Playlist
Show Playlist
Hide Playlist
Acidosis and Alkalosis – Laboratory Diagnostics
-
Slides DiagnosticsAcidosisAlkidosisStep1-2 RespiratoryPathology.pdf
-
Reference List Pathology.pdf
-
Download Lecture Overview
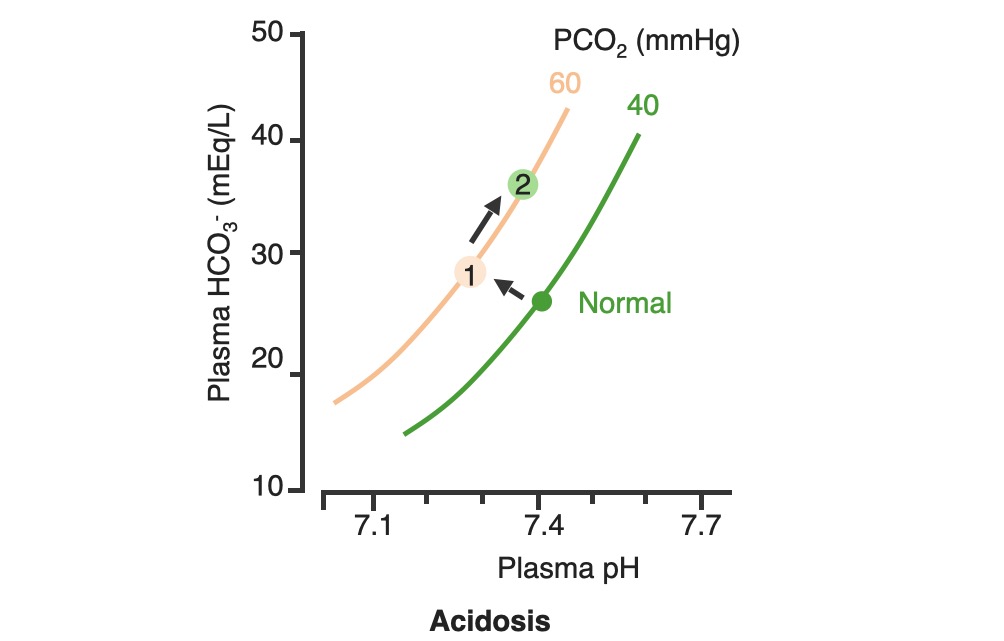
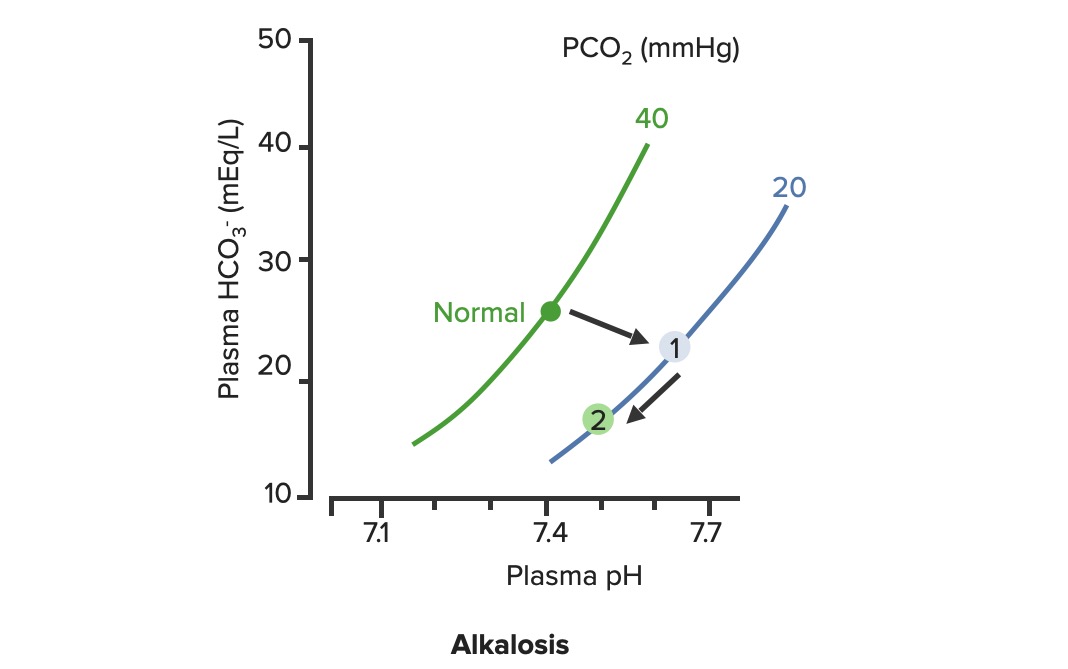
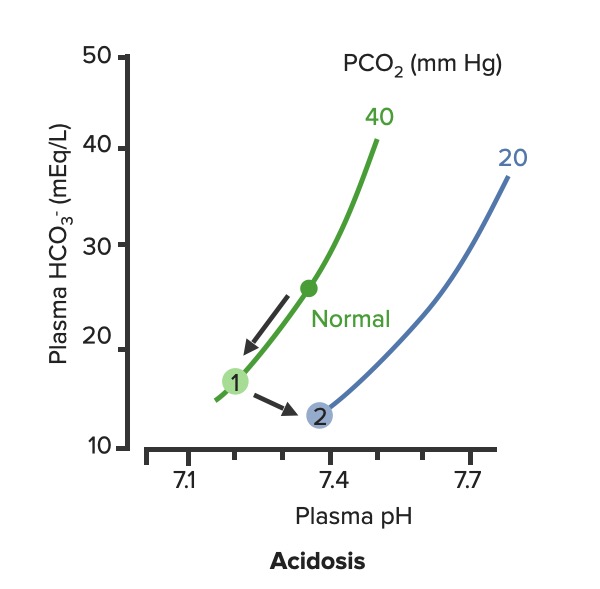
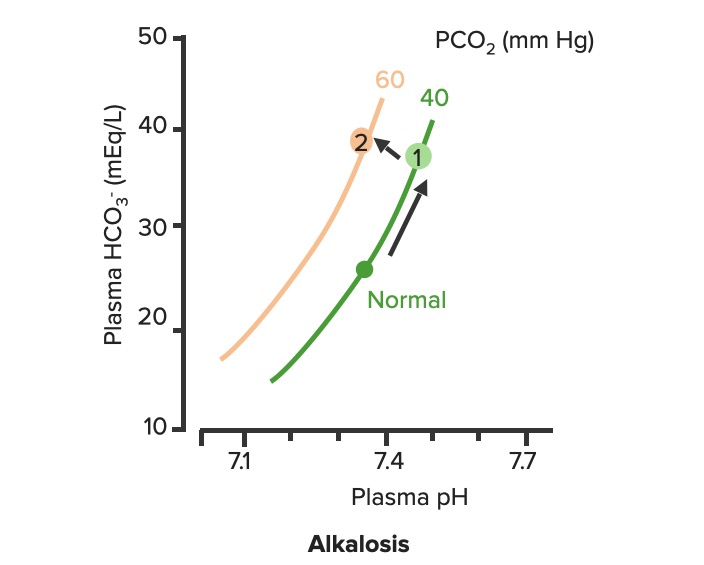
00:01 Okay. Now, what we’ll do here is we're going to dissect the heck out of this flowchart algorithm. 00:06 This is absolutely imperative that you understand as to what goes on because the differential diagnosis that you come up with are crucial for you to properly save your patient. 00:19 What steps do you need to take or what particular path do you need to follow so that I can come up with the proper differentials? Let me set this up. 00:28 At first, we’ll take a look at our arterial pH. 00:32 It is what you will always begin with. 00:34 The arterial pH at least put you in the right category and the right path as I was referring to as to what steps you need to take next. 00:42 If you find your pH to be less than 7.35 this automatically puts you in the realm of acidemia. 00:51 What I’ll do here at first is give you an example of one path and as I said, you’ll utilize all of this and dissect this further but you’ll come back and always take a look at the overview so you quickly know, "Oh, this is acidotic." And then what kind this is, it is metabolic or respiratory? Speaking of which, if it is acidosis then why do we even have this in dissection? Because it's about carbon dioxide, isn’t it? It's about your ability to breathe and what you're doing with your carbon dioxide. 01:21 Off the back, you know that at this point that your pH is decreased, so do then you know it's acidotic? If you know that it’s acidotic, then what's your next step? Well, here, let's say that you find your PCO2 to be elevated. 01:34 What happened do you think at this point? You know the patient’s breathing pattern is probably decreased, correct? Hypoventilation, maybe dyspnea. What do we call this medically? We call this hypercarbia or hypercapnia. 01:48 And once we get into that then we’ll talk about the reasons as to why the patient is feeling hypoventilated. 01:55 There differentials. 01:56 If on the other hand you find your patient have metabolic acidosis, how do you know? Well, you take a look at your carbon dioxide, and you find it to be 25. Stop there. 02:10 Can you think - can you possibly have a level carbon dioxide of 25 and call it respiratory acidosis? How is that possible? Really, your normal is 40. 02:21 Carbon Dioxide to you equates to hydrogen production so if you find your carbon dioxide to be dramatically decreased and I gave you 25, how could that be respiratory acidosis? It's not. So don’t be confused. 02:34 What is your reference point? The pH. You find your pH to be less than 7.35 and you find your carbon dioxide to be 25. 02:42 Wow, that’s not respiratory issues. Who do you turn to next? Good, the bicarb. 02:49 And I told you earlier, bicarb between 22 to 26 and when you get your bicarb levels it will be quite dramatic. 02:56 Here we find your bicarb to be less than 20, maybe it's 18. 03:00 My primary disorder, please? Metabolic acidosis. 03:05 What's your next step in management? Anion gap. 03:08 And we’d get into that, we’ll take a look at our sodium which is your one clinical cation that you use, from this you subtract your anions and there are two of them clinically: chloride and bicarb. 03:22 And this will give you an approximate anion gap of 8 to 12 and when we get into this, we’ll get into our differentials of anion gap and normal, but in the clinical we call this nonanion gap, metabolic acidosis. 03:36 Are you developing a plan as to how to attack this algorithm? And as you do so, at some point, we’ll then going to switch over to the alkalosis and we’ll go to the same thing. 03:46 Let’s begin please. 03:47 So, here we go at first, we're going to check your arterial pH. 03:52 If you find this to be less than 7.35. What's the normal arterial pH? Between 7.35 and 7.45. 03:59 So you're less than 7.35, you know for a fact your patient is in a state of acidemia. 04:05 Next, you take a look at your carbon dioxide and you find that to be elevated, take your time, step-by-step organize your thoughts. 04:11 Your carbon dioxide elevated. 04:13 What is your breathing pattern which then allow the carbon dioxide to be elevated? Hypoventilation. Decrease respiratory rate. You are now holding on to carbon dioxide. 04:24 What is carbon dioxide mean to you? It equals hydrogen, hence pH decreases. Are we clear? You find your carbon dioxide to be above 40, clinically speaking, above 44. 04:34 Now, hypoventilation is my issue, let’s walk through this. 04:39 Hypoventilation maybe there's airway obstruction, maybe acute lung disease, chronic lung disease, opioids, sedatives or weakening of the respiratory muscles. 04:49 That’s a lot of differentials for hypoventilation. 04:51 Guess what? Something else that I'm going to introduce to you here under laboratory findings that are extremely important for you to know the different types of well, why the patient could have issues with breathing? I want you to think about the alveoli - work with me here. 05:10 Go to the alveoli then type 1, type 2 pneumocytes, squamous cell, type of histology. 05:17 very thin that membrane allows for gasses to pass through oxygen moving into the capillaries, carbon dioxide being accepted by the alveoli, right? You move through the interstitium, and then you go in to the capillaries, why am I bringing this up? Because at some point, we have to go through the all-important clinical formula known as the A-a gradient. 05:39 A-a gradient. It’s big A minus little a gradient. 05:45 Why is that important? Why is the actual terms so important? Alphabet. The big A represents what? The alveolar. 05:53 Is that clear? The alveolar. 05:55 What’s the little a represent? The arterial, so that'll be part of your ABG. 05:59 So, what you’ll do here as you go through the A-a gradient is measure the oxygen and see as to whether or not your A-a gradient is it widened or if it’s normal. 06:09 Let me give you a clue. 06:11 Here, under hypoventilation, say that your patient is taking too many opioids, okay. 06:19 Maybe your patient has sickle cell disease and is taking opioids. 06:23 Why sickle cell disease? Because they're in pain. Where? Everywhere. 06:27 "Ow, my hand hurts." Dactylitis. 06:29 "Ow, my chest hurts." Acute chest syndrome. 06:32 Maybe stroke like issues. Sickle cell disease, that’s a lot of pain. 06:36 That pain is relieved by this patient taking opioids. 06:39 In fact, 100% of your sickle cell disease patients are taking opioids. 06:43 Now, these are my problem, the opioids, upon excess consumption causes what? Knocking out your respiratory center. 06:50 Wow, what about that respiratory center? It contains central chemoreceptors and it is detecting, whom? Carbon dioxide in your cerebrospinal fluid. 07:01 Discussion. Physiology bring in into play here, yet, once again. 07:05 Now, if you're going to knock out the respiratory center, what then - how's the patient rendered hypoventilation? Take your time. 07:15 So now, you get the oxygen in the alveoli, any problem there if your respiratory centers been knocked out. 07:20 No, you still get the oxygen in the alveoli so it's filled there in the alveoli from the ambient air, and we’ll walk through the formula, through the trachea and all the way down into the alveoli. 07:32 On the other side of the membrane, where am I? The alveolar membrane, on the other side of the membrane. What is it? Capillaries. From the alveoli onwards, pulmonary capillaries, what kind of blood vessels are these? Oxygenated blood vessels: pulmonary capillary, pulmonary veins. 07:50 And these are filled with oxygen, with approximately PO2 of 100. 07:54 Well, if it’s hypoventilation, and you're not taking in your air because respiration centers has been knocked out. 08:01 I don’t have as much oxygen in my alveoli. 08:05 It gets in there but not as much, so that’s a decrease. 08:08 If your alveolar oxygen is decreased, which direction am I moving? Always forward, meaning into the pulmonary capillary, so if that is decreased, is it safe to say that our arterial oxygen is also diminished? Of course. So both are diminished, the big A and the little a. 08:24 What is your widening of your A-a gradient? Is there one? No. It's a normal A-a gradient. 08:29 One big example is to when it's imperative or crucial for you to use a formula called the A-a gradient, so that you can then determine what's causing this hypoxia. 08:41 Is it of an extrapulmonary issue such as your respiratory center knocked out or is it an intrapulmonary issue? Let's continue. So, we still have acidosis and here we have a bicarb level less than 20. 08:52 So now, we can now determine or confidently diagnose your patient so far with metabolic acidosis. 08:59 Now, the only time that you’ll be using this next formula known as the anion gap is only for metabolic acidosis. 09:07 Keep this simple. Metabolic acidosis your next step is to do anion gap and I’ll tell you why upcoming. 09:14 Anion gap would be once again your major cation is sodium, give me the levels between 135 to 145 and we have bicarb and chloride, chloride 95 to 105, bicarb, how much once again? Between 22 to 26. 09:29 So, at this juncture we have now looked at our acidotic branch and arm, let's take a look at alkalotic. 09:35 Take a look at your arterial pH and if you find your pH to be above 7.45, what realm does this put you in automatically? Good. Alkalosis. 09:44 Close your eyes let me give you the following. 09:47 Say that you found your PCO2 to be less than 40 in fact it’s down to maybe 18. 09:53 So far, what kind of alkalosis is this? Excellent. Respiratory alkalosis. 09:58 Open up your eyes and take a look. 10:00 Less than 36, less than 36 to you should indicate that you're in the state of respiratory alkalosis. 10:06 Next, what do you do? Oh, your patient is probably breathing very, very quickly so this would be hyperventilation. 10:14 When there's hyperventilation, how do you think this occurred? Well, hysteria. What's that mean? Panic, which is not what you're doing right now, but if there is and watch your breathing rate, really rapid hyperventilation. 10:28 Are you blowing off carbon dioxide? Sure you are. 10:30 Look at this one, this is hypoxemia. How does this occur? Hypoxemia in this particular case maybe high altitude. 10:40 What happens when you go into high altitude? Barometric pressure drops. 10:45 Normal sea level, barometric pressure, is what? 760. 10:51 When you go into high altitudes such as Mt. Everest, then what happens to your barometric pressure? Maybe dropped down to 250, that’s really low so there's a lot of decreased pressured oxygen that you're breathing in. 11:04 Tell me about your overall percentage of oxygen? Your FiO2 is still at 0.2. 11:09 That’s not changing I told you, planet Earth, your oxygen percentage will not change but the barometric pressure did. So I'm breathing in decreased oxygen. 11:18 Work with me. I get in to the alveoli. How much oxygen? It gets there but it’s a decreased, same concept. 11:27 What same concept? If you have a decreased in oxygen in the big A, alveoli, is it safe to say that you have decreased oxygen in the pulmonary capillary. 11:38 The little a. Yes, you do. 11:40 If both are decreased tell me about their A-a gradient? Is there a widening or is it normal? Yet, once again, normal. 11:48 So, you have two major causes right now resulting in a normal A-a gradient. 11:53 Strictly, hypoventilation on your left due to something like your respiratory center being knocked out or if you're going to high altitude. 12:02 Well, this is interesting, you're breathing off carbon dioxide and you have respiratory alkalosis but then understand because of that hypoxemia, your respiratory rate is increased with hyperventilation, increased hyperventilation resulting in a normal A-a gradient. 12:19 What else? This is a big one that you wanna keep in mind. 12:22 Salicylates. Early, early. 12:25 Now, there are many theories is out there but you must know if your patient has consumed excess aspirin, salicylates. 12:32 Now, what it does it stimulates your respiratory center, your patient is going to be hyperventilating early and so therefore your patient is gonna be suffering from respiratory alkalosis. 12:42 What else may also result in hyperventilation? Tumor or even PE. 12:46 Hyperventilation, hyperventilation, hyperventilation. 12:49 Respiratory alkalosis. What's your carbon dioxide level? It's less than 36 because you're blowing off carbon dioxide. 12:56 Okay. Now, close your eyes, say that your PCO2 is at 50, but you find your pH to be greater than 7.45. 13:05 There is no way that this is a primary respiratory issue, is there? Because your pH is 7.45, let's say 7.6. 13:14 Alkalosis. Well, let's say that your bicarb is less than or greater than 28. 13:21 So, if you find your bicarb to be greater than 28 then you know that your patient is in a state of metabolic alkalosis. 13:29 How did this occur? Maybe there's loop diuretic, maybe there's vomiting. 13:34 Stop there for one second. Interesting. Loop diuretic and vomiting. 13:39 And what I would like for you to jump to real quick is hyperaldosterone. 13:43 Loop diuretic. How much fluid are you losing? Tons, tons of fluid. 13:48 The more that you deliver sodium to the distal convoluted tubule, what are you doing? You are removing, you're reabsorbing your sodium, and how does it gets rid of? In other words, you find your bicarb increasing, get rid of your hydrogen. 14:02 This is metabolic alkalosis. 14:04 Vomiting. Remember this conversation. With vomiting, what are you doing? You are getting rid of fluid and so therefore you are in a contractile state and when you're in a contractual state, once again you’ll be in a state of metabolic alkalosis. 14:17 There's more steps here. Now, please understand, you’ll never do an anion gap. 14:22 I won't say never, but with metabolic alkalosis highly unlikely that you’ll do an anion gap. 14:29 So, do not waste your time doing an anion gap for metabolic alkalosis. 14:33 Antacid, pretty straightforward, you're literally consuming your bicarb.
About the Lecture
The lecture Acidosis and Alkalosis – Laboratory Diagnostics by Carlo Raj, MD is from the course Pulmonary Diagnostics.
Included Quiz Questions
Which of the following values would represent a normal pH of the arterial blood?
- 7.35 - 7.45
- 7.05 - 7.45
- 7.50 - 7.55
- 7.03 - 7.30
- 7.04 - 7.40
Which of the following would lead to a state of respiratory acidosis?
- Opioids
- Uremia
- Diarrhea
- Loop diuretics
- Methanol poisoning
Which of the following is used to calculate the anion gap?
- Na - (Cl + HCO3)
- Na - (K + HCO3)
- Na - (Cl - HCO3)
- Na + (Cl - HCO3)
- Cl - (Na + HCO3)
Which of the following is not an etiology of anion gap metabolic acidosis?
- Vomiting
- Uremia
- DKA
- Lactic acidosis
- Salicylate overdose
Pick the correct etiology using the following values: pH < 7.35 Bicarbonate = 18 mEq/L Anion gap = 24 mEq/L
- Ethylene glycol
- Spironolactone
- Acetazolamide
- Renal tubular acidosis
- Hyperaldosteronism
Which of the following is the correct process based on the following values? pH = 7.50 pCo2 = 32 mmHg Bicarbonate = 20 mEq/L
- Respiratory alkalosis
- Respiratory acidosis
- Anion gap metabolic acidosis
- Non-anion gap metabolic acidosis
- Metabolic alkalosis
Choose the correct answer using the following values: pH = 7.25 pCO2 = 30 mmHg HCO3 = 16 mEq/L Anion gap = 8 mEq/L
- Non-anion gap metabolic acidosis
- Anion gap metabolic acidosis
- Respiratory acidosis
- Respiratory alkalosis
- Metabolic alkalosis
A-a gradient is used to determine which of the following?
- If hypoxia is extra-pulmonary or intra-pulmonary.
- If metabolic acidosis has an anion gap or not.
- If arterial blood is delivered to the peripheral tissues.
- Relationship between arterial pH and PO2.
Which of the following processes occurs in high altitude sickness?
- Respiratory alkalosis
- Respiratory acidosis
- Metabolic alkalosis
- Anion gap metabolic acidosis
- Non-anion gap metabolic acidosis
Which of the following represents the correct process that occurs during salicylate poisoning?
- Early stage - Respiratory alkalosis, Later stage - Anion gap metabolic acidosis
- Later stage - Respiratory alkalosis, Early stage - Anion gap metabolic acidosis
- Early stage - Respiratory acidosis, Later stage - Anion gap metabolic acidosis
- Early stage - Respiratory alkalosis, Later stage - Non-anion gap metabolic acidosis
- Early stage - Respiratory alkalosis, Later stage - Anion gap metabolic alkalosis
Customer reviews
3,5 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
1 |
| 1 Star |
|
0 |
I still don't understand. I have played the video so many times. Maybe its just a very hard concept.
He explains it so well, but I wish there was subtitles