Playlist
Show Playlist
Hide Playlist
Receptors – Biological Interactions
-
Slides 10 Chemistry Advanced Le Gresley.pdf
-
Download Lecture Overview
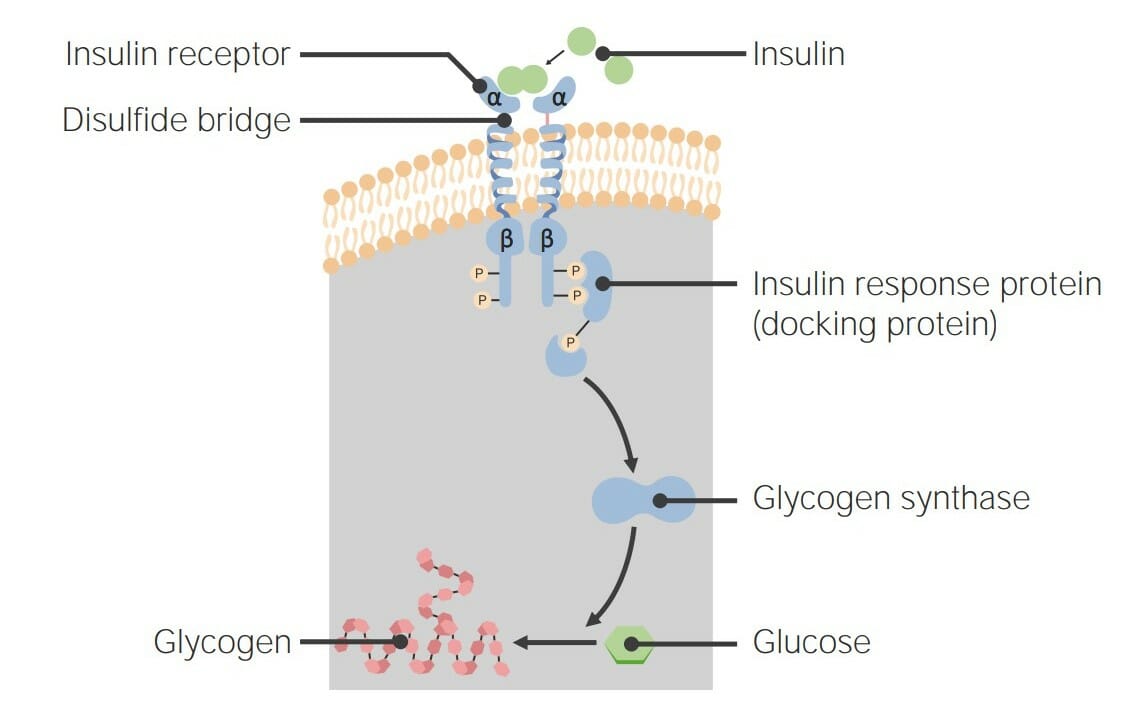
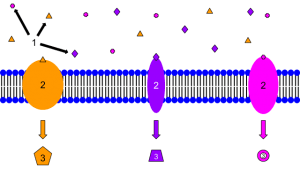
00:01 Now, this apparent contradiction that quaternary ammonium salts were shown to both contract and relax smooth muscle galvanised the idea of a receptor. 00:10 A receptor, in physiological terms, is a site where a drug binds. And then the receptor brings about a physical response. 00:19 In the context of what we were discussing, which is contraction and relaxation, let us consider the cholinergic system. So, this is the idea that we have a receptor which responds by relaxing muscle in its normal state. Let’s have a look at this in terms of our cartoon. 00:38 We have here our drug, which is shown as the yellow circle, and we have our receptor, which is shown here as the green rather hungry pac-man. What’s happening in this scenario is that you have a drug receptor complex. You have selective binding of the drug for the receptor and then this yields an appropriate response. 00:57 This is the idea of receptors and drugs interacting with each other. And it’s pretty basic and straightforward, but it governs a lot of what we observe in terms of the responses we see when we are applying drugs to a given tissue type. 01:13 The drug is a ligand at this receptor and that’s an important term to remember. This is something which is a ligand or ligates at the particular receptor. It is something that binds to it. It is described, in this case, when it results in the production of a response as an agonist. In this case, a muscle relaxant, the receptor was identified by being in the family called the cholinergic receptors, which I mentioned earlier. And a naturally-occurring agonist at this class of receptor is acetylcholine. Hence, the name cholinergic. And indeed, when this was first discovered, it was actually christened by its German name, Vagusstoff. 01:53 The drug was found to contract... were found to contract the muscle were shown to be acting at actually the same receptor. But, of course, they were doing something quite different. 02:03 They were found to be binding to the receptor, but without giving rise to the physical response that was associated with the binding of acetylcholine. And it was hypothesised that what was actually happening, in this case, is that the drug, in here for the sake of clarity represented as the red square, was binding to the receptor and forming a drug-receptor complex. But, this was not yielding a response. The question had to be asked: why? If it’s binding, surely the process of binding results in that response. 02:38 So, the concept of the antagonist was born because the idea was that, because the receptor was actually blocked, acetylcholine itself could not bind and therefore, by not being able to bind, couldn’t actually elicit the appropriate messenger response. Since acetylcholine couldn’t act there, the muscle went into a state of contraction mostly because there are other receptors that actually deal with the sympathetic contraction force. And the cholinergic system is largely concerned with the parasympathetic relaxant force. 03:14 Drugs which block active sites without giving rise to a response are called antagonists. 03:21 Other active sites where drug combined include enzymes. And as we’ll see, an enzyme is something which actually involves the conversion of a molecule into more than one molecule or, sometimes, the bringing together of two molecules to form a third. 03:39 Both receptors and enzymes are made up of proteins. And that’s why I said earlier on in Module III why proteins and polypeptides were so important, as is their understanding. 03:49 Proteins contain a variety of functional groups. So, we have to ignore, for the sake of argument, that it is effectively just a polyamide backbone. Yes, of course, the polyamide or polypeptide primary structure is important. But, it’s the side chains that we saw on the other amino-acids back in Module III which convey on it different types of potential intermolecular interaction. 04:12 We have things like glutamic acid which, of course, as the name suggests, has an acidic extension part coming off the alpha carbon. 04:22 We have neutral species, neutral species, for example, like isoleucine, which is purely aliphatic. We also have basic species like arginine and lysine which, on their side chain, have NH2 and also amidine and guanidinium groups. In addition, we also have the polar, which, for example, could be something like serine, which contains an alcohol group, and non-polar like, for example, valine. So, there’s a whole host of different groups on the side chains which have... can influence the interaction between a drug and a receptor. 05:01 Drugs will form interactions with the functional groups lining the active site. And this is important because in the receptors that we will discuss, there is a bulk of protein and then there is the key active site which contains certain key amino-acids whose interaction through intermolecular forces with our agonist or indeed, with our antagonist, are so crucial for that biological activity. 05:28 Drugs are recognised by their targets by a number of different types of interaction. 05:33 And we’re going to be going through some of these now. 05:36 Drug binding at the same site, but in a different way can give rise to different effects. And this is the difference between agonists and antagonists. You could also get something called a partial agonist. But, we will probably leave that alone for the moment. 05:52 Knowledge of these interactions allows us to change the structure of the drug in order to afford a better interaction. It helps us to work out how a drug binds and it also helps us to design new drugs and predict how they will bind in an ideal world.
About the Lecture
The lecture Receptors – Biological Interactions by Adam Le Gresley, PhD is from the course Medical Chemistry.
Included Quiz Questions
Which of the following can be used as targets in drug therapy?
- Receptors, enzymes and DNA
- Enzymes only
- Receptors and enzymes only
- Enzymes and DNA only
- None of the above
Which of the following statements best describes cholinergic drugs?
- Cholinergic drugs elicit the effects similar to those produced by acetylcholine.
- Cholinergic drugs elicit the production of gastric juices.
- Cholinergic drugs elicit the production of bile juices.
- Cholinergic drugs exhibit an antibiotic effect against prions.
- Cholinergic drugs cause the release of adrenal hormones.
Which of the following is NOT true about acetylcholine?
- When stimulated, vagus nerves can release either a cholinergic agonist or an antagonist, depending on the need.
- Acetylcholine is essential for the functioning of both central nervous system and peripheral nervous system.
- Acetylcholine, an ester of acetic acid and choline, acts as a neurotransmitter.
- Acetylcholine helps in chemical synaptic transmissions.
- D-tubocurarine acts at the same receptor as acetylcholine,
What is the main difference between agonist and antagonist drugs?
- Binding of an agonist to a receptor elicits a biological response, whereas binding of an antagonist to a receptor dampens this response.
- Binding of an agonist to a receptor dampens a biological response, whereas antagonist binding to a receptor elicits this reaction.
- Agonistic drugs act on the RNA of a cell, whereas antagonist drugs act on the DNA.
- Agonistic drugs react with histone proteins of a cell, whereas antagonistic drugs react with phosphodiester bonds of the DNA backbone.
- Agonistic drugs act on the DNA of a cell, whereas antagonist drugs act on the RNA.
Complete the following statement. The knowledge of the interactions between drug molecules and receptors allows us …
- … to modify structures of drug molecules to create effective drugs.
- …to change receptor structures to create effective responses.
- …to immune cells against effects of drug molecules.
- …to minimize the number of interacting groups in the ligand.
- …to maximize the number of carbon atoms in the receptor molecule.
Functional groups present in the molecular structure of drugs do which of the following?
- They determine the water solubility of the drugs, and the interaction between the drugs and the target receptor molecule.
- They determine the water insolubility of the drugs.
- They do not play any crucial role in drug-receptor interactions.
- They react with each other to nullify the drug side-effects.
- They must be removed before the drugs are consumed.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |