Playlist
Show Playlist
Hide Playlist
Human Immunodeficiency Virus (HIV): Signs, Symptoms & Opportunistic Infections
-
Slides HumanImmunodeficiencyVirus InfectiousDiseases.pdf
-
Reference List Infectious Diseases.pdf
-
Download Lecture Overview
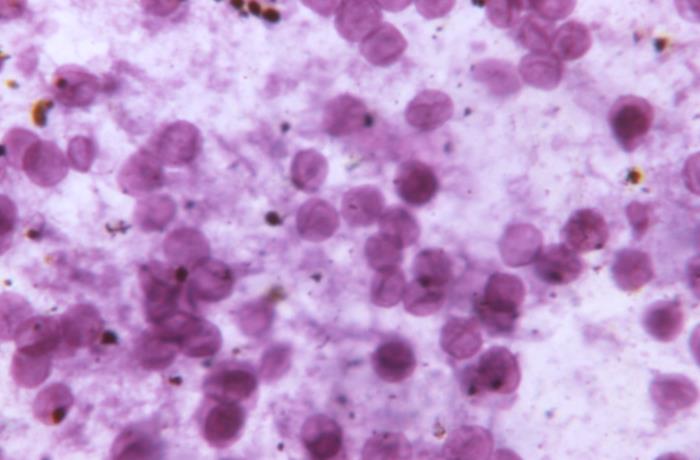
00:01 After initial HIV infection, patients typically develop acute retroviral syndrome within 1 to 6 weeks. 00:09 And the most common presentation is an infectious mononucleosis-like illness with fever, rash, pharyngitis, sometimes aphthous stomatitis. Most patients experience myalgias. Some develop diarrhea. 00:27 And if they come to a physician, the physician may not only find lymphadenopathy, but hepatosplenomegaly during this acute retroviral syndrome. 00:39 So in a study of 209 patients who had the acute retroviral symptom, you can see that 200 of them, or 96 percent, had fever. 00:54 A large percentage also had adenopathy, pharyngitis, and rash. 01:01 And as you can see from these declining numbers, only a minority had thrush, or neuropathy, or encephalopathy. 01:10 The encephalopathy, of course, if it does occur, is very serious. 01:15 In the early days of HIV infection, we were actually staging patients using these following stages. 01:24 Stage 1 was considered having a CD4 T cell count of greater than 500 cells per microliter, or a CD4 T cell percentage of more than 29 percent. As far as clinical evidence, there may be none at this stage. 01:44 Stage 2 was a lower number of CD4 T cells, 200 to 499, or a lower T cell percentage. 01:54 But once again, there was no necessary clinical evidence. No AIDS-defining condition. 02:01 Stage 3 was classified as a CD4 T cell count of less than 200 per microliter, which is the definition of AIDS, or a CD4 T cell percentage of less 14 percent. 02:17 Alternatively, a documentation of an AIDS-defining condition, such as opportunistic infection that we would never find in a normal host with lab confirmation of HIV infection. 02:31 The reason this not as important today, although it’s still used is because we are treating everybody. 02:39 It doesn’t matter what class they’re in. Everyone is recommended to get antiretroviral therapy. 02:50 Now, some of the opportunistic infections are worth spending some time on. Here are just four examples. 02:56 Let’s start out with perhaps the most common opportunistic infection that you’d find, and this is pneumocystis pneumonia caused by Pneumocystis jiroveci. 03:08 And patients who get this generally have a CD4 T cell count of less than 200. 03:16 However, I have seen patients whose T cell count was greater than that. 03:23 The way patients present. They will have dyspnea, fever, and classically a non-productive cough. 03:31 And this may have been for the last several weeks to months. 03:37 On exam, they will have tachypnea, rapid breathing, tachycardia, and in some cases, fine crackles. 03:46 But they could have a completely normal exam on auscultation. 03:53 Their chest X-ray may range from normal to the more common bilateral infiltrates that are interstitial in nature. 04:05 The diagnosis is made by special stains of respiratory secretions, and sometimes bronchoscopy is needed to confirm this. 04:13 But what you see are these cysts that are classic for pneumocystis. 04:21 The cysts are stained black with a GMS silver stain as seen in the photomicrograph in the lower right. 04:29 The treatment of this disease is trimethoprim-sulfamethoxazole (TMP/SMX), or dapsone +TMP. 04:37 Clindamycin and primaquine can be given to patients who cannot take ΤМΡ-ЅΜΧ. 04:43 And as an adjunctive therapy, especially in patients who have hypoxia, we would use tapering courses of corticosteroids. Let me discuss the key features of cryptococcal meningitis using this illustrative case. 05:07 Patients typically present with headache, fever, altered mental status, and visual disturbance. Interestingly, meningismus - the neck stiffness we typically associate with meningitis - is present in less than 40% of patients. 05:22 This is particularly true in immunocompromised patients who can't mount a robust inflammatory response. 05:29 The diagnostic approach is critical. Before performing a lumbar puncture, we need neuroimaging with CT or MRI if we suspect increased intracranial pressure or see focal neurologic deficits. 05:43 Look at this MRI - it shows classic findings in an immunocompetent patient. 05:49 You can see intense enhancement of the basal meninges, which is characteristic of cryptococcal infection. The patient has developed hydrocephalus, which occurs because the inflammatory process at the base of the brain obstructs normal CSF flow through the basal cisterns. 06:06 These imaging findings guide our management in three ways: If we see mass lesions, we need to consider other diagnoses like toxoplasmosis, lymphoma, or TB Hydrocephalus might require neurosurgical intervention with a shunt Increased intracranial pressure affects how we approach the lumbar puncture and often requires therapeutic CSF drainage This case beautifully demonstrates how imaging findings not only help establish the diagnosis diagnosis but also guide critical management decisions. 06:41 Let me walk you through the critical aspects of the lumbar puncture in cryptococcal meningitis. 06:47 When we perform an LP, measuring opening pressure is absolutely crucial - we're aiming for 20 centimeters of water or less. 06:55 In many cases, we'll need to perform serial therapeutic LPs to manage elevated pressures. 07:01 The CSF analysis typically shows some characteristic findings: a low white cell count, fewer than 50 cells per microliter, with a predominance of mononuclear cells. You'll usually see slightly elevated protein and low or normal glucose. 07:19 However - and this is really important - about a quarter to a third of patients can have completely normal CSF parameters even with proven infection, so don't let a normal CSF profile rule out the diagnosis. 07:34 For specific testing, we have three key tools. The cryptococcal antigen by lateral flow assay, or LFA, is our most sensitive and specific test. Culture is our confirmatory test. 07:49 And India ink - which you can see in this photomicrograph - gives us rapid preliminary results. 07:56 In the India ink preparation, you can see the characteristic appearance of Cryptococcus: round yeast cells with their distinctive clear capsular halos against the dark background. 08:07 This capsule is pathognomonic for Cryptococcus, though remember India ink is less sensitive than CrAg testing. 08:15 Treatment success depends heavily on managing that intracranial pressure, so these CSF studies guide both our diagnosis and ongoing management. 08:25 Let me cover the treatment approach for cryptococcal meningitis. 08:28 Treatment follows a two-phase strategy. We start with two weeks of induction therapy using amphotericin B plus flucytosine. 08:36 This is followed by eight weeks of consolidation therapy with fluconazole. 08:41 This regimen is based on strong clinical evidence and is our standard of care. 08:46 Throughout treatment, one of our biggest concerns is managing intracranial pressure. 08:50 We need to monitor ICP carefully as it's one of the main determinants of outcome. 08:56 If we see hydrocephalus develop, we need neurosurgical consultation to consider CSF diversion procedures. 09:03 There's one critical point I want to emphasize: in HIV patients with CD4 counts below 100, we should have a very high index of suspicion for cryptococcal disease. 09:14 In these patients, we should check serum cryptococcal antigen by lateral flow assay even before they develop obvious symptoms of meningitis. 09:24 This screening strategy can help us catch and treat the infection early, potentially preventing progression to meningitis. 09:32 Toxoplasma encephalitis is another one. 09:36 And this is generally found in patients with profoundly low CD4 T cell counts. 09:42 And these patients have symptoms of headache, confusion, and lethargy. 09:50 They may present with a seizure in up to 30 percent of the patients. 09:54 So serious manifestations because this essentially produces a space occupying lesion in the brain. 10:02 And so, on exam, you’re gonna have focal neurologic signs in maybe 70 percent of patients. 10:08 And that includes hemiparesis, cranial nerve palsies, ataxia, and sensory deficit. 10:15 And I think you can see from these images that these lesions are not only multiple, but maybe quite large and are common cause of increased intracranial pressure, and possibly even herniation. 10:32 You make the diagnosis by doing a head CT scan or MRI. The MRI is much more sensitive than the CT scan. 10:43 So if you had a positive serology for toxo, it’s really non-specific because a lot of us have a positive serology for toxo. 10:56 But if a patient has these lesions on imaging and their toxo serology is negative, that’s very much against the diagnosis of toxoplasma encephalitis. 11:09 We treat this with pyrimethamine plus sulfadiazine plus folinic acid for at least 6 weeks, and maybe more. 11:19 And the final example of an opportunistic infection I want to present is that of disseminated Mycobacterium avium complex, or MAC infection. 11:29 And these patients too, usually have a CD4 count of less than 50 cells per microliter. 11:38 And the way these patients present. They often present with high fever, marked weight loss, night sweats, abdominal pain, diarrhea. These are very sick patients and they’re often very wasted. 11:52 On exam, they have lymphadenopathy. Most of them have hepatosplenomegaly. 11:58 And because the bone marrow is involved, you’re gonna find severe anemia. 12:04 You’re gonna have an increased alkaline phosphatase. And so, the diagnosis is made primarily by drawing blood cultures. 12:14 And mostly, the blood cultures are going to be found to be positive. Or if you isolate the organism from another normally sterile site. 12:25 This is treated with either clarithromycin or azithromycin, plus ethambutol, plus or minus rifabutin. 12:38 It’s important nowadays to speak to you about the immune reconstitution inflammatory syndrome, otherwise known as IRIS. 12:49 And think about what’s happening. 12:51 We’re giving everybody antiretroviral therapy and some of the patients that we’re treating have opportunistic infections. 13:01 So as their immune system function improves with therapy, they may start attacking the organisms that are causing opportunistic infection. 13:13 And actually, when they were immune-suppressed, they weren’t having as much inflammation. 13:21 Now, we’re providing them with the ability to have inflammation and they’re kind of lighting up these infections as their immune system begins to attack them. 13:31 So what’s happening is, an inflammatory response to the pathogen is triggered by the initiation of antiretroviral therapy. 13:40 We thought we were helping them, but in some cases, we’re actually hurting the patients. 13:46 So let me give you some examples. 13:50 If they had a viral infection and their immune system is coming back, it may worsen. 13:55 If they had a fungal infection, it may worsen. 13:58 Or a bacterial infection like the Mycobacterium avium complex infection we talked about.
About the Lecture
The lecture Human Immunodeficiency Virus (HIV): Signs, Symptoms & Opportunistic Infections by John Fisher, MD is from the course Genital and Sexually Transmitted Infections. It contains the following chapters:
- HIV – Signs/Symptoms
- Opportunistic Infections – Four Examples
- Cryptococcal Meningitis
- Toxoplasma Encephalitis
- Disseminated Mycobacterium Avium Complex Infaction
Included Quiz Questions
Which of the following best describes the common findings in acute retroviral syndrome?
- Fever, lymphadenopathy, sore throat, rash, myalgia/arthralgia, headache
- Cervical, mediastinal, and axillary non-tender lymphadenopathy
- Exudative pharyngitis and hepatosplenomegaly
- Fever, bilateral throbbing headache, blurred vision
- Pneumonia, altered level of consciousness, organ failure
Which of the following is the antibiotic treatment of choice for Pneumocystis jirovecii pneumonia in patients with HIV?
- Trimethoprim-sulfamethoxazole
- Azithromycin
- Clindamycin
- Ceftriaxone
- Levofloxacin
Which of the following types of infection is an AIDS-defining condition?
- Pneumocystis jirovecii pneumonia
- Staphylococcus cellulitis
- Escherichia coli pyelonephritis
- Hepatitis C infection
- Oral candidiasis
What structure does india ink stain allow the observer to see on microscopy?
- Cryptococcal capsules
- Cryptococcal hyphae
- Mycobacteria cell walls
- Toxoplasma nuclei
- Toxoplasma cell walls
Which of the following is the most common method of confirming the diagnosis of disseminated infection caused by mycobacterium avium complex (MAC) in persons with HIV?
- Blood cultures
- Serology
- Abdominal computed tomography scan
- Brain magnetic resonance imaging
- Gram stain
Which of the following refers to paradoxical worsening of a preexisting infectious process following initiation of antiretroviral therapy in a patient with HIV infection?
- Immune reconstitution inflammatory syndrome
- Acute retroviral syndrome
- Acquired immunodeficiency syndrome
- Hyperlactataemia syndrome
- Differentiation syndrome
Which of the following is the imaging modality of choice in the diagnosis of Toxoplasma gondii encephalitis?
- Magnetic resonance imaging
- Compute tomography scan
- Ultrasound
- Thallium single photon emission computed tomography
- positron emission tomography
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
I am an RN student, 2nd semester. This lecture is very informative. I like when it describing the opportunistic infection, the lecture provides info from the type of illness, s/s, type of diagnostic tests, and treatments.