Playlist
Show Playlist
Hide Playlist
Role of Water – Biological Interactions
-
Slides 10 Chemistry Advanced Le Gresley.pdf
-
Download Lecture Overview
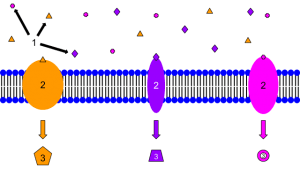
00:01 Now, let's bring on the role of water. 00:04 Any system will adopt the lowest energy configuration. 00:08 This is to do with enthalpy and it's also to do with entropy. 00:13 In chemical systems, this means that the participant will form as many bonds as possible of the strongest type. 00:19 The more bonds it can form of the strongest type, the more stable that system will be. 00:24 For example, if we look at water molecules hydrogen bonding with each other, let's say in ice, each molecule can make it possible for hydrogen bonds. 00:34 In liquid water, however, hydrogen bonds are continuously forming and breaking. 00:40 On average, each molecule makes around 3.4 hydrogen bonds. 00:46 So, let's have a look and see what that is. 00:49 If you look at the hydrogen-bonded cluster on the left-hand side, you can see that this is where the average is derived from. 00:56 At any one time, there will be highly bonded hydrogen-bonded clusters, ice-like ones, and areas with very few hydrogen-bonded clusters. 01:06 And therefore, you can see we have free molecules there. 01:10 Now, let's have a look at what this means in terms of ion solvation. 01:16 This doesn't just relate to the solvation of ion such as those we discussed in Module Two, but also indeed any charged species, be it organic or inorganic. 01:25 When water dissolves and solvates other polar molecules, remember, like dissolves like, a shell of water is formed around the polar molecules, which prevents them from interacting with each other. 01:37 This enables the water and also the drug to achieve its lowest energy configuration. 01:43 Now, let's have a look at solvent effects. Life on Earth uses water as a solvent, which is good in many respects as it dissolves polar drugs, and also biomolecules such as proteins and DNA will also dissolve. 01:59 However, the bad point between this is that interactions between the molecules are weakened as a result of the water effectively getting in the way. 02:08 To understand how this problem is overcome, we must first consider the interactions of nonpolar molecules in water. 02:15 And that's what we're gonna do in the diagram on the next slide. 02:18 So, let's consider a drop of oil and water. 02:22 I'm sure you've all done this, either by accident or by design. 02:25 Vegetable oil and water doesn't -- actually, is not miscible, it is immiscible. 02:30 And therefore, it exists as an emulsion. 02:33 When you have a small amount, it exists as a drop, usually forming on the surface. 02:38 The oil in this case is represented by the yellowy orange circle, and the water, obviously, in blue. 02:44 So, as we add a drop of water to the system -- of oil to the system, what happens is that the water itself cannot hydrogen bond with the oil. 02:52 So, whereas with, let's say for the sake of argument, ethanol, is equally dispersed throughout the water, because hydrogen bonding is possible between alcohol and water. 03:02 In the case of an oil, it tends to preferentially bind to itself. 03:07 Now, ostensibly, you may think this is odd. 03:09 Why would something not bind to itself when there are hydrogen bonds available to it? The point is that fats themselves are highly lipophilic. 03:17 They're not capable of bonding in a hydrogen bonding fashion they are only capable of bonding in a Van der Waals fashion. 03:24 And as we've said, Van der Waals forces are very, very weak. 03:28 And so, that which holds them together is only the Van der Waals. 03:31 And what happens is the water tries to adopt the best possible configuration around this oil droplet, forming a water cage. 03:40 Okay, so let's consider the thermodynamics in this system. 03:44 The energy of the system as a whole goes up because there are fewer hydrogen bonds, and as a consequence, disorder decreases. 03:53 I refer you to the Gibbs free energy equation, which you can look up. 03:57 Entropy decreases when you have a more ordered system. 04:02 Entropy increases when you have greater disorder. 04:06 Now, let us consider two oil drops. 04:09 They will sit in two water cages, so the energy in the system is even higher. 04:15 The energy can be decreased by merging the drops into a larger one, and doing so releases some of the ordered molecules back into solution. 04:25 The water squeezes the nonpolar molecules together, but they do not have a strong affinity for each other, but this is the thing I want to get back to, water does for itself, being capable as we know, of hydrogen bonding to itself. 04:40 So, let's see what this means in terms of a drug and a potential receptor. 04:44 We have here the drug shown on the left hand-side in red. 04:47 Around it, we have ordered water molecules, regions of structured water. 04:53 As the structure of water is displaced from the binding side and also from the region of which the drug binds. 05:01 So, this actually results in a decrease in energy when you're dealing with something which is lipophilic as a receptor, and also relatively lipophilic as a drug. 05:11 So, this obviously decreases the energy available to it.
About the Lecture
The lecture Role of Water – Biological Interactions by Adam Le Gresley, PhD is from the course Medical Chemistry.
Included Quiz Questions
What happens when a polar compound is dissolved in water?
- The formation of water shells around each molecule of compound prevents their interactions with each other and enables them to achieve the lowest energy configuration.
- The formation of water shells around each molecule of compound prevents their interactions with each other and allows them to reach the highest energy configuration.
- The formation of water shells around each molecule of compound helps their interactions with each other and allows them to reach the highest energy configuration.
- The formation of compound molecule shells around each water molecule prevents their interactions with each other and allows them to gain the lowest energy configuration.
- The formation of compound molecule shells around each water molecule helps their interactions with each other and allows them to gain the maximum energy configuration.
Complete the following statement. The stability of a system is ...
- ... directly related to the number and strength of the bonds.
- ... indirectly related to the number and strength of the bonds.
- ... directly related to the strength of bonds, but inversely proportional to the number of bonds.
- ... independent of the number and strength of bonds.
- ... directly related to the number of bonds, but inversely proportional to the bond strengths.
With an increase in the entropy of the system, the system does what?
- Becomes more disordered.
- Becomes more ordered.
- Remained unaffected.
- Gives a highly disordered pattern with evenly distributed patches of ordered symmetry.
- Gives a highly ordered pattern with evenly distributed patches of disordered symmetry.
What happens when a lipophilic drug interacts with a lipophilic receptor?
- The energy of the system drops down.
- The energy of the system shoots up.
- The water molecules get condensed around the drug-receptor complex.
- The water molecules facilitate the hydrogen bonding between the drug and receptor molecules.
- The water molecules provide hydrophilic nature to the drug molecule.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |