Playlist
Show Playlist
Hide Playlist
Peripartum and Arrhythmogenic Cardiomyopathy
-
Slides Cardiomyopathy.pdf
-
Reference List Pathology.pdf
-
Download Lecture Overview
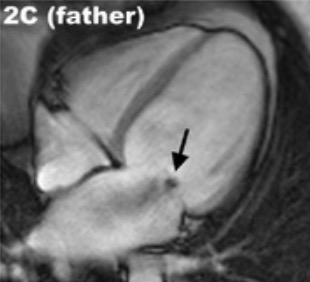
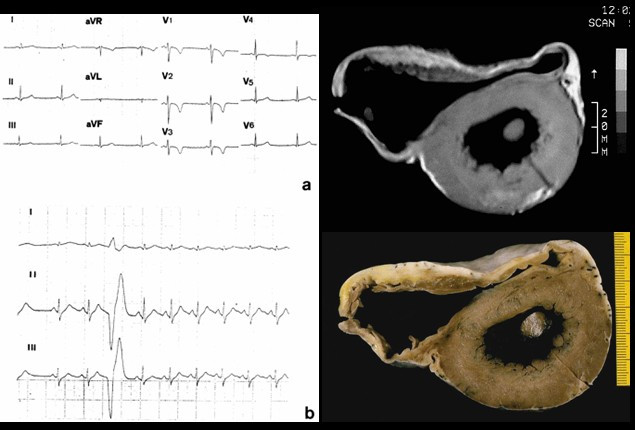
00:01 The rates are highest in the African American population accounting for just a little under 50% of cases but it can occur in any ethnic group. 00:10 Typically occurs in the maternal population that's a little bit older, so 30 to 40 years old, but it can occur in teens and 20 year olds. 00:23 It occurs late in gestation and we'll talk a bit about the the pathogenesis in a minute, or can occur up to several weeks following delivery of the child. 00:32 Half of these patients fortunately, while they will develop a transient peripartum cardiomyopathy, a dilated cardiomyopathy will spontaneously recover. 00:42 So that's the good news. 00:45 What is causing this? So there is hypertension associated with many cases of pregnancy, although this is not the only cause. 00:55 Volume overload in fact, there are significant swings in volume overall, in trying to maintain adequate perfusion of the developing fetus. 01:05 And so that volume overload in the setting of hypertension may feed into the pathophysiology. 01:11 If you have multiple pregnancies, or you have twins or triplets, that increases the overall risk by mechanisms that may be related to what's going on in the placenta, as we'll talk about. 01:26 There are nutritional deficiencies. 01:27 So mothers who do not get adequate antenatal care, do not get adequate vitamin supplementation may end up with a functional beriberi, for example, inadequate thiamine, which can cause a dilated cardiomyopathy. 01:41 For tocolysis, meaning that we are giving drugs to diminish the uterine contraction so we're trying to maintain a pregnancy so the baby can go to term. 01:52 Use of those agents may actually cause a dilated cardiomyopathy because of the effect on the vasculature of the heart. 02:02 You can also have metabolic derangement. 02:04 So metabolic gestational diabetes may also feed into this pathway. 02:10 And then the immunologic responses have been implicated, although there's not a good literature on that, but immune responses potentially related to recognition of foreign HLA from dad's contribution to the fetus may drive some of this response. 02:30 The current thinking is that there is the major driver for peripartum cardiomyopathy is probably due to proteins that are being elaborated from the developing placenta. 02:47 So and also from a best part of the prolactin secretion so we know that prolactin from the posterior pituitary, those cleavage products have a propensity to drive a dilated cardiomyopathy in some animal models. 03:05 The placenta also makes antagonist to vascular endothelial growth factor. 03:11 So up to a certain point, the placenta is very angiogenic, but as we get nearer and nearer and nearer to delivery, the placenta does not want to have more angiogenesis. 03:22 In fact, it wants to stop it so that at delivery, and as we deliver the placenta, the mother doesn't bleed to death. 03:30 So there are placental derived antagonists that inhibit vascular endothelial growth factor particularly late in pregnancy. 03:37 Both of these prolactin cleavage products, and the antagonists, will drive an anti-angiogenic effect overall. 03:46 And the vasculature within the heart is constantly remodeling if we bathe that in anti-angiogenic factors that can lead to impaired angiogenesis in that tissue leading to ischemic injury. 03:58 As they say, these two pathways are thought to be the most likely causes for peripartum cardiomyopathy. 04:06 But whether the others are contributory and many cases, some cases they will be, overall we have diminished myocardial contractility and a dilated cardiomyopathy. 04:17 Arrhythmogenic cardiomyopathy again previously called arrhythmogenic right ventricular cardiomyopathy is one that will be another cause of dilated cardiomyopathy. 04:27 It is a genetic disease of the heart muscle characterized by fibrofatty replacement, classically the right ventricle, but we can also get fibrofatty replacement involving parts of the left ventricle. 04:40 What's going on here? The major presentation is right sided heart failure because the dominant involvement involves the right ventricle. 04:48 And as we are changing the relationship of cardiac myocytes, one to another, we develop significant severe rhythm disturbances hence the name arrhythmogenic cardiomyopathy. 05:01 The main proteins that seem to be involved are those that regulate cell-cell connections. 05:08 So we are looking at a variety of proteins that connect one cardiac myocyte to another cardiac myocyte at the level of the intercalated disc. 05:18 And there are desmocolins, desmogleins at the top, there's a membrane, And then there are these proteins that bridge into the cardiac myocyte that are responsible for maintaining these interactions again at the intercalated disc, and the one in the green box, plakoglobin is probably the most important known mutation that causes a arrythmogenic cardiomyopathy. 05:43 What does it look like? So the right ventricular wall is markedly thin. 05:47 All that yellow, that's fat that used to be kind of that brown tan color, and now it's been replaced with fat. 05:55 Pretty remarkable, because there's just a little tiny sliver of cardiomyocytes that are still within the right ventricular wall. 06:04 That fibrofatty infiltration, there's some fibrous connective tissue really kind of pervades along the entire right ventricular wall, and as they say, can also involve the interventricular septum, as you see down at the bottom of the picture. 06:18 It can also involve focally, the left ventricular wall as well. 06:22 On histology, this looks pretty much like what we thought based on the gross, most of the myocytes have been replaced. 06:30 So kind of the maroon colored cells, those are going to be the myocytes. 06:35 That entire thickness should be myocytes. 06:38 And in fact, it's not, it's been replaced by fibrosis, which is the blue strands, and also by adipose tissue. 06:47 So the cardiac myocytes are dying, because they've lost connection one to another. 06:51 And as they vigorously contract, they tend to pull apart, that's our thinking. 06:56 And when they do that, they undergo apoptosis. 06:58 And then the myocytes or the myocardium, replaces that with scar and fat. 07:05 So the mutations that we do know about involve those desmosomal junctional proteins at the intercalated disc, and particularly plakoglobin. 07:14 There are also some mutations that are known that interact the desmosomes in the intermediate filaments inside the cell. 07:19 So connecting that bridging between the cardiac myocytes to the rest of the cell. 07:28 Desmosomal detachment, particularly during strenuous exercise is kind of the thinking leads to apoptosis. 07:35 As the cells are dying, we are also getting some degree of inflammation. 07:39 That combination of apoptosis and inflammation is going to be clearly myocardial cell death, and then we will get dilation of the ventricles and leading to ventricular arrhythmia and dysfunction. 07:52 That dilation of the ventricle and loss of myocytes is the dysfunction part. 07:57 The arrhythmias is because we've lost connection cell to cell to cell, and that's how we get a normal conduction wave from throughout the ventricle.
About the Lecture
The lecture Peripartum and Arrhythmogenic Cardiomyopathy by Richard Mitchell, MD, PhD is from the course Cardiomyopathy.
Included Quiz Questions
Which of these factors increases the risk of peripartum cardiomyopathy?
- A multiple-gestation pregnancy
- Hypotension
- Asian ethnicity
- Age < 25 years
- Hypovolemia
What pathological finding is seen in a heart with arrhythmogenic cardiomyopathy?
- Fibrofatty replacement of the right ventricular myocardium
- Granulomatous inflammation and scarring
- Constrictive pericarditis
- Rupture of an atherosclerotic plaque
- Subendocardial ischemia
Arrhythmogenic cardiomyopathy is...
- ...an inherited disorder of the heart muscle that leads to rhythm disturbances and right-sided heart failure.
- ...a spontaneous defect in heart muscle that leads to rhythm disturbances and right-sided heart failure.
- ... an inherited disorder of the heart that leads to mitral valve prolapse.
- ...a spontaneous defect in the pericardium that leads to right-sided heart failure.
- ...a spontaneous defect in valvular tissue that leads to left-sided heart failure.
What morphological changes are seen with arrhythmogenic cardiomyopathy?
- Myocyte replacement with fat and thinning of the ventricular wall
- Myocyte replacement with fat and thickening of the ventricular wall
- Granulomatous scarring of the ventricular wall
- Hypertrophy and thickening of the ventricular wall
- Atrophy and thinning of the ventricular wall
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |