Playlist
Show Playlist
Hide Playlist
Hypersensitivity: Type 2
-
Slides Hypersensitivity.pdf
-
Reference List Immune System.pdf
-
Download Lecture Overview
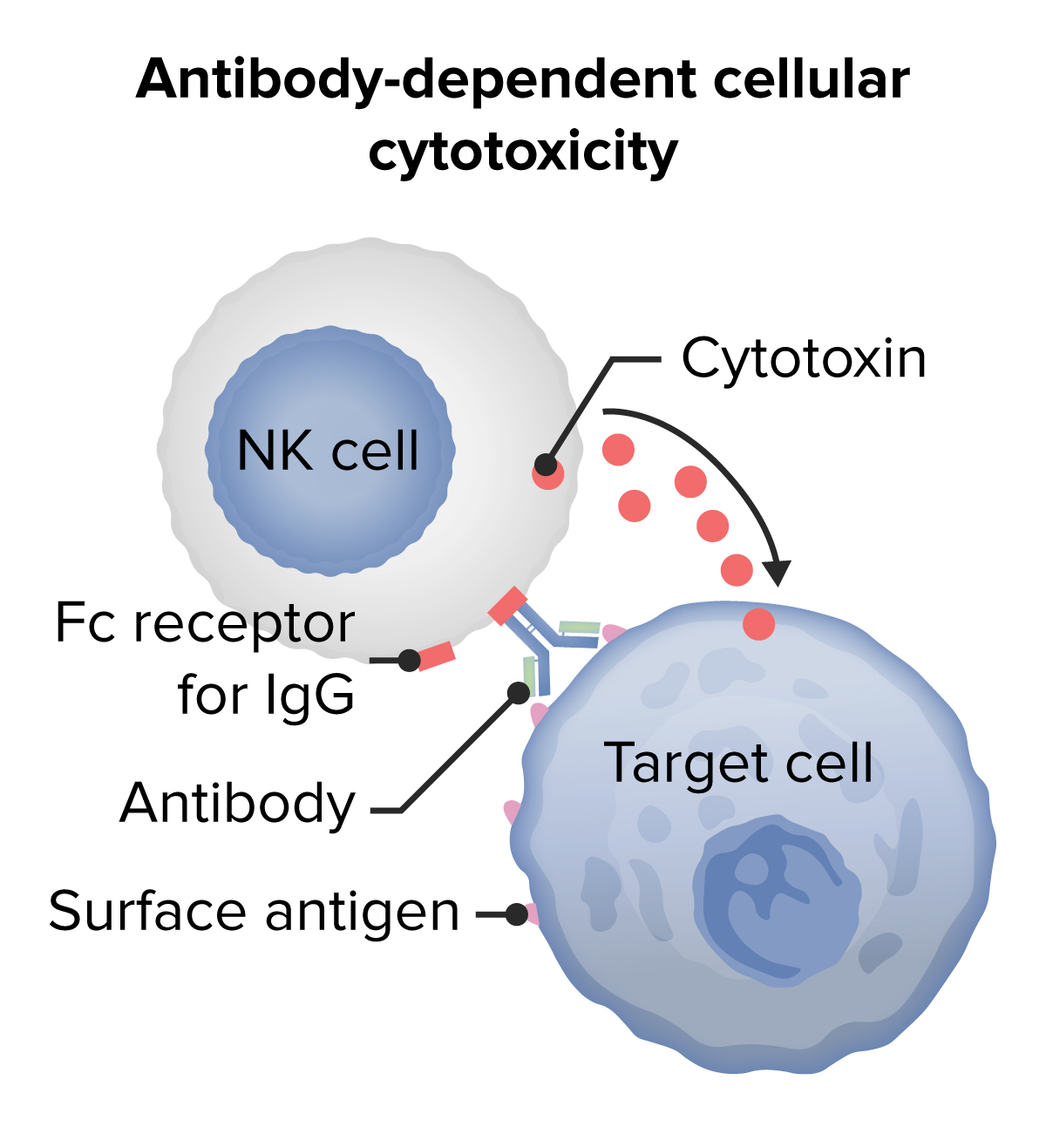
00:01 Type II hypersensitivity is antibody against cell surface antigens. 00:10 Examples of Type II hypersensitivity include transfusion reactions, autoimmune hemolytic anemia, pernicious anemia, hemolytic disease of the fetus and newborn, autoimmune thrombocytopenic purpura, acute rheumatic fever, Goodpasture syndrome, Graves’ disease and myasthenia gravis. 00:47 There are antibody-mediated effects due to these antibodies binding to cell surfaces. 00:55 This can lead to complement activation, and the activation of other white blood cells, in other words leukocytes. 01:04 This results in complement and Fc receptor-mediated inflammation and tissue injury. 01:13 Alternatively, antibody-mediated effects may result in abnormal physiological responses without cell and tissue injury. Two examples are given here. 01:26 On the left hand side, we can see in an individual with a particular type of autoimmune disease called thyroid autoimmune disease, more specifically Graves’ disease which is one type of thyroid autoimmune disease. 01:41 That this individual has antibodies that are binding to the thyroid stimulating hormone receptor, the TSH receptor that is present on the surface of thyroid epithelial cells. 01:54 These autoantibodies, because they are antibodies being produced against a self antigen, the TSH receptor. 02:02 These antibodies stimulate the receptor without needing any TSH there at all. 02:07 So there’s a constant stimulation of the thyroid gland and an overproduction of thyroid hormone. 02:13 On the right hand side of the slide, you will see that in this case there are autoantibodies to the acetylcholine receptor, which is found at the neuromuscular junction. 02:26 And these autoantibodies result in another type of autoimmune disease, in this instance myasthenia gravis. 02:34 The antibody inhibits binding of the ligand to the receptor, in other words it gets in the way of acetylcholine binding to its receptor. 02:43 And it also has a secondary effect of causing the loss of receptors from the surface of the muscle cell. 02:56 A very familiar situation to all of you I’m sure, is the fact that if you need a blood transfusion, you need cross matching of the blood between the donor and the recipient. 03:07 And the most important blood group antigens for eliminating transfusion reactions is to make sure you cross match the ABO group of antigens. 03:20 As you can see in this diagram, these antigens are based upon sugars that are linked together. 03:28 The sugar N-acetyl-galactosamine, N-acetyl-glucosamine, fucose and galactose. 03:35 Individuals that are blood group A have on the surface of their red blood cells, the A antigen. 03:44 They produce antibodies, natural antibodies, ones that occur in the absence of any obvious stimulation, against blood group B antigen. 03:55 So why do they do this? Well it seems that there are many common microorganisms that we come across that have structures that are rather similar and we produce these natural antibodies. 04:05 So because they are Type A, in other words have A antigen; that’s a self antigen, they’re tolerant to that antigen, but they’re not tolerant to B. 04:15 So they make anti-B. 04:17 In contrast, people who are blood group B will have the B antigen on their surface and they will make anti-A antibody. 04:28 Individuals that are group AB possess both A and B antigen, and therefore they don’t produce any antibodies against either A or B. 04:37 In contrast, individuals that are group O, lack both A and B antigens, and therefore they produce these natural antibodies that have the potential to recognize both the A and the B blood group antigen. 04:52 So in a transfusion reaction, if an individual is blood group A or O and therefore will possess anti-B antibodies. 05:01 If they are given blood of either type B or of type AB which will have both the A and also importantly in this case the B antigen, they will cause lysis. 05:14 The IgM antibodies will cause lysis of the transfused erythrocytes. 05:20 And likewise, an individual that is blood group B or O will have anti-A and therefore would lyse any transfused cells from a blood group A or blood group AB donor. 05:40 There’s a condition that is referred to as hemolytic disease of the fetus and newborn (Rhesus D incompatibility). 05:49 This is a situation where a mother is Rhesus D negative. 05:54 She lacks the Rhesus D blood group antigen. 05:57 But her partner is Rhesus D positive, and therefore the fetus has Rhesus D positive red blood cells. 06:07 This is not a problem whatsoever during the first pregnancy. 06:11 There is very little mixing of the fetal and maternal circulation, and there is not a substantial amount of fetal red cells that enter the maternal circulation. 06:24 However, during labour there is a significant release of fetal red blood cells into the maternal circulation during the rupture of the placental membranes. 06:37 Therefore, the mother gets exposed to these Rhesus D red blood cells which to her are foreign antigens. 06:46 She’s Rhesus D negative, so she recognizes Rhesus D as being foreign and makes an antibody response. 06:56 And the fetal red cells survive long enough to elicit this IgG antibody response. 07:03 Now, this is also not a problem at this point in time. 07:06 The baby has been born, so it’s now not exposed to the maternal antibodies. 07:11 And the mother herself is Rhesus D negative, so it doesn’t really matter that she’s got some anti Rhesus D antibody. 07:19 However, a significant problem arises, is if during a subsequent pregnancy the partner is again Rhesus D positive, because now the maternal anti-D antibodies can cross the placenta. 07:34 Because one of the things that the mother does of benefit to her fetus, is to transfer IgG antibodies across the placenta. 07:43 Most of those antibodies will be good antibodies against common pathogens that the mother is coming across. 07:48 And obviously any pathogens that the fetus will come across have gone through the mom. 07:52 So this will be really useful antibodies. 07:54 But in this instance, amongst all those good antibodies, there are also some bad antibodies, in respect that these are anti-Rhesus D and the infant is Rhesus D positive. 08:05 And these transferred antibodies will attack the fetal red cells, leading to the development of this condition called hemolytic disease of the fetus and newborn. 08:15 However, this condition is now incredibly rare because of a very simple procedure which is Rhesus D prophylaxis. 08:24 So at 28 weeks of pregnancy, and then again within 72 hours of birth, the mother is injected with antibodies against Rhesus D which is essentially cover up the Rhesus D antigen on the fetal erythrocytes so that they are not able to sensitize the mother’s immune system.
About the Lecture
The lecture Hypersensitivity: Type 2 by Peter Delves, PhD is from the course Hypersensitivity and Autoimmune Disease. It contains the following chapters:
- A Closer Look on Type II Hypersensitivity
- The Role of Antibodies in Type II Hypersensitivity
- The Transfusion Reactions
- Hemolytic Disease of the Fetus and Newborn
Included Quiz Questions
What is an example of a type II hypersensitivity reaction?
- Autoimmune hemolytic anemia
- Systemic lupus erythematosus
- Eczema
- Tuberculin sensitivity
- Asthma
Which of the following options best describes the mechanism behind the hypersensitivity reaction of Graves disease?
- Autoantibodies stimulate the thyroid stimulating hormone (TSH) receptor to overproduce thyroid hormones.
- Autoantibodies inhibit the thyroid stimulating hormone (TSH) receptor from producing thyroid hormones.
- Downregulation of thyroid stimulating hormone (TSH) receptors leads to hypothyroidism.
- Autoimmune destruction of the thyroid gland leads to spillage of thyroid hormones into the circulation.
- Autoimmune upregulation of thyroid stimulating hormone (TSH) receptors leads to hyperthyroidism.
A person with group A red blood cells has which of the following antigens and antibodies, respectively?
- Type A, anti-B
- Type A, anti-A and anti-B
- Type B, anti-A
- Type AB, anti-A
- Type B, anti-A and anti-B
Which of the following blood groups have neither anti-A or anti-B antibodies?
- AB
- A
- B
- O
- A and B
How does Rh sensitization occur during pregnancy?
- Mother's Rh-negative blood is exposed to the fetus's Rh-positive blood.
- Fetus's Rh-positive blood is exposed to the mother's Rh-negative blood.
- Mother's Rh-positive blood is exposed to the fetus's Rh-negative blood.
- Fetus's Rh-negative blood is exposed to the mother's Rh-positive blood.
- Mother's Rh-negative blood is exposed to the fetus's Rh-negative blood.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |