium since I have obviously included it in the equation. The reality is that water, as a liquid, always have a… has a value of unity or one and therefore, it’s unimportant to actually put it into the equation since we just would end up multiplying the concentration of the carboxylic acid by one. If we take the -log to the base 10 of Ka, we obtain pKa.
About the Lecture
The lecture Carboxylic Acids and Their Derivatives: Introduction by Adam Le Gresley, PhD is from the course Organic Chemistry.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |