Playlist
Show Playlist
Hide Playlist
Catabolism and Anabolism — Metabolism and Regulation
-
14 Basic PrinciplesOfMetabolism2.pdf
-
Biochemistry Free and Easy.pdf
-
Reference List Biochemistry.pdf
-
Download Lecture Overview
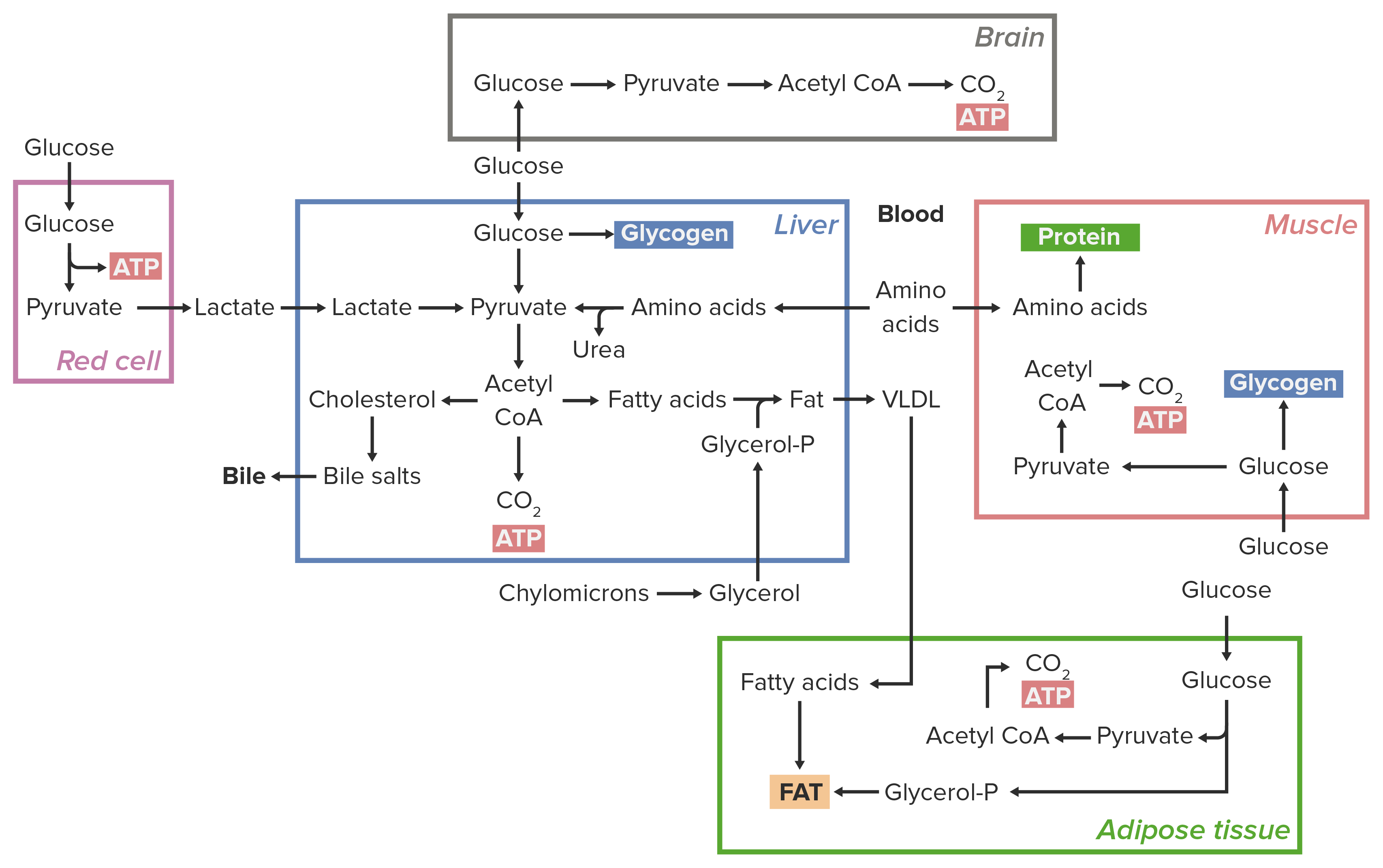
00:01 Um leben zu können, müssen Zellen Energie erzeugen und diese Energie zur Herstellung von Molekülen verwenden. 00:07 Das ist die Funktion dessen, was wir Stoffwechsel nennen. In dieser Präsentation werde ich Ihnen die Prozesse des Katabolismus und Anabolismus und Energiebetrachtungen im Hinblick auf biochemischen Reaktionen sowie die Regulierung dieser Mechanismen erläutern. 00:22 Zunächst muss ich einige Begriffe definieren. Katabolismus ist ein Prozess, bei dem größere Moleküle, die die Zelle aufnimmt, in kleinere Bestandteile zerlegt werden. Sie können auf dem Bildschirm sehen, dass zum Beispiel Proteine, Polysaccharide und Fette jeweils in Aminosäuren, Zucker und Fettsäuren zerlegt werden. Diese Vorläufer oder diese Bausteine, werden zur Herstellung von beispielsweise Muskel-, Nerven- oder Knochenmasse benötigt. 00:48 Eine Vielzahl dieser Komponenten, die hergestellt werden, hat einen gemeinsamen Punkt, auf den sie alle zulaufen, und as ist Acetyl-CoA. Acetyl-CoA kann im Citratzyklus leicht oxidiert werden, um ATP-Energie zu erzeugen. 01:01 Der Prozess der Oxidation erzeugt natürlich Elektronen und diese Elektronen muss die Zelle irgendwie unterbringen. Die Elektronen in den Zellen werden auf Elektronenträger, entweder NAD+ oder FAD, übertragen. Wenn NAD+ Elektronen und ein Proton aufnimmt, wird es zu NADH, wie Sie in der Präsentation sehen können. 01:22 Der Prozess des Anabolismus ist im Wesentlichen das Gegenteil des Katabolismus, d.h. kleinere Moleküle werden zu Vorläufern aufgebaut und diese Vorläufer werden zu den Bausteinen der einzelnen Körperteile wie etwa Muskelmasse gemacht. 01:33 Wir können hier zum Beispiel sehen, dass die Vorläuferstoffe zu Aminosäuren, Zuckern und Fettsäuren zur Herstellung von Proteinen, Polysacchariden bzw. 01:43 Fetten aufgebaut werden. Weil dieser Prozess Energie benötigt, ist ein ATP-Einsatz erforderlich und das erzeugte ATP von den katabolen Prozessen wird verwendet, um die Moleküle hier in den anabolen Prozessen herzustellen. 01:57 Der Anabolismus ist in der Regel reduktiv, d.h. er benötigt Elektronen und damit Elektronenquellen, um diese größeren Moleküle herzustellen. Elektronenquellen gibt es in vielen Bereichen, aber normalerweise kann NADH verwendet werden. 02:13 Wie ich bereits in der vorherigen Präsentation erwähnt habe, werden Zellen von den Regeln des Universums bestimmt. 02:17 Sie können nicht die Art und Weise verletzen, in der Energie verwendet und gespeichert wird, also müssen sie innerhalb diese Zwänge agieren. Betrachten wir nun zum Beispiel, was Zellen bei der Oxidation von Glukose tun müssen. 02:30 Dieser Stoffwechselweg ist als Glykolyse bekannt und beinhaltet in der allerersten Reaktion die Anlagerung eines Phosphats an Glukose. Wenn die Zelle versuchen würde, einfach ein Phosphat auf Glukose zu packen, würde es zu der Reaktion kommen, die ganz oben dargestellt ist, wo Glukose an ein Phosphat angefügt wird, wodurch ein Molekül entsteht, das als Glucose-6-phosphat bekannt ist. Jetzt ist delta G (ΔG0') für diese Reaktion +15 kJ pro Mol. Es ist möglich, diese Reaktion ablaufen zu lassen, aber dieser sehr positive ΔG0'-Wert stellt eine Art Hindernis dar und macht es schwierig für die Zelle, Glukose-6-Phosphat herzustellen. Anstatt also zu versuchen, diese Reaktion übermäßig zu forcieren, haben die Zellen eine weitere Alternative, die sie nutzen können, um die Reaktion zu begünstigen. Die Überlegung, die die Zellen haben, ist, dass sie eine Energiequelle nutzen und die Energiequelle, die sie haben, ist ATP. Nun kann ATP, wie Sie in der zweiten Zeile sehen können, mit Wasser hydrolysiert werden, um ADP plus Phosphat zu erzeugen. Nun ist das ΔG0' für diese Reaktion sehr negativ, -31 kJ pro Mol. 03:37 Was die Zellen also tun, um die Barriere der ersten Reaktion zu überwinden, ist, dass sie diese mit der zweiten Reaktion koppeln. Wenn diese beiden Reaktionen zusammengeführt werden, wird die Reaktion insgesamt energetisch günstig. In diesem Fall wird Glukose plus ATP verwendet, um Glukose-6-Phosphat plus ADP erzeugen, und für diese Reaktion beträgt ΔG0' -16 kJ pro Mol, das ist die Summe der beiden vorherigen Reaktionen. Durch die Paarung wird also eine Energie freisetzende Reaktion ausgelöst, auch Kopplung genannt, mit der Zellen Energiebarrieren überwinden können.
About the Lecture
The lecture Catabolism and Anabolism — Metabolism and Regulation by Kevin Ahern, PhD is from the course Biochemistry: Basics.
Included Quiz Questions
Which of the following is true of catabolism?
- The larger molecules are broken into smaller ones.
- Energy is required for catabolism.
- The process is usually non-oxidative.
- Polysaccharides are made.
- It is the process of making larger molecules from smaller ones.
Which of the following is true regarding energy coupling?
- It makes energetically unfavorable processes more favorable.
- It is usually the mechanism of making ATP.
- It is necessary to make catabolic processes occur.
- It occurs when NAD+ is produced.
- It occurs when FAD is produced.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
Made something so complex easy. I've always had trouble here.