Playlist
Show Playlist
Hide Playlist
T-Cell Deficiency, Severe Combined Immunodeficiency (SCID), Ataxia-Telangiectasia, Wiskott–Aldrich syndrome (WAS) & DiGeorge Syndrome
-
Slides T-Cell Deficiencies.pdf
-
Download Lecture Overview
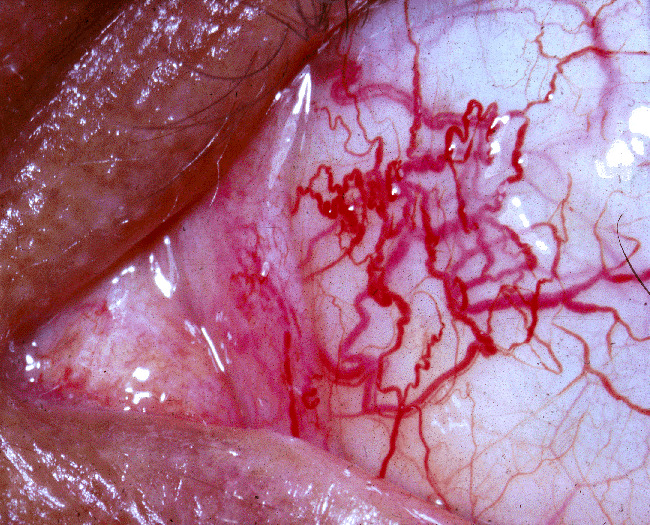
00:01 In this lecture, we'll discuss congenital T-cell deficiencies and combined immune deficiency disorders. 00:09 Let's review what the T-cell is doing. 00:11 The T-cell is responsible for destroying intracellular and other invading organisms like bacteria, viruses, fungi, or parasites. 00:22 The T-cell also facilitates the B-cell in producing antibodies. 00:26 Therefore, patients with T-cell disorders may have humeral deficiency as well. 00:33 So, let's do some examples. 00:35 In kids, we may see severe combined immune deficiency or SCID. 00:40 This is what the bubble boy had. 00:42 We may see patients with ataxia telangiectasia, and we'll talk about that. 00:47 We'll also talk about Wiskott-Aldrich syndrome. 00:51 Lastly, we'll close with a discussion of DiGeorge syndrome. 00:54 These are all examples of T-cell disorders in children. 00:58 So, typically in children with defunct T-cells, they'll present relatively early in childhood, between 2 and 6 months of age. 01:06 Usually, they present with severe infections from organisms that don't cause severe disease in those with a normal immune system. 01:15 An example would be, a severe infection with a common virus, or a severe infection with yeast, such as oral candidiasis that's gone horribly wrong, or mycobacterium, or pneumocystis. 01:30 Pneumocystis is a common presentation for patients with no T-cells. 01:35 So, how do we diagnose a T-cell disorder? Well, we may see a reduced lymphocyte count on CBC, or on the CBC smear, we may notice abnormal morphology of the T-cells. 01:50 But if we wish to make the diagnosis definitively, we order a flow cytometry. 01:55 The flow cytometry will demonstrate deficiency of T-cell subpopulations or of all T-cells, depending on the problem. 02:03 A chest x-ray may also reveal something in that in children with an absent thymus, which we can't see on the x-ray, those children may have a T-cell deficiency. 02:15 So, let's think about how patients present when they have T-cell deficiencies. 02:21 We'll start with SCID or severe combined immunodefiecency. 02:26 These patients have a severe defeciency of both B-cells and T-cells. 02:32 They are extremely susceptible to infection in infancy and it's very rare that we would fail to make the diagnosis relatively quickly. 02:41 They often will have failure to thrive. 02:44 The way we're going to treat these children is by treating with a bone marrow transplant before the age of 3 months. 02:51 We need to avoid public exposure prior to transplant. 02:55 In general, with these patients, when they have so few T-cells, we will provide them PJP prophylaxis with trimethoprim/sulfamethoxazole usually starting at about a month of age. 03:07 These patients may recieve IVIG infusions because IVIG persists for maybe 3 weeks, and this will replace their missing immunoglobulin from their B-cell dysfunction as a result of their T-cell deficiency. 03:25 In patients with ataxia telangiectasia, they may present slightly differently. 03:32 These patients have a combined deficiency of T-cells, immunoglobulins, and they also have neurocutaneous findings from which you can easily make the diagnosis. 03:43 The defect of ataxia telangiectasia is in a gene called the ataxia telangiectasia mutated protein or ATM. 03:52 This protein facilitates DNA repair, and as a result, these patients are at risk for immunodeficiency. 04:01 Generally, they will present with truncal ataxia by the age of 2 years of age. 04:06 Very quickly, they will be wheelchair-bound, usually by school age. 04:11 What's key is we can see eye telangiectasias typically by the age of 5. 04:17 This is an example of an eye telangiectasia. 04:20 It's a very complex bed of capillaries. 04:23 They then get skin telangiectasias as well usually by the age of 7. 04:29 These patients will have immunoglobulin deficiency and infections are common, especially sinopulmonary infections. 04:37 They can also develop leukemia or lymphoma which occurs in about 10% of patients. 04:44 Remember, they can't do DNA repair, so they're likely to develop cancer. 04:49 Let's switch to Wiskott-Aldrich Syndrome. 04:53 This is an X-linked recessive trait. 04:56 So, the boys are much more likely to be affected and an infected girl is usually just a carrier. 05:04 This is a defect in the function of T-cells and B-cells and also platelets. 05:10 It's usually diagnosed in the first year of life. 05:13 20% can go on to develop lymphoma and 40% have autoimmune disease. 05:19 Most of these patients sadly die before they are 10 years old. 05:24 In Wiskott-Aldrich syndrome, about 30% of them or a third have the classic triad, which is eczema, thrombocytopenia, and chronic otitis media, which is an example of an infection they can get as a result of immunodeficiency. 05:41 But they also get all kinds of other infections because of their immunoglobulin deficiency, sinopulmonary infections, for instance pneumonia or sinusitis, and they are in risk for viral and other opportunistic infections as well. 05:56 Lastly, let's touch on DiGeorge syndrome. 05:59 DiGeorge syndrome is another disease where patients can get T-cell deficiency, but they don't always have it. 06:07 There's a wide variety of severity of DiGeorge syndrome. 06:11 It's a deletion of 22q11 and these patients will have midline defects. 06:17 It affects the brain. They may have effects on the face such as hypertelorism or a cleft palate. 06:22 It affects their midline glands like their thyroid or their parathyroid, but it can also affect their thymus. 06:30 And the thymus is responsible for making T-cells, so they may have a T-cell defeciency. 06:35 Keep in mind, they can also have heart findings and multiple congenital heart defects are possible. 06:42 So, patients with DiGeorge who have their thymus involved will develop thymus aplasia resulting in a T-cell defeciency. 06:51 It's highly variable how severe their disease is, and we generally provide supportive care. 06:58 So that's my summary of the congenital reasons why children might have T-cell deficiency. 07:03 Thanks for your attention.
About the Lecture
The lecture T-Cell Deficiency, Severe Combined Immunodeficiency (SCID), Ataxia-Telangiectasia, Wiskott–Aldrich syndrome (WAS) & DiGeorge Syndrome by Brian Alverson, MD is from the course Pediatric Allergy and Immunology. It contains the following chapters:
- T-Cell Disorders
- Severe Combined Immunodeficiency (SCID)
- Ataxia Telangiectasia
- Wiskott-Aldrich Syndrome
- DiGeorge Syndrome
Included Quiz Questions
Which of the following is true about ataxia telangiectasia?
- Immunoglobulin deficiency is common.
- The majority develop leukemia.
- Eye telangiectasias are universally present at birth.
- Patients with this disorder typically are never able to walk.
- The defect is in a gene affecting telomere function.
Blood and sputum cultures from septic child are sent to the lab. Which of the following organisms, if identified, will most likely indicate immunodeficiency in this patient?
- Pneumocystis jirovecii
- Escherichia coli
- Staphylococcus aureus
- Haemophilus influenzae
- Streptococcus pneumoniae
Which of the following is not a T-cell disorder in children?
- Fanconi syndrome
- Ataxia telangiectasia
- Wiskott-Aldrich syndrome
- DiGeorge syndrome
- Severe combined immune deficiency
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
2 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
Very precise and lecturer makes the subject interesting! Thank you so much!
Great lecture as usual. The teacher succeeds in simplifying a complex topic for the practicing generalist pediatrician.