Playlist
Show Playlist
Hide Playlist
Pediatric Bleeding Disorders: Hemophilia
-
Slides Bleeding Disorders.pdf
-
Download Lecture Overview
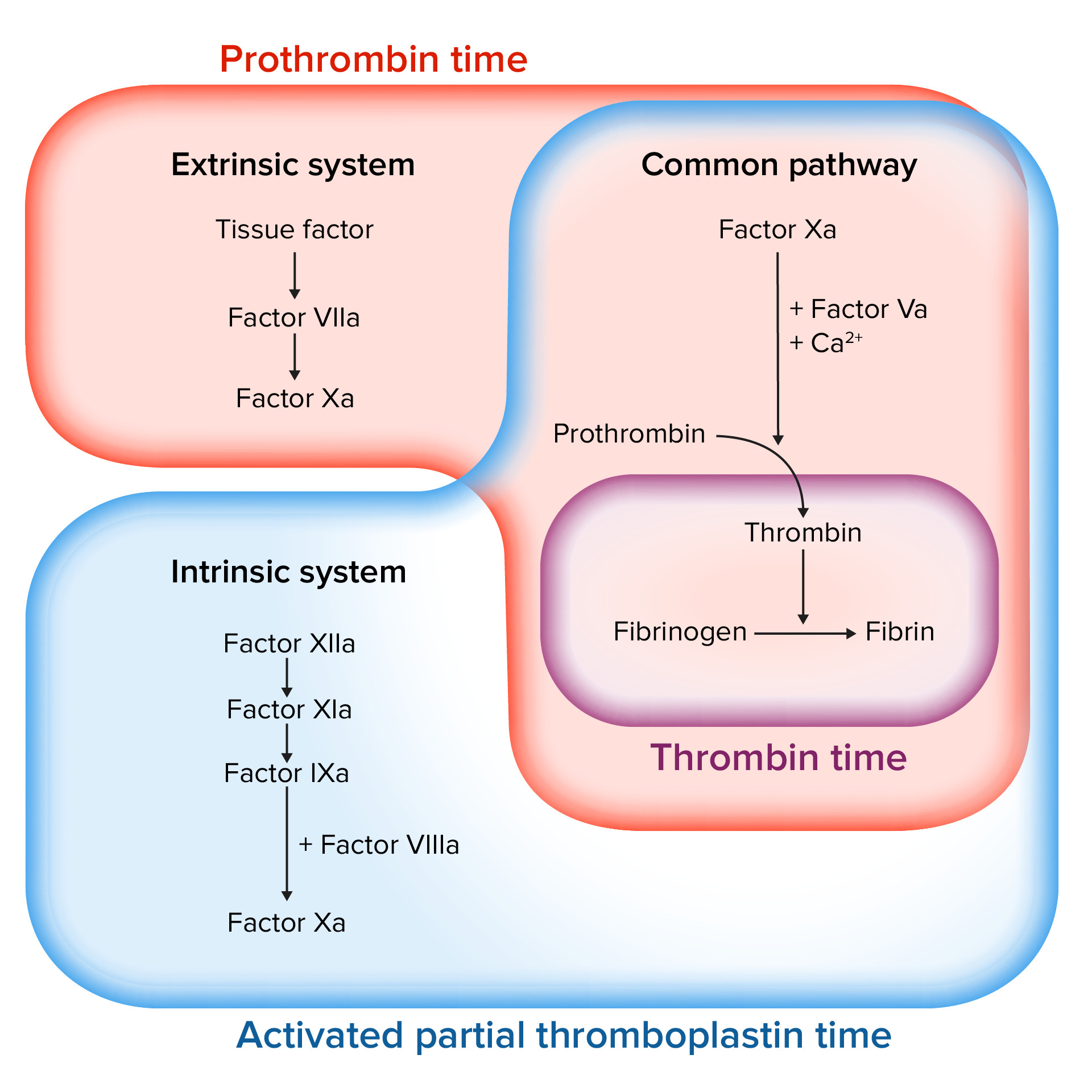
00:01 Okay, let’s switch gears now and talk about the clotting cascade, which really happens first, but we’re presenting it in a slightly backwards order. 00:09 So in the clotting cascade, we have to recall that there is an intrinsic pathway of clotting and an extrinsic pathway of clotting. 00:17 I don’t think it’s so important clinically to remember which clotting factor goes to which clotting factor but there are 2 that are very important to remember and those are factors VIII and IX which are circled on the slide. 00:30 You can see these are both part of the intrinsic pathway and thus, the PTT is the test of choice for determining whether patients have a deficiency of either factor VIII or factor IX. 00:41 A deficiency of factor VIII or factor IX is called hemophilia. 00:46 So, hemophilia A is a deficiency of factor VIII and hemophilia B is a deficiency of factor IX. 00:54 Both of these diseases are X-linked recessive. 00:57 That means almost always these are male patients who have the disease and females are simply carriers, although very, very rarely female can have the disease. 01:07 A variable severity exists with patients with this disease. 01:11 They may have mild bleeding or they may have very severe bleeding. 01:15 So we may expect to see a variety of severity in patients with hemophilia. 01:22 So let’s look at how this disease typically presents. 01:27 Especially on patients with more severe disease, it can absolutely present in a newborn period. 01:33 Classically, about half of undiagnosed infants, infants who weren’t diagnosed prenatally through genetic testing, will present because of bleeding after a circumcision. 01:44 So the circumcision is done, the child bleeds excessively after the procedure and the doctor puts two and two together and says, “Yeah, maybe this baby has hemophilia.” The patients may develop intracranial hemorrhage at birth. 01:59 This is because they bleed excessively as a result of a normal amount of birth trauma. 02:04 They may have a large cephalohematoma or parents may notice that after the umbilical stump falls off between weeks one and four, that the umbilical stump bleeds excessively. 02:17 There can be a later presentation especially those in more mild disease. 02:21 They can present with hemarthrosis or blood in the joint and on average, patients present with their first hemarthrosis around 10 months of age. 02:31 Hematomas can present, but not usually bruising or petechiae, that’s more consistent Von Willebrand’s. 02:38 In these patients, they simply have a large swelling hematoma. 02:42 Patients may present with a GI bleed or they may present with an intracranial bleed. 02:48 And keep that in mind, intracranial bleed in a child with minimal trauma might be hemophilia. 02:54 There are very rare cases reported where people have such mild disease that they don’t even present well into adulthood, but most people will present in childhood, usually by the age of 4. 03:06 So what sort of spontaneous bleeds can happen in these patients? They could spontaneously have an intracerebral bleed and that is devastating. 03:14 Those are usually the more severe patients. 03:16 They often have frequent bleeding from nose and mouth, again, the nose bleeds and the bleeding after brushing their teeth and they may have spontaneous joint bleeds, which are real problem and we’ll talk about that in a little bit. 03:28 Patients may also have posttraumatic bleeds, but what’s key is these may be delayed, so they have their trauma and then later they start bleeding especially with the joints. 03:39 So the long term complication that’s really debilitating for these patients is the hemophilic arthropathy. 03:46 It turns out blood is very irritating to joints and it causes degradation of that joint and these patients end up with very severe arthritis. 03:56 They also can occasionally get infection from transfused blood products. 04:01 There was a period of time when we didn’t know how to screen for HIV in the blood supply or we didn’t know HIV existed yet. 04:08 And patients with hemophilia were frequently getting those diagnosed with HIV because of their transfusions. 04:17 So nowadays, transfusions are very safe at United States and blood is screened very effectively for all kinds of disease. 04:26 However, there are still areas where transfused blood isn’t screened for all diseases and so these patients may be at risk. 04:33 Patients overtime can develop autoantibodies against recombinant factor we’re using to treat them and we’ll talk about treatment in a bit, but we’re using recombinant factor VIII. 04:44 And it is a human recombinant, but they can nonetheless develop autoantibodies. 04:49 This is a devastating thing to have happened. 04:52 These patients are at much greater risk for severe bleeding events because we are having difficulty transfusing a medicine to which they’re allergic. 05:01 Because of the constant autoimmune nature of this disease, just demounting of autoimmune response will result in things like delayed growth and delayed sexual development. 05:13 So this is a real problem if children develop autoantibodies against their treatment. 05:19 So how do we make the diagnosis? If we suspect the patient has hemophilia, we will obtain a PT and PTT and we’ll notice, “Oops, the PTT is prolonged”. 05:28 Remember that’s the intrinsic pathway involving factors VIII and ix. 05:32 They should have a normal PT or INR. 05:35 So it’s not liver failure or some other cause of prolonged bleeding. 05:39 When we get a platelet function assay, we should and it should be normal because now, we’ve ruled out for example Von Willebrand’s disease. 05:48 Factor VIII and factor IX levels are available and we can check plasma level so we can distinguish whether if this is hemophilia A or B and that’s important for therapy. 05:59 If factor 8 is low, don’t forget to check the Von Willebrand’s disease factor as this could be Von Willebrand’s disease. 06:06 Okay, how do we treat these patients? For severe patients, we’re going to give them factor all the time to maintain them at a certain level. 06:15 However, for less severe patients and really for all patients who bleed and have a bleeding event, we have to treat them immediately. 06:24 So patients with hemophilia will come in to be treated for acute bleeds for which we give them recombinant factor. 06:31 The dose depends on the desired percentage of factor that needs to be achieved based on the weight of the child and what percent you want to get to. 06:40 In what percent you want to get to has to do with what the bleed is. 06:45 You probably don’t have to memorize that for an exam, but just to understand that it’s going to vary depending on the severity of bleed and how bad off the patient is and how much they weigh. 06:56 Typically, for severe patients, we will give primary prophylaxis. 07:02 So these are patients with, for instance, less than 1% of their normal amount of factor and these patients will get typically between two and three times per week doses of recombinant factor. 07:16 The goal is to raise these severe patients up to the 1% level, which equivalent to moderate disease. 07:23 The reason why we don’t do this for everyone, it is extremely expensive. 07:28 Recombinant factor costs a lot of money and so we have to be careful about not giving it when it’s not needed. 07:35 Patients will often require transfusions because they’re bleeding too much and that’s generally indicated for severe bleeds, especially in patients with hemodynamic consequences. 07:45 So we don’t necessarily transfuse a patient who has a low hemoglobin if their vital signs are stable and their bleeding is controlled. 07:52 But in an active bleed or a patient with tachycardia, we need to transfuse. 07:58 Don’t forget, we usually have to transfuse these patients if they’ve had severe bleeds. 08:03 If they have tachycardia or if they’re actively bleeding, we must consider transfusion. 08:09 For a patient with a lowish hemoglobin but who’s no longer actively bleeding and their heart rate is okay, we generally don’t transfuse. 08:18 So that’s my summary of bleeding problems in children. 08:21 Thanks for your attention.
About the Lecture
The lecture Pediatric Bleeding Disorders: Hemophilia by Brian Alverson, MD is from the course Pediatric Hematology. It contains the following chapters:
- Von Willebrand Disease
- Unusual Platelet Aggregation Problems
- Hemophilia
- Hemophilia – Factor Replacement
Included Quiz Questions
Which of the following is the most common initial presentation in patients with hemophilia?
- Bleeding after circumcision
- Retinal hemorrhage
- Hematomas after trauma
- Brisk bleeding with first menses
- Bleeding between menses
Which of these diseases is an X-linked recessive disorder?
- Hemophilia A
- Type 1 vWD
- Type 2A vWD
- Glanzmann thrombasthenia
- Aplastic Anemia
Which of the following clinical manifestations of bleeding more likely reflects hemophilia than a platelet disorder?
- Hemarthrosis
- Ecchymoses
- Petechiae
- Purpura
- Blood in stool
Which of the following is NOT a long term complication of hemophilia?
- Accelerated sexual development
- HIV infection after transfusion
- Arthropathy
- Autoantibodies against factor VIII
- Intracranial bleeding
Which of the following is consistent with hemophilia?
- Prolonged PTT
- Prolonged BT
- Prolonged PT and PTT
- Increased international normalized ratio
- Decreased von willebrand factor
Which of the following does NOT influence the dose of recombinant factor 8 in the treatment of an acute bleed due to hemophilia?
- Patient's age
- Location of bleeding in the body
- Severity of hemophilia
- Target percentage of factor 8 to be reached
- Patient's weight
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
Excellent lecture. Hematology is often poorly explained. This is not the case here! Thanks!