Playlist
Show Playlist
Hide Playlist
Inotropes – Heart Failure and Angina Management
-
Slides Inotropes Heart Failure Angina.pdf
-
Reference List Pharmacology.pdf
-
Download Lecture Overview
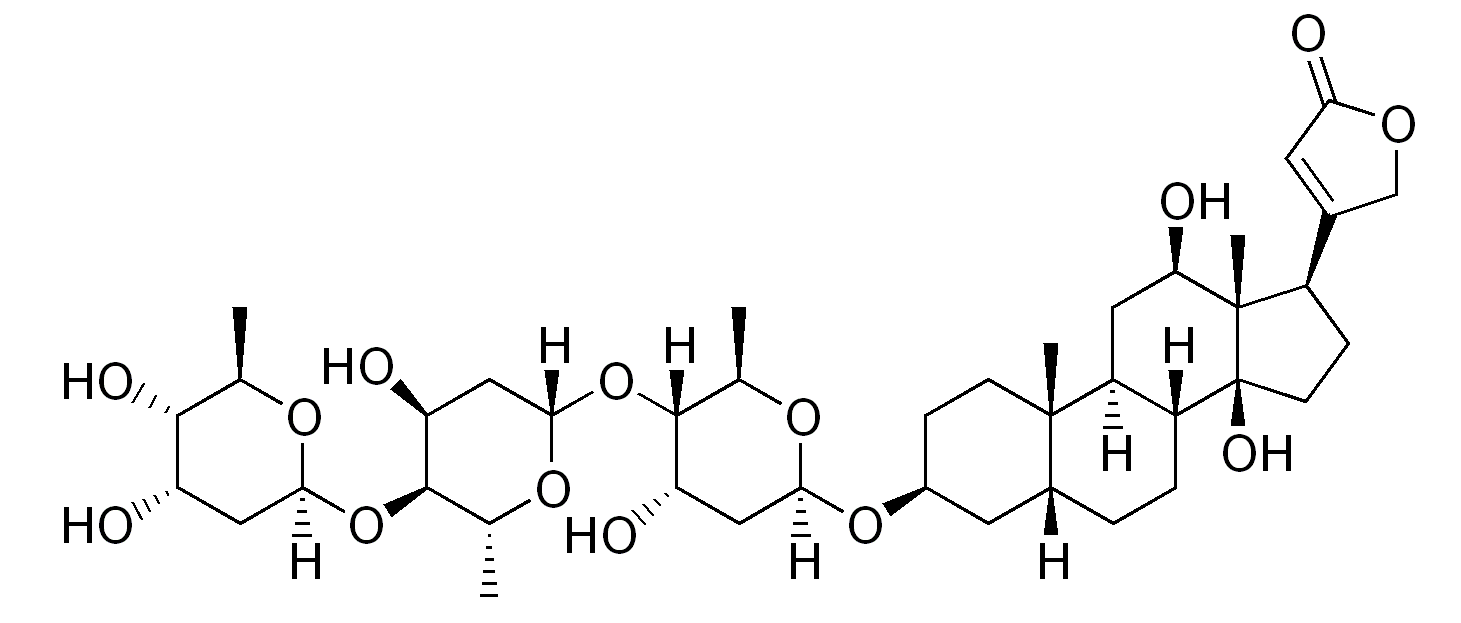
00:01 Welcome to pharmacology by Lecturio. 00:04 I'm Dr. Praveen Shukle, and today, we're gonna talking about the pharmacology of heart failure. 00:09 When we look at treating heart failure, we have several different ways of making the heart more efficient and a less likely to fail. 00:17 We can use drugs called inotropes. 00:20 These are medications that increase the heart’s contractility, or the force with which the heart contracts. 00:25 We can also use vasodilators. 00:27 Vasodilators open up the blood vessels so that the heart has less work to do to push the blood forward. 00:35 And finally, we have drugs that remodel the heart or affect the neurohormonal activity which is the brain hormone activity of heart failure itself. 00:46 Let's start out with the oldest of the heart failure drugs: the cardiac glycosides. 00:52 The prototypical example of the cardiac glycosides and probably, I would say, the one that is used 90% of the time in terms of this drug classes, digoxin. 01:03 Digoxin and digitalis are essentially the same thing. 01:06 Digitalis is the parent natural product that is derived from a plant. 01:13 How this drug works is that it inhibits the sodium potassium ATPase in the cell. 01:21 Now, what happens is, is that you end up having an increase in the amount of intracellular sodium. 01:28 So, you can see that the sodium level goes up, and because there's more sodium, there's going to be less excretion of calcium through the calcium/sodium antiporter. 01:39 Now, I was saying before that digoxin is derived from a plant. This is the plant called foxglove. 01:46 This was a weed where I grew up so if a farmer didn't use an herbicide to kill off this plant, you would see fields and fields of this brilliant purple flower everywhere. 01:58 So, it's quite beautiful but it was an annoyance for the farmers. 02:02 Anyway, let's get back to the drug. 02:03 Digoxin reduces the symptoms of heart failure. 02:07 It reduces hospitalizations due to heart failure but it didn't actually show a reduction in death from heart failure. 02:14 Now, admittedly, that information comes from a relatively old study. 02:18 We may find that a long-term study of patient shows that there's an improvement. 02:24 We may find that if we were to redo the studies today, that there could be a reduction in death but at any rate right now, what we're doing and what we're using this mediation for is to reduce hospitalization. 02:36 In terms of the dosing, the problem with digoxin is that it has a very narrow therapeutic index. 02:41 So, what we have to do is we have to monitor digoxin levels in some of our patients and patients are at high risk for becoming digitalis toxic or digoxin toxic. 02:55 In terms of drug interactions, there's multiple drug interactions that can occur with this agent. 03:01 First of all, it interacts with quinidine which technically is also a glycoside. 03:06 The toxicity can be increased with low serum potassium, low serum magnesium, and high serum calcium, and if you think about the way that the sodium potassium cotransporter works, you can kind of understand how that's happening. 03:24 Toxicity can result in arrythmia, nausea, vomiting, and diarrhea. 03:30 You could also get arrhythmia specifically due to calcium overload so you have to be careful using this agent. 03:36 In terms of other inotropes, we also have Beta I agonists. 03:42 Now, Beta I agonists are, generally speaking, dobutamine and dopamine. 03:48 Now, these are drugs that are used in the coronary care unit or the intensive care unit. 03:54 These are intravenous drugs. 03:56 These are not oral medications and you have to have a very good handle on how these medications are used. 04:01 They are not appropriate for chronic heart failure. 04:05 They are, however, pro-strength or inotropic agents. 04:11 One of the problems with these medications is you can develop a tolerance to them which means that the more you use it in terms of, say, a patient in the ICU or CCU, the less effective it becomes. 04:23 The other problem is that they're potentially pro-arrhythmia generating or proarrhythmogenic or arrhythmogenic drugs. 04:31 Again, I will also emphasize that this is an intravenous medication so there are no oral equivalents to this medication. 04:40 The next category I wanna talk about are the phosphodiesterase inhibitors. 04:44 So, this is kind of an exciting development in heart failure. 04:47 Now, what does phosphodiesterase do? So, let's take a look at a sympathetic nerve that you see drawn here. 04:55 It releases norepinephrine. 04:57 It can also -- this discussion can also apply to epinephrine being released from the adrenal gland. 05:03 Now, these agents will act on the Beta I or Beta II receptors in the heart. 05:10 This activates a Gs-protein. Gs means stimulatory G protein. 05:16 Now, here's the cardiac muscle in the grey. 05:19 What the Gs protein does is that it will activate adenylyl cyclase which will convert ATP into cyclic AMP. 05:27 When you have more cyclic AMP, you have increased calcium influx and you have more contraction. 05:35 The cyclic AMP-dependent phosphodiesterase is responsible for converting cyclic AMP to non-cyclic AMP or just plain AMP. 05:47 So, a phosphodiesterase inhibitor, when you think about it, is going to increase levels of cyclic AMP, and therefore, increase contraction. 05:58 Milrinone is the prototypical drug in this class. There are a couple of nuance out. 06:03 They haven't made it to market yet. 06:05 They're used mostly in the intensive care unit or in the critical care unit and again, this is an intravenous drug. It has an inotropic action. 06:15 It increases the cyclic AMP by inhibiting the breakdown enzyme called PDE3. 06:21 This increases the intracellular levels of calcium and it increases the strength of contraction or inotropy. 06:29 Now, additional factors to consider when using this med, it can be vasodilatory. This is because it also exerts its effects on PDE3 in vascular smooth muscle, so we need to watch for potential hypotension. 06:41 It’s also important to remember that it can be pro-arrhythmogenic. 06:45 We tend to reserve this medication for the management of acute heart failure or as short-term inotropic support after heart transplantation. It is not typically used for chronic heart failure. 06:56 In patients where it's been used long term in chronic heart failure, there is actually an increase in mortality in a small study so it's important to be aware of this. 07:06 Okay, that's great. You have managed to get through this part of the lecture. 07:11 We'll move on to other agents used in heart failure.
About the Lecture
The lecture Inotropes – Heart Failure and Angina Management by Pravin Shukle, MD is from the course Cardiovascular Pharmacology. It contains the following chapters:
- Overview Heart Failure
- Cardiac Glycosides
- β1 Agonists
- Phosphodiesterase Inhibitors
Included Quiz Questions
What is most likely to potentiate digoxin toxicity?
- Quinidine
- Decreased serum calcium levels
- Increased serum magnesium levels
- Increased serum phosphate levels
- Normal serum calcium levels
What increases the risk of digoxin toxicity? Select all that apply.
- Low serum potassium
- Low blood hemoglobin
- Low serum magnesium
- High serum calcium
- High serum phosphate
Which statement regarding cardiac glycosides is most accurate?
- They typically have a narrow therapeutic window.
- They cause arrhythmias due to calcium depletion.
- They typically have a wide therapeutic window.
- Their minimum effective concentration is much lower than their minimum toxic concentration.
- They may be used to treat nausea.
What is the primary mechanism of action of digoxin?
- Inhibition of sodium/potassium adenosine triphosphatase, mainly in the myocyte.
- Inhibition of sodium/potassium adenosine triphosphatase, mainly in the blood vessels.
- Activation of sodium/potassium adenosine triphosphatase, mainly in the myocyte.
- Activation of sodium/potassium adenosine triphosphatase, mainly in the blood vessels.
- Activation of the calcium/sodium antiporter.
Which statement below best describes digoxin?
- It decreases heart failure symptoms but does not decrease the mortality rate.
- It decreases heart failure symptoms and mortality rate.
- It decreases the mortality rate.
- It decreases arrhythmia symptoms.
- It decreases the mortality rate of cerebrovascular disease.
What best describes the mechanism of action of milrinone?
- It increases cAMP levels leading to increased contractile force.
- It decreases cAMP levels leading to increased contractile force.
- It increases cAMP levels leading to decreased contractile force.
- It decreases cAMP levels leading to decreased contractile force.
- It increases ADP levels leading to decreased contractile force.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
Thank you for covering pharmacokinetics and clinical aspects of positive inotropes in heart failure management.