Playlist
Show Playlist
Hide Playlist
Review of [H⁺] and pH
-
Slides 07 pHBuffers AcidBaseBalance GeneralPhysiology.pdf
-
Download Lecture Overview
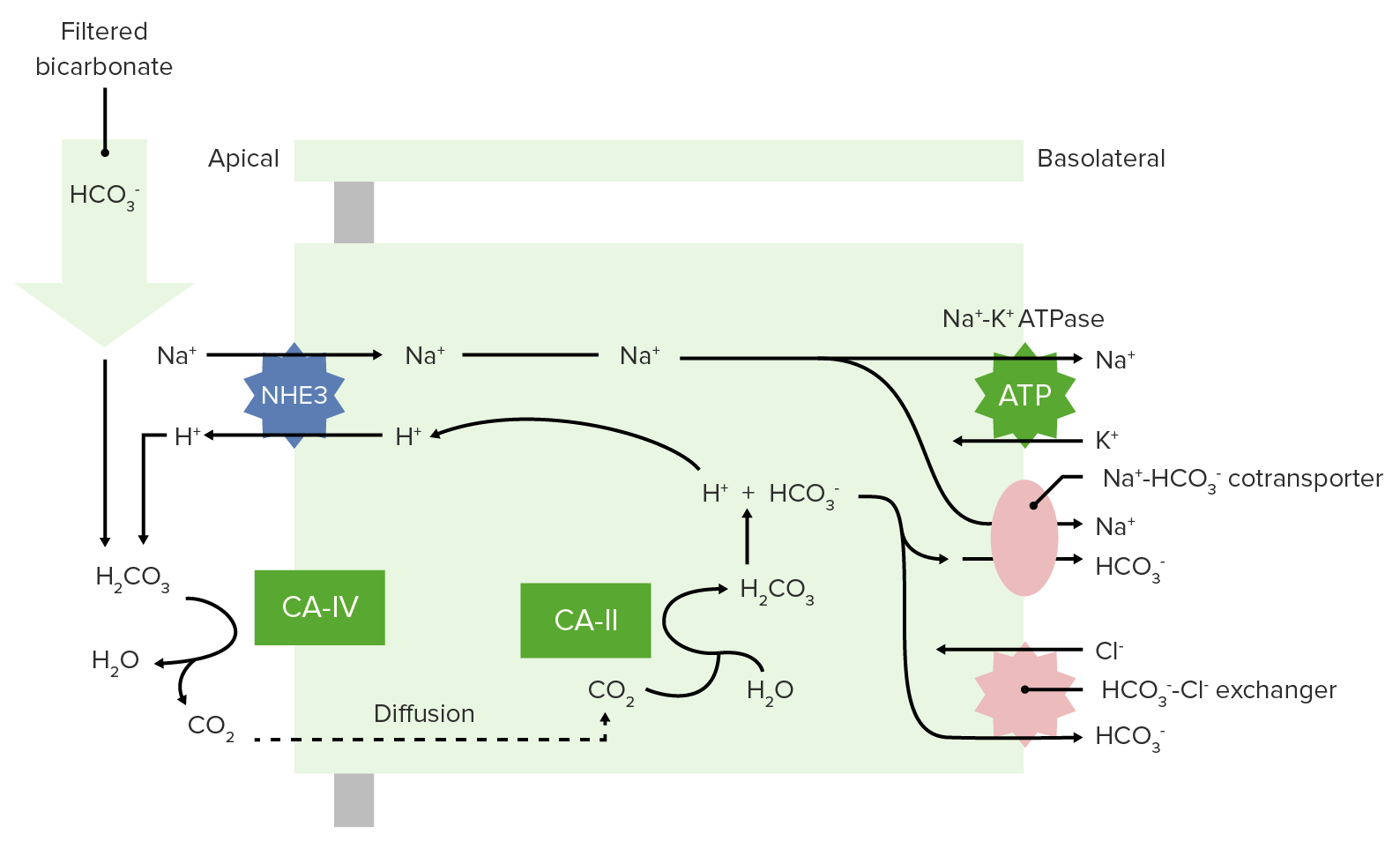
00:00 Acid- Based Balance Fundamentals of pH Acids, Bases and Buffers We are to go through and review a lot of things that you probably are familiar with. 00:13 But we wanna make sure that everyone is on the same page. 00:16 And this will allow us then to delve in deeper to acid base balance in the body if we review some Chemistry basics. 00:24 So, you remember that the pH is a logged rhythmic scale. 00:28 So, when you have an average or normal arterial blood pressure pH of 7,4, it really is a very small concentration of hydrogen ions. 00:40 You can see how many decimal places you are and that is your concentration of hydrogen ions. 00:47 In terms of the body's ability to change pH, especially in the GI system, you can go all the way from a pH of about 1 or 2 pH for about 8. 01:01 The pH of 1 will be in the stomach. And the pH of 8 will be the secretions of the pancreas. 01:12 So, when you think about pH, it's probably easiest to start off with a pH indicator strip. 01:19 You all use these in Chemistry class at one point. 01:22 You take this, you dip it into a liquid, you pull it out and you see what color came up. 01:28 If you get to the more darker colors, such as a blue, a purple or violet, that is a basic pH. 01:36 If you’re at the red end of the spectrum, that is an acidic pH. 01:43 So, we’re going to utilize this kind of color scheme as we move forward to try to understand, what acid- based balance in the body looks like. 01:51 So, I have a couple memory tricks for you. 01:55 First memory trick, is… oh! That is our acidic gnome. 02:01 Oh! Those are your normal pH gnomes. 02:07 And this final one, that is you basic gnome. 02:11 We’re going to utilize these color schemes as we move forward or utilize our little gnome friends here as examples so you’ll be able to think about as soon as you see one, whether it’s either acidic, a normal pH or basic. 02:30 So, for go through all the different pH examples of the fluids of the body besides the blood. 02:36 Although the blood is the most important and we’ll spend the most time on it. 02:39 Let’s go through other pH’s in the body. 02:43 Gastric secretions I said before, right around a pH of 1, maybe of a little bit less. 02:50 This is because of the hydrogen ion pump, which is known as an H+-K+-ATPase. 02:58 This is what is kicking out hydrogen ions into your stomach, making hydrochloric acid. 03:06 Lysosomes also are acidic. 03:08 These are cells, cellular structures that will breakdown various proteins once they come in contact with this lysosomes. 03:17 Different Chromaffin granules are also very low pH. 03:23 If you think about water at 37°C, it’s pH is 6,81. 03:31 The typical cell, is just a little bit basic. It is about 7,2. 03:39 The range though was pretty wide with any particular cell. 03:45 If you undergo anaerobic metabolism, you’re going to be producing more hydrogen ions and therefore your pH will be lower. 03:52 If metabolism is very, very slow, your pH could rise very slightly. 03:59 Cerebrospinal fluid has a little bit higher pH 7,3, but here the plasma of the blood has a pH of 7,4. 04:09 This is very tightly regulated. 04:12 Some of these other pH’s are less regulated as arterial blood plasma. 04:18 The range is plus or minus (±) 0,05. 04:22 So, it’s going to be somewhere between 7,35 and 7,45. 04:28 The Mitochondrial inner matrix within a cell that is about 7,5. 04:34 And finally our highest pH is in pancreatic secretions. 04:39 These pancreatic secretions are going to put enzymes into the GI tubule. 04:45 This secretions involve a lot of bicarbonate. 04:50 The reason why you have this high bicarbonate secretions are to buffer the pH that’s coming directly from the stomach. 04:57 So, you’re going through from the stomach into the small intestine known as the duodenum. 05:04 These are ductal cells that secrete the bicarbonate. 05:10 and these bicarbonates are secreted as they leave the pancreatic ducts.
About the Lecture
The lecture Review of [H⁺] and pH by Thad Wilson, PhD is from the course Acid-Base Balance.
Included Quiz Questions
Which of the following has the highest hydrogen ion concentration under normal physiological conditions?
- Lysosomes
- Cytosol
- Cerebral spinal fluid
- Arterial plasma
- Mitochondrial inner matrix
What does a pH of 1 mean?
- [H+]=0.1
- [H+]=0.01
- [H+]=1
- [H+]=0.001
- [H+]= 1/0.1
What is the hydrogen ion concentration of pancreatic secretions?
- 0.00000001
- 0.000000008
- 0.00000009
- 0.8
- 8
Where is the hydrogen ion concentration (pH) most tightly regulated?
- In the blood
- In the cerebrospinal fluid
- In the inner mitochondrial matrix
- In the lysosomes
- In the cytosol
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
ok me gusto mucho esta leccion , gracias por hacer lecturio para estudiar