Playlist
Show Playlist
Hide Playlist
Amides Formation from Esters – Carboxylic Acids and Their Derivatives
-
Slides 08 Chemistry Advanced Le Gresley.pdf
-
Download Lecture Overview
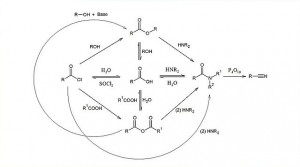
00:00 Right. Amide formation from esters. 00:04 If you recall a couple of slides ago, I talked about the biological input or importance of this because activation of certain amino acids by transesterification is important for them to then be converted into amides and we will see this in Module IV when we start looking at the biology behind it. 00:22 In this particular case, I am sharing a relatively simple addition elimination reaction. In the case of methylamine or methylamine, depending on which side of the water you are from, and you have the lone pair of nitrogen again attacking, in this case, the carbonyl carbon, opening it up and, in this case, kicking off a methoxide ion. Now, this in itself is not a reaction which would necessarily always go to completion and you can tell this by the fact you have an arrow going in one direction and an arrow going in the other direction, which suggest that this is in equilibrium. And so, you should be trying to remove the amide acid that is formed to push the equilibrium in the direction of the desired product. 01:04 It should be stressed that esters cannot be formed from amides, although having said this from a chemical perspective, it’s not possible, but in biology, enzymes do achieve this and as I said, we will see more of that a little later on. 01:16 It is also possible to reduce esters to alcohols. This reduction can take place through the use of a hydride transfer reagent such as lithium aluminium hydride or lithal, for short, followed by proteination to remove the lithium salt. As you can see, the ester is converted, first, to an aldehyde and then in this case, back down to the primary alcohol. It is also possible to reduce amides to amines using the same technique, although indeed, another reducing agent [Unaware 00:33:10] is also possible to reduce this as well. 01:51 Note, we are converting the carbonyl into an alcohol in the first instance, which forms an amine, which is then reduced even further. So, in this particular case, we have converted our amide carbonyl into a CH2 group, as you can see in the bottom right hand corner. So, care must be taken and indeed, there are a number of text books available when you actually have got multiple functional groups that you only want to reduce one or other, but not usually both or maybe several of them. So, you have to be careful to select your reagents in order to make sure that you have achieved the desired outcome in your reaction. 02:24 So, in summary, substitution reactions, and this is what we started off with, involves nucleophilic attack and a carbonyl carbon with a reasonable leaving group in the form of that denoted by Z. This opens up that SP2 hybridised carbonyl carbon, generates a tetrahedron intermediate and then the negative charge on the oxygen kicks back in, reforms the carbonyl and discharges the Z leaving group, shown here as Z-. 02:53 There are a number of different substituents which forms the category of Z, in this case, but when you are trying to substitute in that position, you need to make sure your nucleophile is a worse leaving group than your Z, whatever that may be. 03:08 So, Z- must be a better leaving group than the nucleophile, otherwise the reverse reaction can take place. The nucleophile must also be strong enough to actually attack whichever the carboxylic acid derivative you are looking at. It also must be an electrophile good enough to react with the nucleophile in the first place and this is where the amide falls down. 03:32 Bearing in mind, if we go back to the structure of the amide, you actually have the lone pair of the nitrogen donating back into the carbonyl carbon and decreasing the size of the dipole. 03:41 And so, finally, this is a scheme which shows not just how carboxylic acid derivatives can be formed, which is in the first part in the center, but also on the right hand side, the reactions and the inter conversions that can take place. And my recommendation is that you actually study this and make sure you are familiar with those interconversions. 04:02 Even if you are not necessarily 100 percent certain about the particular mechanisms, you should be aware of how acid chlorides can be converted to a wide variety of different other carboxylic acid derivatives and vice versa.
About the Lecture
The lecture Amides Formation from Esters – Carboxylic Acids and Their Derivatives by Adam Le Gresley, PhD is from the course Organic Chemistry.
Included Quiz Questions
Amino acid activation in the living cells involves which of the following?
- Transesterification of amino acid -COOH group with -OH group at 2’ or 3’ position of ribose sugar of CCA 3’ end of a tRNA molecule by aminoacyl-tRNA synthetase enzyme.
- Transesterification of amino acid -COOH group with -OH group at 2’ or 3’ position of ribose sugar of CCA 5’ end of a tRNA molecule by aminoacyl-tRNA synthetase enzyme.
- Transesterification of amino acid -COOH group with -OH group at 2’ or 3’ position of ribose sugar of AAC 3’ end of a tRNA molecule by aminoacyl-tRNA synthetase enzyme.
- Transesterification of amino acid -COOH group with -OH group at 2’ or 3’ position of ribose sugar of AAC 5’ end of a tRNA molecule by aminoacyl-tRNA synthetase enzyme.
- Transestarification of amino acid -COOH group with -OH group at 5’ position of ribose sugar of CCA 3’ end of a tRNA molecule by aminoacyl-tRNA synthetase enzyme.
Which of the following is an INCORRECT statement?
- Carboxylic esters readily give amides during reduction reactions under standard laboratory set-ups.
- Carboxylic ester and amide reductions take place in the presence of a strong reducing agent.
- Carboxylic esters first get reduced to the aldehyde, followed by subsequent reduction to the corresponding primary alcohols in the presence of LiAlH4.
- An amine is formed when an amide gets reduced in the presence of LiAlH4.
- The amide group donates the lone pair of nitrogen back to the carbonyl group, hence reduce the polarity of the functional group.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |