Playlist
Show Playlist
Hide Playlist
Genetic Susceptibility to Autoimmune Diseases and Mechanisms
-
Slides Autoimmune Disease.pdf
-
Reference List Immune System.pdf
-
Download Lecture Overview
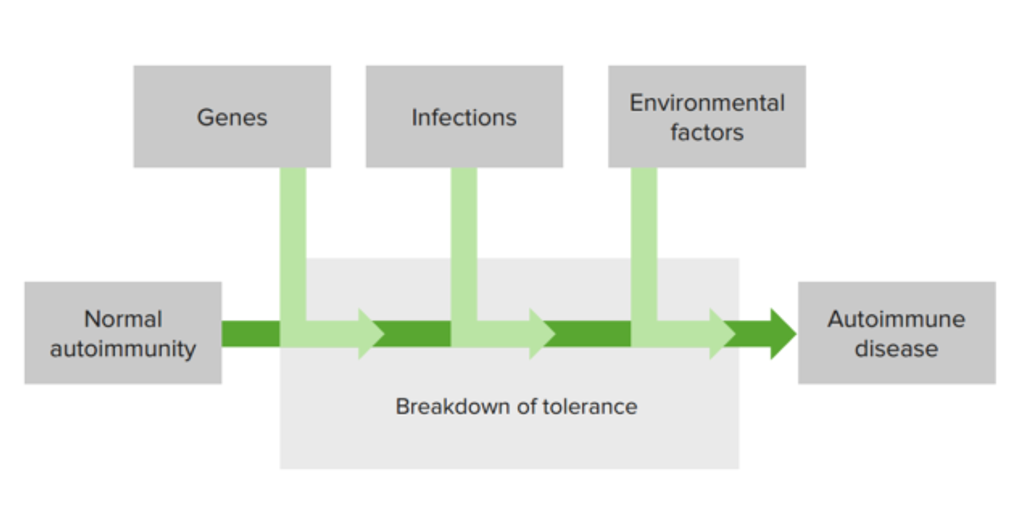
00:01 So there’s a genetic susceptibility, in the vast majority of cases. 00:07 This is many different genes acting together resulting in a failure of self tolerance and the development of self reactive lymphocytes. 00:17 Coupled with these multiple genetic factors, are environmental stimuli such as tissue injury, inflammation, infection and so on. 00:26 This can lead to the activation of tissue antigen presenting cells. 00:31 And these antigen presenting cells can activate self reactive lymphocytes, which then become self reactive effector lymphocytes that are actually going to carry out the damage that is occurring. 00:45 There’ll be tissue injury, and the result will be autoimmune disease. 00:50 So let's have a look at some of the genes that have been implicated. 00:55 In animal studies, it’s quite clear that maybe in a given autoimmune disease, perhaps as many as 20 or 30 different genes can contribute. 01:06 And the genes that have been identified both in animal studies and in human patients include the following - The MHC is nearly always implicated. 01:19 And the MHC of course is involved in antigen presentation to T-cells. 01:25 And this is a gene that virtually all polygenic autoimmune diseases, which is 99% of autoimmune diseases, have a MHC influence on their development. 01:38 CTLA4 which is a molecule that you find on the surface of T-cells, and actually acts as a kind of negative regulator of T-cells, and stops excessive T-cell responses. 01:52 This gene or polymorphisms of this gene have been implicated in thyroid autoimmune diseases, in type I diabetes and in rheumatoid arthritis. 02:04 A molecule called PTPN22, which is involved in antigen receptor signaling has also been implicated in those particular diseases. 02:16 In other words, thyroid autoimmune disease, type I diabetes and rheumatoid arthritis. 02:21 Polymorphisms of complement components, particularly C1q, C2 and C4 are involved in the development of systemic lupus erythematosus. 02:35 Meanwhile, polymorphisms in the interleukin-2 gene; interleukin-2 is involved in stimulating lymphocytes. 02:42 This gene is implicated in rheumatoid arthritis, type I diabetes and multiple sclerosis. 02:49 Another cytokine, in this case an immunosuppressive cytokine IL-10. 02:54 Polymorphisms of the IL-10 gene have been implicated in SLE and type I diabetes. 03:02 And the BLK molecule involved in B-cell signaling has also been implicated in SLE as well as in rheumatoid arthritis. 03:11 So this is just a few examples of many genes that have been implicated in the development of autoimmune diseases. 03:19 Let’s have a look at some of the MHC associations in autoimmune disease. 03:25 Looking at MHC Class I, in this example we can see that individuals that are HLA-B8 have a relative risk of three for the development of myasthenia gravis. 03:39 This means that if an individual happens to be HLA-B8 rather than other HLA-B variants, they are three times more likely to develop myasthenia gravis. 03:52 So for example, this individual may be HLA-B6 and B8, and they would be three times more likely to develop myasthenia gravis than somebody that was for example HLA-B1 and HLA-B14. 04:08 Ankylosing spondylitis - the relative risk is 87 for individuals with HLA-B27. 04:17 So a huge relative risk. 04:22 Let me share a little bit of confidential medical information with you. 04:26 I know that I am HLA-B27, but I don’t have ankylosing spondylitis. 04:33 In fact only around about 10% of individuals that are HLA-B27 develop ankylosing spondylitis. 04:41 So although the relative risk is really pretty high, you need other genes and you need environmental factors in order to develop ankylosing spondylitis. 04:51 So it’s an important contributory factor, but it requires several other contributory factors for the disease process to occur and for the individual to develop ankylosing spondylitis. 05:02 So 90% of individuals that are HLA-B27 positive do not develop ankylosing spondylitis. 05:09 Turning to MHC Class II, these genes are also implicated in the development of autoimmune disease. 05:14 So for example, individuals with Hashimoto’s disease, an autoimmune disease of the thyroid gland, HLA-DR5 is more commonly seen in these individuals, giving a relative risk of three. 05:28 In type I diabetes, HLA-DQ8 confers a relative risk of 14. 05:35 If an individual has both HLA-DQ8 and HLA-DQ2, the relative risk goes up to 20. 05:45 Some MHC genes are actually protective against autoimmune disease. 05:51 And here we see an example where DQ6 gives a relative risk of 0.2 In other words, an individual that has HLA-DQ6 is protected against the development of type I diabetes. 06:06 One of the non-genetic factors that has been implicated in the development of autoimmune disease is infection. 06:14 Usually, encounter with self antigens will lead to tolerance, because there is an absence of co-stimulatory molecules. Here we see a resting dendritic cell that isn’t expressing CD80 and CD86 at any great level, and therefore will not provide co-stimulation to the T-cell. And this will result in self tolerance usually by anergy. However, microorganisms contain Pathogen Associated Molecular Patterns that will be recognized by the Pattern Recognition Receptors on dendritic cells. 06:53 And that recognition will cause the up-regulation of co-stimulatory molecules on the dendritic cell. 07:00 For example the B7 molecules, B7.1 CD80, B7.2 CD86. 07:06 These will interact with the molecule CD28 on the surface of the T-cell causing this self reactive T-cell to become activated leading to autoimmunity, which in some cases may be pathogenic autoimmunity, leading to autoimmune disease. 07:24 There are a number of ways in which infection may lead to the development of autoimmune disease. 07:31 One of them is a phenomenon referred to as molecular mimicry. 07:37 This is caused by a sharing of either structures or sequence between self antigens and microbial antigens. 07:47 So here we have a self reactive T-cell that is also capable of recognizing a microbial peptide, because there’s a sequence in the microbial peptide that is similar to a sequence in a self antigen. 08:01 Therefore there will be activation of T-cells due to this sharing of sequence between the microbial protein and the self protein. 08:10 So the peptide sequence sitting in the MHC will be the same between the microbe and self. 08:17 So a normal immune response if you like, is developed against an infection. 08:22 But unfortunately, some self peptides have the same sequence and therefore the T-cells will recognize not only the foreign microbial proteins but also self proteins again leading to the development of autoimmunity which in some cases may be pathogenic and lead to autoimmune disease.
About the Lecture
The lecture Genetic Susceptibility to Autoimmune Diseases and Mechanisms by Peter Delves, PhD is from the course Hypersensitivity and Autoimmune Disease. It contains the following chapters:
- Mechanisms of Autoimmune Disease
- MHC Associations in Autoimmune Disease
- Role of Infection in the Development of Autoimmune Disease
Included Quiz Questions
Which major histocompatibility complex (MHC) protein is most strongly associated with the development of ankylosing spondylitis?
- HLA-B27
- HLA-B2
- HLA-B7
- HLA-B8
- HLA-B72
Which of the following best describes the action of the protein receptor cytotoxic T-lymphocyte-associated protein 4 (CTLA4)?
- It transmits an inhibitory signal to T cells.
- It transmits a stimulatory signal to T cells.
- It transmits an inhibitory signal to B cells.
- It transmits an inhibitory signal to natural killer cells.
- It transmits a stimulatory signal to macrophages.
Which of the following cytokines mainly has inhibitory effects in the immune system?
- Interleukin-10
- Complement component 4 (C4)
- Interleukin-2
- B lymphoid tyrosine kinase (BLK)
- Complement component 1q (C1q)
Which of the following human leukocyte antigen (HLA) protein is most likely protective against diabetes type 1?
- HLA-DQ6
- HLA-DQ8
- HLA-DQ2
- HLA-DR5
- HLA-B27
What is molecular mimicry?
- A similarity of structures or sequences between self and foreign antigens, leading to autoimmunity
- A B-cell mediated reaction which results in the production of identical autoantibodies
- Destruction of healthy tissue cells because they resemble damaged tissue cells
- Autoimmune complexes that mimic antigens and deposit in lymphocytes
- Inability to clear antigens which are deposited in damaged tissues, leading to tissue destruction
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |