Playlist
Show Playlist
Hide Playlist
Electrophilic Aromatic Substitution – Aromatic Compounds and Eas
-
Slides 06 Chemistry Advanced Le Gresley.pdf
-
Download Lecture Overview
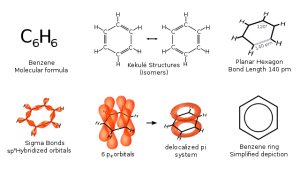
00:01 So, electrophilic aromatic substitution. This is the general default model for this particular reaction and this is one of the most common ones. Because, of course, the benzene ring is electron rich even though the bonds are a bit stronger, you tend to only really see these types of reactions unless you have somehow activated your benzene ring with electron withdrawing groups. 00:25 What happens in this case is that two of the electrons from one of the idealised alkene bonds in our aromatic compound attacks; remember, electrons move, protons do not move, neither do positively charged species. Electrons attack on to, in this case, idealised electrophile, here E+. What this does is that it affectively breaks open or destroys the aromaticity of that ring temporarily. We note we only have two double bonds now. We don’t have 6 pi electrons. 00:56 And so, what then happens in order to reform the stability associated with the aromatic compound is one or two things must happen. Either E+ is lost again, just so that we can regenerate aromaticity, or we lose the single hydrogen that is attached to the carbon on which the electrophile becomes attacked. 01:17 In this case, we have shown a successful electrophilic aromatic substitution where we lose H+ and a new substituent in its place is E, our electrophile. E+ is usually an electrophile which is generated in situ via a preliminary reaction. 01:34 And these are the different reaction types. Each of these mechanisms is almost identical to the one I have shown you earlier. 01:42 Halogenation is achieved via Cl+/Br+ which is usually generated in situ, for example, bromine in the presence of iron bromide. Nitration, which is generated in situ via the reaction of sulphuric acid and nitric acid to give the NO2+ electrophile. Sulfonation, usually it is sulphur trioxide in the presence of acid. This gives rise to a sulphonation electrophile SO3H+. Alkylation is one of the more desirable reactions that you want to do on an aromatic compound and that cannot directly be achieved by reacting it with, for example, an alkyl halide. You cannot just add that over a double bond. Friedel-Crafts Alkylation takes place in the presence of a Lewis acid catalyst. Usually, for example, in the presence of a chloroalkane, you would use something like aluminium trichloride to facilitate that reaction. 02:42 And the same applies with Friedel-Crafts Acylation. This is particularly important when you want to introduce an acyl group. This effectively will resemble a ketone group being formed on your benzene ring. 02:56 Usually, as we will see, we use something like an acid chloride again in the presence of an aluminium trichloride catalyst. Electrophiles are formed in situ in a preliminary step. 03:09 So, here we have an example of such an electrophilic aromatic substitution taking place. We have substituted E+ for Cl+ and in the first step, we can see the electrons from the benzene ring, taking it from an idealised double bond, attack our electrophile leaving electrons on to that and forming a sigma bond between the two. What we then see is this intermediate, this positively charged species which has had the aromaticity of the ring disrupted. 03:37 The second step involves the elimination of the H+ and the real formation of the benzene ring and is this reformation of the benzene ring which is a driving force. And this is important to bear in mind, where there is the possibility for a structure to adopt an aromatic state, this is usually good energetic reason why it would take place.
About the Lecture
The lecture Electrophilic Aromatic Substitution – Aromatic Compounds and Eas by Adam Le Gresley, PhD is from the course Organic Chemistry.
Included Quiz Questions
Which of the following is an INCORRECT statement?
- Friedel-Crafts alkylation of an aromatic compound does not need any Lewis acid as a catalyst.
- During the chlorination of benzene, aluminum chloride acts as a catalyst.
- During nitration of benzene, in situ generation of an electrophile (NO2+) takes place by a reaction between sulfuric acid and nitric acid.
- Upon heating with conc. H2SO4, benzene forms benzenesulfonic acid as a product.
- The production of phenylethanone from benzene and ethanoyl chloride at 60°C in the presence of AlCl3 is an example of Friedel-Crafts acylation of benzene.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |