Playlist
Show Playlist
Hide Playlist
Henderson-Hasselbalch Equation: Examples
-
Slides 07 pHBuffers AcidBaseBalance GeneralPhysiology.pdf
-
Download Lecture Overview
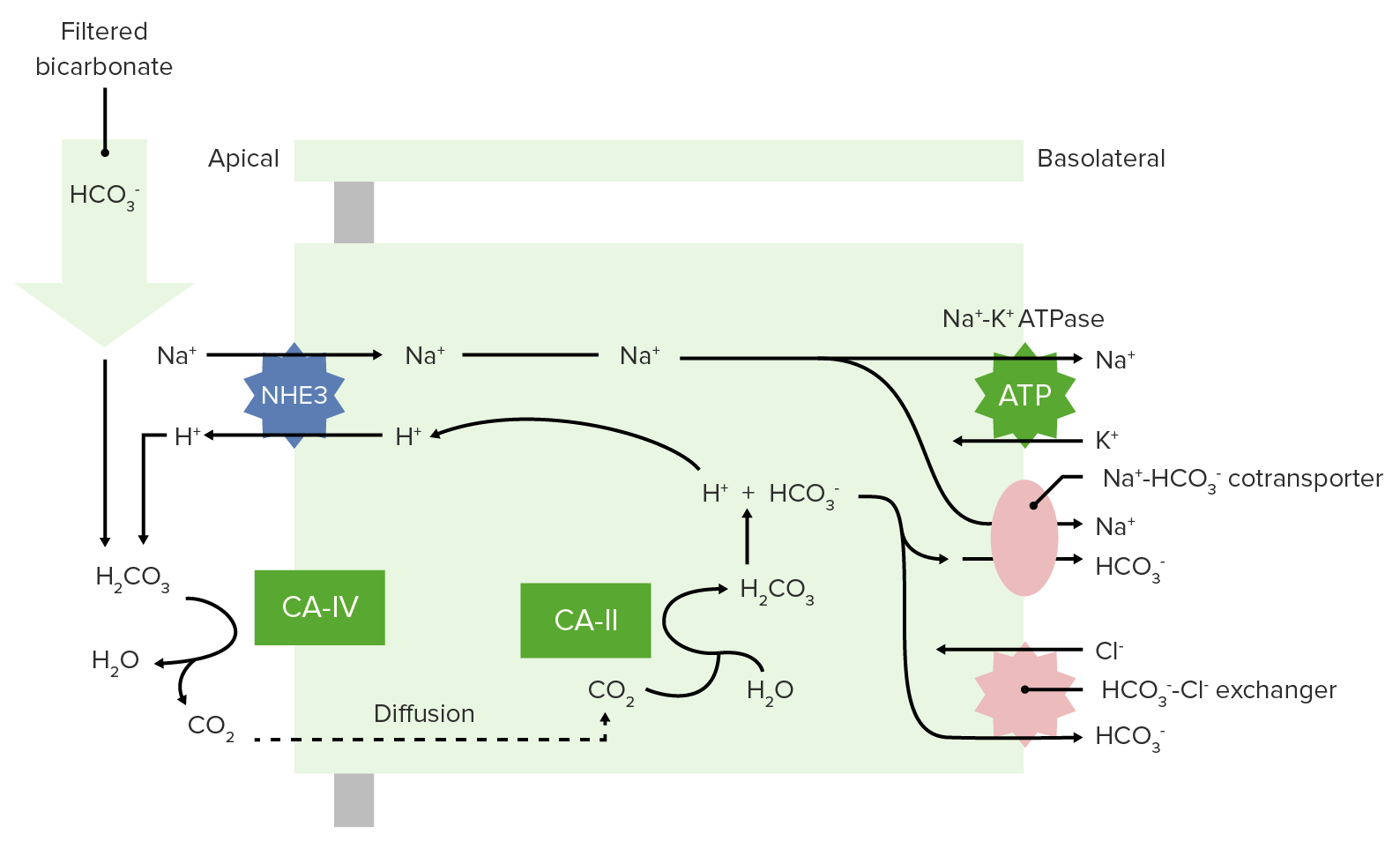
00:00 These types of equations that we utilize from this example formula. 00:07 Let’s go through three examples. 00:11 The first one is normal arterial blood. 00:15 So you get a blood gas from someone. The bicarb comes back at 24 millimolar. 00:20 The PaCO2, this is PaCO2 is 40 millimeter of mercury. 00:25 You plug into this equation and you should get a pH of 7,4. 00:30 How to encourage you to be able to do that? So you can make sure you can get the same numbers as we’re showing here. 00:37 Now, let’s take a different example. 00:39 This is an acidic environment. So they might were expecting that they might have an acidemia. 00:46 They have a bicarb of 26, PaCO2 0f 60. 00:51 The result of pH of 7,26. So it is confirm, this person has an acidemia. 01:00 If we expect an alkalemic blood, what we wanna do is get a blood gas from that person. 01:07 We find that we have a bicarb of 22 millimolar, and we have a PaCO2 of 20 millimeter of mercury. 01:16 We put both of those into this equation. 01:18 And again, an equation is 6,1 plus the log of the bicarb, divided by the PCO2. 01:25 But remember, PCO2 we have to multiply it by a factor to get it into a dissolve form. 01:31 Should yield 7,66. 01:34 So that’s confirmed this is an alkalemia. 01:38 So you can use this Henderson-Hasselbalch equations. 01:41 And we can see how it was develop via titration curves to calculate anybody’s pH if we have an arterial blood gas.
About the Lecture
The lecture Henderson-Hasselbalch Equation: Examples by Thad Wilson, PhD is from the course Acid-Base Balance.
Included Quiz Questions
An arterial blood gas shows the following: HCO3- = 18 mM; PaCO2 = 35 mmHg. What is the pH?
- 7.33
- 7.23
- 7.43
- 7.36
- 7.42
An arterial blood gas shows the following: HCO3- = 27 mM; PaCO2 = 50 mmHg. What is the pH?
- 7.35
- 7.28
- 7.38
- 6.89
- 7.42
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |