Playlist
Show Playlist
Hide Playlist
Pediatric Pyruvate Kinase Deficiency, Autoimmune Hemolytic Anemia (AIHA) & Hereditary Spherocytosis
-
DLM 09-65 Anemia Alverson v011.pdf
-
Download Lecture Overview
00:01 Let’s switch goals now towards another cause of anemia that you should know about and we often think about as a metabolic condition. 00:09 This is pyruvate kinase deficiency. 00:12 It is extremely rare, but it sometimes shows upon test, so let’s chat about it. 00:17 It’s autosomal recessive and basically there is a reduction in the production of pyruvate. 00:23 Because they can’t do glycolysis down to pyruvate, they are having a hard time putting pyruvate into the citric acid cycle. 00:33 And remember, the citric acid cycle is responsible for making ATP. 00:38 Now if you have a deficiency of ATP, it’s the cell without a mitochondria. 00:44 The cell that’s relying on its own endogenous stores, that’s going to be suffering first and that cell is the red blood cell. 00:52 So in patients with pyruvate kinase deficiency, they have an intracellular buildup of sodium. 01:01 These cells then die prematurely and they result in hemolytic anemia. 01:06 Because these patients are having such a rapid breakdown of cells, they often get gallstones where the byproduct of bilirubin is stored up and they can turn into stones. 01:17 The patients also may develop at birth kernicterus. 01:22 For that rapid breakdown, you can see that they get so jaundiced and they get an unconjugated hyperbilirubinemia that can result in kernicterus. 01:32 Furthermore, because they require multiple transfusions, they are at risk for iron overload, so you have to keep an eye on that. 01:40 So let’s switch gears now to an acquired anemia such as autoimmune hemolytic anemia. 01:47 Okay. 01:48 This is a primary problem that’s isolated to the red blood cells. 01:52 Patients are going to have an antibody attack on their red blood cells. 01:58 Alternatively, this could be a secondary problem from a generalized autoimmune condition like lupus or Crohn’s or juvenile idiopathic arthritis. 02:08 Most of the time, it's primary. 02:11 We describe these patients as having warm or cold agglutinins. 02:17 The way I like to think about this is we know that when things are warm, it’s easier for them to react. 02:23 So if I have an IgG with only one FAB region, it better be warm for me to react. 02:31 If I have 5 FAB regions, like an IgM would have, I can react when it’s colder. 02:39 So when you hear cold agglutinins, those are IgMs and when you hear warm agglutinins, those are IgGs. 02:47 It doesn’t really matter so much we’re going to manage this fairly similarly but that’s the distinction. 02:52 How do we make this diagnosis? Well, we’ve talked about it already really, it’s that Coombs test we talked about. 02:58 Okay. 02:59 So you get a Coombs test and the patient has antibodies against their red blood cells. 03:05 If we see that, we’re going to treat with prednisone. 03:09 The IgG mediated autoimmune hemolytic anemia will respond a little bit better to steroids than the IgM will. 03:18 If patients get very bad off, we will need to transfuse and care must be taken to transfuse because we have to transfuse them blood, for which they are not attacking by antibodies and we’ll use the indirect Coombs to figure that out. 03:33 In worst case scenarios, if we can’t find a type of blood that works for them, we may need to do plasmapheresis to try and take out those antibodies. 03:43 Overall, the mortality of this condition is pretty high. 03:47 It’s around 10%, but many patients will recover without too many problems. 03:53 Let’s move on to the hereditary spherocytosis. 03:56 This is a disorder that is in a family of disorders where the problem is the proteins that anchor the red cell membrane into its classic form. 04:06 So there are many different types of these disorders. 04:09 One is hereditary spherocytosis, which is the most common type and you can see it on this slide. 04:16 These cells don’t have their classic pattern. 04:18 They are small spheres. 04:20 You can also get hereditary elliptocytosis. 04:24 This is where the cells form an elliptoid-type pattern. 04:28 You may have hereditary pyropoikilocytosis or South Asian ovalocytosis or hereditary stomatocytosis. 04:38 The point is there are many different kinds, all of which were involved in these proteins on the outer surface of the cell. 04:45 So let’s look a little bit deeper. 04:47 Ankyrin in patients with hereditary spherocytosis is the problem. 04:52 Ankyrin normally holds the red blood cell into that shape that we expect. 04:57 In patients with hereditary spherocytosis, the abnormal cells are cleared prematurely, mostly in the spleen. 05:05 So these patients will have an anemia. 05:09 Here’s an example of that disease and you can see that the cells are not accommodating a normal picture that we would see. 05:16 You can make this diagnosis pretty much on a blood smear. 05:21 Many patients with hereditary spherocytosis are asymptomatic and really require no therapy other than perhaps folate supplementation, because remember since they are making so many cells, they are burning through the problem. 05:35 More severe patients are treated with splenectomy. 05:38 What this does is it reduces that clearance, so that they’re less likely to have severe anemia. 05:44 They may get chronic transfusions and that’s actually why we’re not necessarily supplementing with iron because they may end up iron overloaded from those transfusions. 05:54 We will play each case like it comes. 05:56 Another issue that these patients have, because there are so much hemoglobin turnover, is some may develop gallstones and eventually the need for cholecystectomy. 06:06 This is true of all of our rapid turnover red blood cell anemias. 06:11 So that’s a summary as it were of most of the types of anemia that we see in children and how we break them down and how we make that diagnosis. 06:21 Thanks for your time.
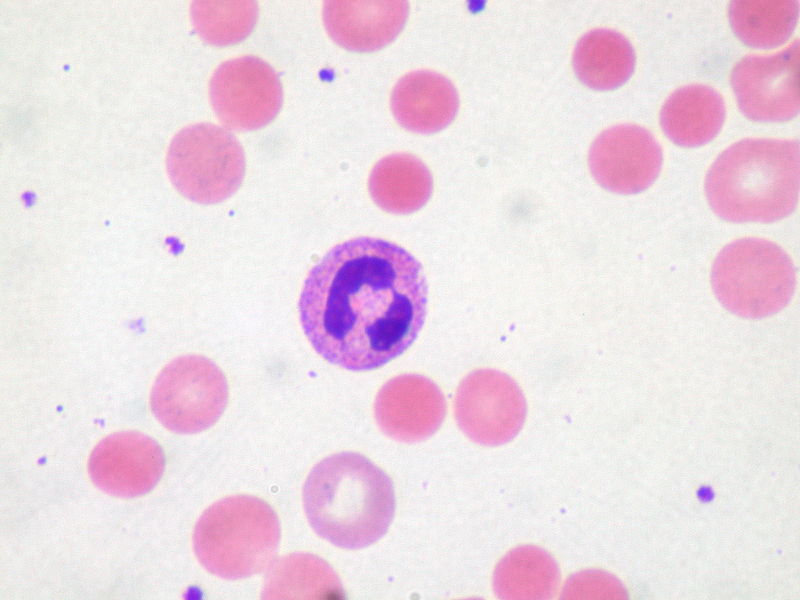
About the Lecture
The lecture Pediatric Pyruvate Kinase Deficiency, Autoimmune Hemolytic Anemia (AIHA) & Hereditary Spherocytosis by Brian Alverson, MD is from the course Pediatric Hematology. It contains the following chapters:
- Pyruvate Kinase Deficiency
- Autoimmune Hemolytic Anemia
- Hereditary Spherocytosis
Included Quiz Questions
Which of the following is often seen in patients with pyruvate kinase deficiency?
- Gallstones
- Iron deficiency anemia
- No jaundice at birth
- Agenesis of the corpus callosum
- Eczema
Which of the following is a complication of pyruvate kinase deficiency?
- Kernicterus
- Iron deficiency anemia
- Congenital heart disease
- Acute myeloid leukemia
- Renal failure
Which of the following is the most appropriate initial test when autoimmune hemolytic anemia is suspected in a patient with acute symptomatic anemia?
- Coombs test
- Indirect Coombs test
- Direct bilirubin
- Serum iron
- Serum lactate dehydrogenase
Which of the following is NOT indicated in the treatment of autoimmune hemolytic anemia?
- Platelet transfusion
- Packed red blood cell transfusion
- Plasmaphoresis
- Prednisone
- Spleenectomy
Customer reviews
4,0 of 5 stars
| 5 Stars |
|
0 |
| 4 Stars |
|
1 |
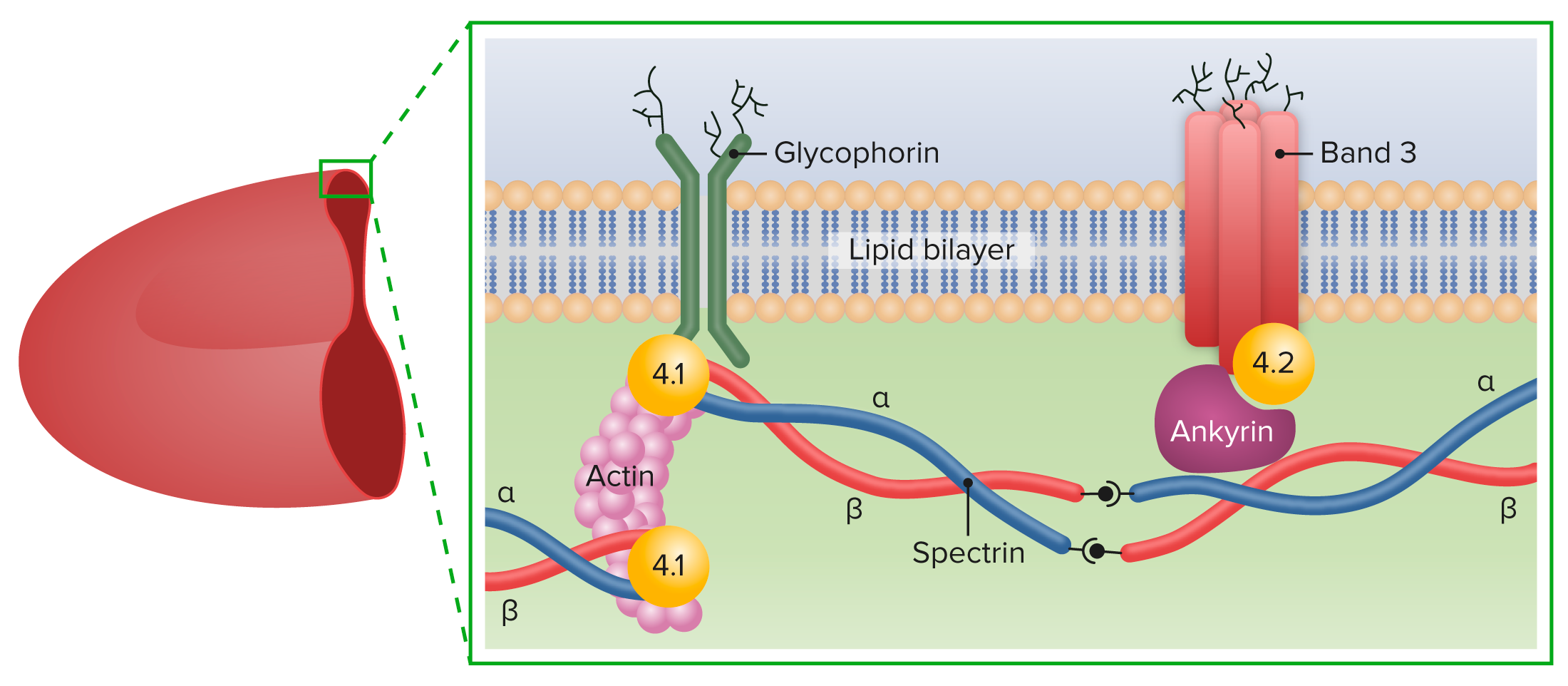
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
1 customer review without text
1 user review without text