Playlist
Show Playlist
Hide Playlist
Heparin vs. Warfarin
-
Slides Hematology-Pharmacology.pdf
-
Download Lecture Overview
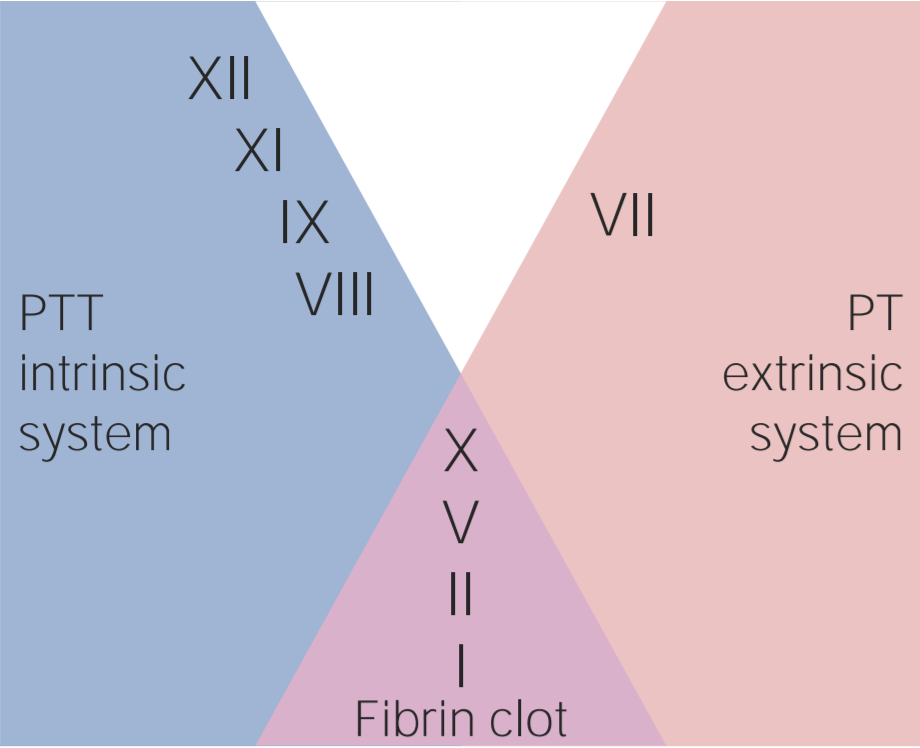
00:01 Here’s an important table to compare heparin versus warfarin. 00:05 Heparin, number one, large; warfarin, small lipid. 00:11 What does that mean to you? Passes through the blood-placental barrier. 00:14 Next, administration. Heparin parenteral only, IV or subcutaneous. 00:21 Warfarin a.k.a. Coumadin, oral. 00:24 Site of action, heparin blood; warfarin, why the liver? Good, the vitamin K dependent factor is epoxide reductase. 00:32 Vitamin K epoxide O, is the abbreviation and then you have the letter C, complex. 00:41 The onset of action is rapid with heparin hence it’s a bridge therapy so you start the heparin first. 00:50 Warfarin will take time for it to inhibit the enzyme. 00:54 The mechanism of action, heparin activate antithrombin III, warfarin we talk about vitamin K dependent factor and specifically epoxide reductase. 01:02 As for its duration of action while heparin will be acute, whereas warfarin will be chronic. 01:07 In terms of inhibition coagulation in vitro, heparin, it will show it to you. 01:13 Warfarin not so much because heparin works so quickly in antithrombin III. 01:17 Treatment of acute overdose, protamine sulfate whereas with warfarin you should be thinking about replenishing those vitamin K-dependent factors. 01:26 So maybe fresh frozen plasma or something called prothrombin complex concentrates or PCC. 01:34 How many letters in PCC? One, two, three. Now there are different types of PCC, you can have a four element or a three element. 01:43 Now what does that even mean? Keep this in mind, if I say prothrombin to you what branch of the coagulation pathway were you thinking? Extrinsic. If you're thinking about extrinsic and you're thinking warfarin, you should be thinking about the Vitamin K dependent factors. 01:59 What are the four major prothrombotic agents? II, VII, IX, and X. 02:04 You can have clinically PCC, prothrombin complex concentrate with not as much factor VII in it. 02:12 So now all I have left is II, IX, and X. That’s the one that you'd want to know. 02:16 In other words what I'm trying to tell you is you have a patient who is in a state of hemorrhage, right, too much anticoagulation. 02:24 You want to replenish your coags, you give FFP or PCC, keep that in mind. 02:30 Monitoring, we talk about heparin will be PTT, warfarin will be [00:02:35] the BEPT the BEPT. 02:38 How many seconds for PT? 11-15 seconds, you add a T, you add more time that time for PTT is 25 - 40 seconds. 02:48 You can use which one of these tests as well for as a liver function test, please? PT. Site of action of warfarin is deliver. 02:58 And we talked about placenta crossing. 03:03 Warfarin will wait to where upon the fetus, it’s a teratogen, do not use it. 03:07 Heparin could still be used during pregnancy. 03:10 Then we’ll take a look at thrombolytics. 03:12 You see a patient walking through the door. 03:14 Patient come in and has chest pain, tachycardia, diaphoresis. 03:18 The pain is radiating up to the jaw sub-sternal, left side shoulder. 03:23 Take a look at the labs and you will find an increase in troponin I and upon let’s say ECG leads, II, III and AVF, then you'll find an ST elevation. 03:33 At this point, your thinking, most likely transmural type of infarction taking place of the right coronary artery obviously having a myocardial infarction. 03:42 At this point, your objective as a clinician has to make sure that you bust this clot as quickly as possible. 03:48 Welcome to thrombolytics, alright? Now, you may either convert directly or indirectly, the enzyme here of course would not be fibrin nor would it be thrombin, you bring it to discussion the conversion of plasminogen into plasmin and as plasmin is then going to break up this clot very, very quickly, isn't it? Now what's really fascinating about this is the fact that you would find an increase in PT, PTT when you're using a thrombolytic. 04:16 Here’s every possibility that you may also bring about reperfusion injury, remember that? But the discussion with perfusion injury is all about risks and benefits. 04:24 And if you know the time is salvation, in other words three hours, if you are able to get in there and bust that clot, the amount of damage that you're going to cause to the heart and to the tissue will be rather minimal, correct? Reperfusion injury. 04:39 Now, with all that said, you use the thrombolytic, you create or activate your plasma, however, what's never going to decrease when you use a thrombolytic obviously is a no change in platelet count but you may have an increase in PT, PTT though because the coagulation factors are busy. 04:56 The clinical use here would be obviously myocardial infarction; however, the toxicity becomes important. It may result to massive, massive type of bleeding. 05:06 Now what you'd really be careful of is, as far as you're concerned, remember, you are not in practice yet, you, when you become let’s say a neurosurgeon, then you do what you want in terms of your patient, knowing also what’s the consequences are. 05:20 However, at this point the standard is contraindicated in patients with active bleeding, absolutely contraindicated in patients with history of intracranial bleeding. 05:32 Contraindicated in recent surgery, or if there is a known bleeding diet disease for example your patient has DIC or obviously severe hypertension. 05:46 Now if there is toxicity that you're worried about with providing thrombolytic and you have too much, too much, too much bleeding, then I'm gonna show you in a diagram upcoming the mechanics of aminocaproic acid which is then going to be inhibitor of fibrinolysis. 06:03 So there's some important antidotes, aren’t there, that we've talked about, there's four. With heparin, what is it called? Remind me? Protamine sulfate, number one. 06:15 With warfarin, you wanna replenish the coagulation factors. 06:19 Fresh frozen plasma or prothrombin complex concentrates. 06:26 If it’s TPA, so we're gonna talk about tissue plasminogen activators either direct or indirect, then you're thinking about aminocaproic acid. 06:36 Let’s take a look at that diagram. 06:38 Let me set this up for you, the objective of this entire diagram is to make sure that you bust this clot. 06:47 I want you to take a look at where we are down at the bottom here. 06:51 You see the word fibrin, please? Well, can you picture a clot with fibrin? Can you picture that for me? Good, I mean that fibrin, it was acted upon by thrombin and it was in-between the glycoprotein 2b3A and you form the stable insoluble clot. 07:08 That’s what you're dissolving, are we clear? In order to dissolve this clot, then you need to activate whom? Now you go to the top, take a look at plasminogen. 07:19 So our objective is to take this clot that had fibrin, is to activate plasminogen into plasmin and look at plasmin’s doing right there smack in the middle, do you see that? That plasmin is taking the fibrin which is the clot form right now and dissolving it. 07:36 Now what do we call this clinically? How do we know if we are breaking down clot? How do you know? You are looking at a lab test in your hand or on a screen, how do you know, that you know that the clot is being broken down? You are looking for D-dimers. 07:50 So what are D-dimers specifically? You must know the D-dimers are not fibrinogen but fibrin split products. 07:57 So you can expect whenever plasmin, when it’s activated, when it’s breaking down your fibrin, no doubt you would expect to find D-dimers. 08:05 Now, what are different ways is in which you can use a thrombolytic or what are the thrombolytics that would activate plasminogen? On your left you'll notice we have activators either direct or indirect. 08:19 These activators include while you're focusing upon the suffix A-S-E, I don’t wanna say it out loud because it sounds like ACE and I don’t want to do that because it’s going to confuse you, right? Because whenever you talk about ACE inhibitors obviously talking about enalapril and so on and so forth but I want you to be able to identify A-S-E, and when you do so then this is going to be your plasminogen activators, using alteplase tPA, tissue plasminogen or reteplase, or streptokinase, or tenecteplase. 08:52 I said earlier, in a previous discussion, contraindications. 08:57 If you find that your patient has previous surgery, is in a state of bleeding that is an intracranial bleeding then these drugs will be contraindicated, is that clear? Let’s say that there is toxicity with too much bleeding, and I told you that there would be an antidote and that antidote would be one in which it would inhibit fibrinolysis by inhibiting the conversion of plasminogen into plasmin. 09:23 Take a look over to the right and I need you to focus on aminocaproic acid. 09:28 So that will be your antidote as far as if you have a patient who's suffering from tPA toxicity. 09:34 By tPA I'm referring to tissue Plasminogen Activator. 09:38 These are your thrombolytics.
About the Lecture
The lecture Heparin vs. Warfarin by Carlo Raj, MD is from the course Hemostasis: Basic Principles with Carlo Raj. It contains the following chapters:
- Heparin vs. Warfarin
- Thrombolytics
Included Quiz Questions
Where in the body does warfarin target the vitamin K epoxide reductase complex subunit 1?
- Liver
- Blood
- Endothelial cells
- Bone marrow
- Lymph nodes
Which of the following routes of administration is most commonly used for warfarin?
- Oral
- Sublingual
- Intravenous
- Intramuscular
- Subcutaneous
Which coagulation factors are mainly affected by warfarin therapy?
- II, VII, IX, X
- II, XII, XII, VIII
- VII, XIII
- III, V, X
- VII, VIII, IX, X
Which of the following medications is most appropriate for the immediate reversal of heparin overdose?
- Intravenous protamine sulphate
- Oral protamine sulphate
- Fresh frozen plasma
- Intravenous vitamin K
- Oral vitamin K
Heparin administration is most appropriately monitored via which of the following laboratory investigations?
- Partial thromboplastin time
- International normalised ratio
- Bleeding time
- Clotting time
- Prothrombin time
Customer reviews
4,5 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
1 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
Dr Raj is amazing teacher, the wsy he explains things is wonderful!! Thank you so much doc
The professor is knowledgeable and able to explain in detail.