Playlist
Show Playlist
Hide Playlist
Biologically Relevant Examples – Aromatic Compounds and Eas
-
Slides 06 Chemistry Advanced Le Gresley.pdf
-
Download Lecture Overview
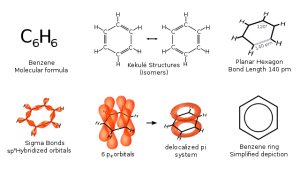
00:00 So, that’s basically for the nomenclature. 00:03 So, let’s move on to biologically relevant examples of such benzene or aromatic-containing compounds. So, natural amino acids are tyrosine and phenylalanine and also, if you look carefully tryptophan also contains a benzene ring system. This is part of an overall indol ring which is beyond the terms of reference for this lecture, but you may wish to investigate it further. 00:27 Note how we have monosubstitution in the case of phenylalanine and 1,4 di-substitution in the case of the tyrosine ring. Bearing in mind, however, these are, of course, the accepted names for these amino acids. The proper IUPAC name would be a lot more complicated, but thankfully, when you are either purchasing these for experiments or when you see these described, they will always use these names given for the amino acids. 00:55 A little bit more complicated and something which has considerable biological relevance is folic acid. This is this large molecule shown here. Vitamin Bc or pteroyl L-glutamic acid is the other name and aside from being essential for the production and maintenance of new cells, not least because it’s responsible for the conversion of, for example, uracil into thiamine, which is very important via methylation reaction, which is again beyond the terms, is also a very, very important molecule when you consider, for example, the role of sulfa drugs in the treatment of certain bacterial infections because, indeed, pteroyl synthase, which is the enzyme responsible for the conversion of para-aminobenzoic acid by bacteria into this folic acid, is inhibited by those sulfa drug. And so, an appreciation and understanding of what you can make that mimics a natural substrate is important in the biological sense. 01:54 So, what do we mean by aromatic? The problem is, it is used by organic chemists in a rather different way to that which is normally applied. The origins of it are effectively from folklore where, for example, cinnamon bark, wintergreen leaves… wintergreen leaves, vanilla beans and anise seeds contain these fragrant compounds that had common, but also unexpected properties. 02:20 Such compounds, as we have seen, have a very low carbon to hydrogen ratio by virtue of the double bonds. It was, therefore, hypothesised that they may indeed contain more than one. 02:30 Nevertheless, they were much more stable than the correlating alkenes and did not give some of the double bond typical reactions. 02:38 You may recall when we were talking about alkenes in one of the previous lectures within this modules that we talked about the idea of decolourising bromine water, a way of actually qualitatively identifying an alkene is effectively to add some bromine water to it or vice versa and observe the decolourisation as the addition reaction takes place to give rise to a halohydrin, or in the case of just pure bromine, a dibromoalkane which is colourless. 03:07 But, benzene does not give this reaction. This suggests that the double bonds themselves are actually not as prone to addition and therefore, cannot be considered to be as nucleophilic as conventional alkenes. 03:22 One of the first identified aromatic compounds, the structure of which I have already shown you, is benzene.
About the Lecture
The lecture Biologically Relevant Examples – Aromatic Compounds and Eas by Adam Le Gresley, PhD is from the course Organic Chemistry.
Included Quiz Questions
Which of the following is one of the characteristics of benzene?
- Higher stability than that of an alkene.
- Ability to decolorize bromine water.
- Absence in the structure of proteins due to its toxic nature.
- Presence in almost all aromatic compounds found in nature.
- Relatively high carbon-to-hydrogen ratio.
Which of the following is NOT a correct statement about folic acid?
- It is a non-essential fat-soluble vitamin.
- The sulfonamide antibiotics inhibit dihydropteroate synthase (DHPS), a key enzyme in the folate pathway of bacteria.
- Folic acid takes part in the synthesis and repair of nucleic acids.
- Pteroyl-L-glutamic acid helps in the production and maintenance of new cells.
- Folate acts as a coenzyme in the metabolism of amino acids like methionine, cysteine, serine, glycine and histidine.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |