Playlist
Show Playlist
Hide Playlist
Free Radicals Effect on Cellular Activities
-
Slides Cellular Pathology Mechanisms of Injury.pdf.pdf
-
Reference List Pathology.pdf
-
Download Lecture Overview
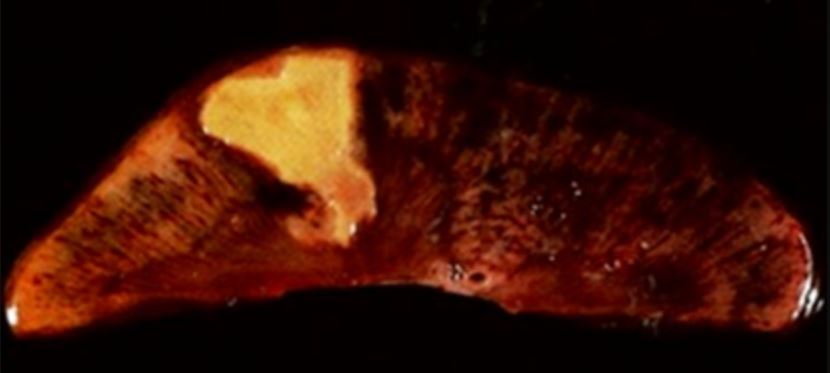
00:00 Right now, let's just look at an example of what happens in terms of protein synthesis, they kind of give this some reality. 00:06 So here we have a bit of a membrane. 00:09 This would be rough endoplasmic reticulum. 00:11 And on the surface of the rough endoplasmic reticulum is ribosome it's translating its protein through into the middle of the looming of the rough ER. 00:20 Life is good, except now we have ingested or inhaled or done something where we've gotten a fair little high dose of carbon tetrachloride. 00:32 This is a compound used in dry cleaning fluid, fairly common, and we metabolize it in enzymes that live in the endoplasmic reticulum. 00:43 In particular the cytochrome p-450 system, which is not a detoxifying system. 00:49 It is a solubilization system. 00:52 It's taking something that's lipid soluble and converting it to something that is more water soluble, so it could be excreted. 00:59 Cytochrome p-450, in that same endoplasmic reticulum, takes our carbon tetrachloride and converts it to carbon trichloride with a free radical. 01:10 Oops, now we have a free radical in the middle of a whole bunch of lipid. 01:15 What's gonna happen? It can be predicted. 01:18 We're going to start in an autocatalytic way, breaking down that entire membrane, and we're getting more and more and more free radicals as because it's autocatalytic. 01:27 As we now break down the rest of lipid, and the lipid is falling apart. 01:33 Our membrane is dissolving, it's become a detergent. 01:36 When that happens, our ribosome goes away. 01:39 It can't bind anymore, so we're protein synthesis goes to zero. 01:43 Not so good. 01:46 Fortunately, we do know that we're going to encounter things like this, so we have ways to sup up, to scavenge free radicals. 01:54 And vitamin E is one of those, it lives in membranes in various places, including the endoplasmic reticulum. 02:00 And it can be a free radical scavenger, so it is a way to potentially stop this. 02:05 There are limits, you only have a certain amount of vitamin E in your tissues, so you cannot make infinite amounts of free radicals and expect to get away with it. 02:13 But it is a mechanism by which we protect ourselves. 02:17 And now there's some other mechanisms by which we protect ourselves from free radicals. 02:23 So here's the effect on cellular activities and death. 02:27 If we look at our normal rough endoplasmic reticulum, and our normal mitochondria, they have a certain structure and architecture, and they look fine. 02:36 If we now hit them with a free radical, in this case, carbon tetrachloride. 02:41 The ribosomes have left rough ER and our mitochondria are swollen. 02:45 So we're not making protein and we're not making ATP. 02:49 When that happens, we will get cell death. 02:51 So this is on the right hand side is a liver. 02:54 In an experimental animal that's been exposed to carbon tetrachloride, and we're getting these areas of necrosis. 03:01 Cell death, cellular homicide and those are pointed to with the arrows.
About the Lecture
The lecture Free Radicals Effect on Cellular Activities by Richard Mitchell, MD, PhD is from the course Cellular Injury.
Included Quiz Questions
Which of the following is responsible for converting carbon tetrachloride to carbon trichloride with a free radical?
- Cytochrome P450
- Cytochrome C oxidase
- NADPH oxidase
- Superoxide dismutase
- Catalase
Which of the following will result from the accumulation of free radicals in the cell?
- Swelling of the mitochondria
- Increased ATP production
- Increased protein synthesis
- Increased activity of the ribosomes
- Release of calcium ions to the mitochondrial lumen
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |