Playlist
Show Playlist
Hide Playlist
Neutrophil Chemotaxis
-
Slides Acute and Chronic Inflammation Cellular response.pdf
-
Reference List Pathology.pdf
-
Download Lecture Overview
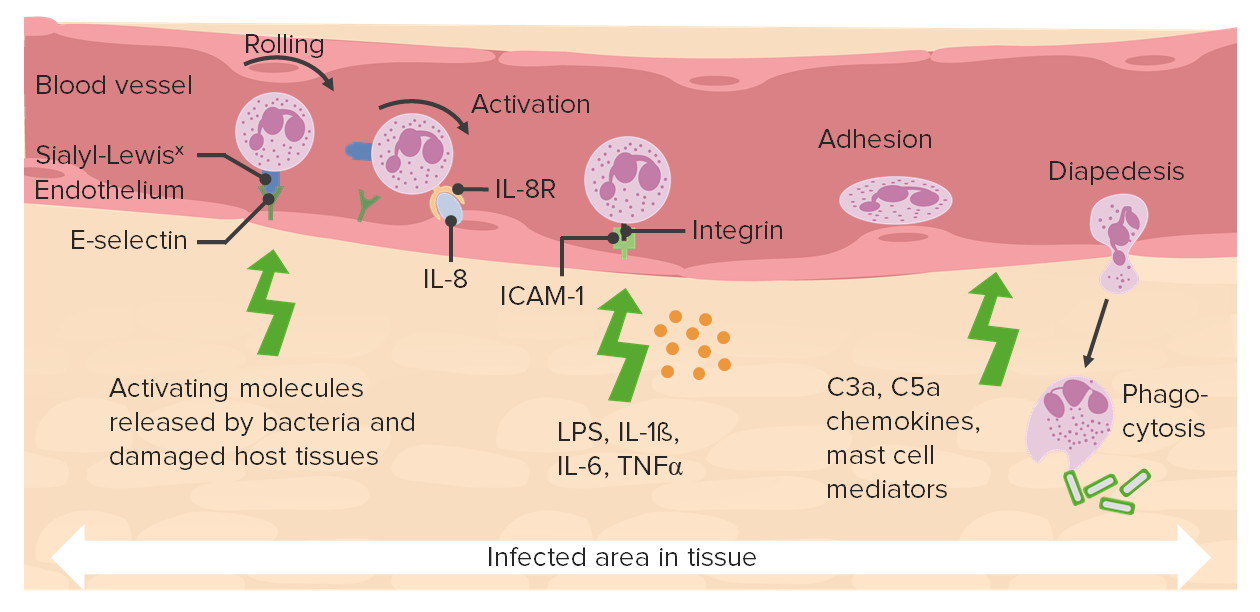
00:01 So how do we chemotax, specifically to the area where the bugs are? One of the major things that's used, particularly for bacterial infections, is N-formyl methionine. 00:13 An N-formyl methionine is the first amino acid attached to the beginning of every bacterial protein. 00:20 If the neutrophil sees N-formyl methionine, it knows, "There's a bacteria here someplace. And I'm gonna go chase after it." Leukotrienes these are part of those eicosanoids. 00:31 So leukotrienes will also be elaborated by the inflammatory cells that are sitting in the extravascular space, that's also very profoundly chemotactic and we'll say, "Come here." Complement fragments. 00:44 Activation of the cascade, that's part of innate immunity, the proteins that are going to be important for forming pores will also make peptide fragments along the way that are going to be very intensely chemotactic. 01:00 Platelet-activating factor. 01:03 So it was originally described as something that activated platelets, but it has other effects, including prominent chemotaxis. 01:11 And it's made by a variety of inflammatory cells. 01:15 And then just other chemokines. 01:17 So the CXC chemokines. Remember those, CXC. 01:21 Those seem to be the ones that will recruit neutrophils, and so they will also be involved in this chemotaxis. 01:26 This moving to find where the action is happening. 01:31 An important point about chemokines and the receptors. 01:35 These are actually really poor targets for therapy because a single chemokine, shown here on the right hand side, can bind to multiple different chemokine receptors. 01:50 Moreover, any individual receptor can bind multiple different chemokines. 01:58 So the interactions are incredibly redundant and they're incredibly promiscuous. 02:02 Meaning if I've just blocked one, it's probably going to do an end around using a different receptor or a different chemokine. 02:09 So these are not useful targets for therapy. 02:14 But it's also impressive that we make so many of them and this is important for probably some degree of specificity in any particular circumstance. 02:24 Okay, as promised, here's a way to think about chemokine functions. 02:29 And if you take this away, you're good for at least the chemokines. 02:33 At very low dose, they activate the leukocyte integrins Remember, we went from the closed configuration to the open configuration. 02:40 When that happens, now you get firm adhesion. 02:44 So low dose chemokines do that on the neutrophil. 02:47 At medium dose, they are that with of perfume in the air, attracting the leukocyte to move out of the bloodstream. 02:57 And at very high dose, they're now going to activate antimicrobial functions that we'll talk about in the next part of this topic discussion. 03:06 Okay, so keep those in mind. 03:10 After it migrates, what does the neutrophil recognize? How does it know that there isa foe out there? And how does it know to just attack that foe and not normal tissues. 03:22 So there are a number of so called pathogen associated molecular patterns. 03:26 These are called PAMPs. Whatever... 03:30 Not Pampers... PAMPs So the pathogen associated molecular patterns include things like Double-stranded RNA An unmethylated CpG islands, so thats cytosine phosphorylated, wanting islands of DNA, these are things that bacteria make, and we don't make. 03:49 we as human beings, as mammals we don't make. 03:53 So if we see these double-stranded RNA or those islands of CpG DNA that says that there is a bug out there. 04:00 And that allows the neutrophil to have some specificity and what it's recognizing and going to kill Lipopolysaccharides (LPS) a very important constituent of the bacterial cell wall. 04:13 And it's important driving septic shock, but it's also an important recognition motif of pathogen associated molecular pattern. 04:20 Mannose is also recognized. 04:22 And you would say, "Well, there's mannose and all kinds of glycoproteins and glycolipids, and mannose." Yes, but we never put it on as the last sugar. 04:30 So, if we have mannose is the last sugar that's almost always a bug. 04:35 And phosphorylcholine, another one of the phospholipids. 04:38 when there is high density of that, and we can see them, that's usually a signal. 04:43 So we have a variety of things. 04:44 We can also make microorganisms pathogens tasty. 04:50 That's a process of opsonization And opsonins are nothing but proteins that will bind to a particular pathogen and make them tasty. 05:01 So opsonins include antibody and complement fragments. 05:06 So those are the kinds of things that we can recognize, that says, "Here's the foe. Attack it." And then we have specific receptors. 05:15 I'm not going to go into these details, but you will hear about them. 05:18 And some of these are important for different kinds of pathogens, but we have all kinds of receptors that can recognize these pathogen associated molecular patterns. 05:26 And we also have immune receptors that can recognize antibody. 05:31 So antibody, when it binds undergoes a conformational change. 05:35 And the FC portion of the antibody now is able to bind to specific receptors on neutrophils and macrophages. 05:43 We also have complement receptors. 05:45 So you get the picture, that we do have a way to have some degree of specificity in terms of attacking just the foe and not something else.
About the Lecture
The lecture Neutrophil Chemotaxis by Richard Mitchell, MD, PhD is from the course Acute and Chronic Inflammation.
Included Quiz Questions
Which of the following is involved in neutrophil chemotaxis?
- platelet-activating factor
- Interleukin 10
- TGF-B
- Interferon-alpha
- C-reactive protein
What is the effect of chemokines at a low concentration?
- Activation of leukocyte integrins
- Induction of fibroblast migration
- Activation of the neutrophilic anti-microbial functions
- Attracting leukocytes to move out of the bloodstream
- Upregulation of L-selectin expression
Which of the following is recognized by the neutrophils as a pathogen-specific molecule?
- Lipopolysaccharide
- Leukotriene B4
- Phosphatidylserine
- Double-stranded DNA
- Methionine
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |