Playlist
Show Playlist
Hide Playlist
Biological Interactions: Introduction
-
Slides 10 Chemistry Advanced Le Gresley.pdf
-
Download Lecture Overview
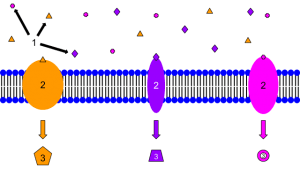
00:01 Hello. 00:01 What we’ll be doing is taking a number of different modules: Modules I, II and III, bringing them together so that we can see the biological and, hopefully, medical application of some of those principles which we’ve learnt. 00:13 We’re going to start off by looking at basic biochemical interactions to best understand the concept of receptors as drug targets. Specifically, we’ll be looking to introduce the concept of agonists and antagonists of these receptors. 00:30 And consider receptors not just as enzymes which, as we will see, facilitate the changes in matter or the conversion of one molecule into more than one molecule or, sometimes, just its functional alteration. But also, we’ll be looking at the types of interaction. 00:47 And this is where the functional group comes into play. 00:50 If you recall from Module III, what was so important is that you understood the basics of the individual functional groups, everything through from carbonyl compounds through to amides. And this is vital in terms of understanding the types of interaction which drugs may make with their targets be they, for example, enzymes receptors or indeed some of these macro-molecular structures that you find, such as DNA. 01:16 We’ll also be considering the role of water in drug-receptor binding and furthermore, considering the impact of this on the thermodynamics and entropy of the interaction as well. 01:27 Also, we’ll be considering the roles of confirmation and shape. So, this is not just about the electron density at a particular part facilitating the best interaction, but also, as we will see, the shape which forces the adoption of the best possible orientation for receptor binding. 01:46 So, when we go back in history, when researchers first began to isolate active ingredients, most different things like plants or, occasionally, from purified water organisms such as plankton and so on so forth, it was not known how they exerted their effects specifically on the body. It was known that they had an effect, but it wasn’t sure what or why or how the effect was mediated. 02:15 Gradually, chemical structure came to be linked to biological activity. And this is something called structure-activity relationship. By varying the structure, we can actually alter the biological activity of our putative drug or indeed, our lead compound as a putative drug. 02:33 For example, it was discovered that certain compounds, aromatic ones as well, with a quaternary amine were often found to either relax or contract a muscle. If you look at the bottom right hand side near the microscope, you’ll see that there is an example here of a quaternary amine. 02:50 Note, the difference between a tertiary and a quaternary is, in this case, you have a permanent positive charge. This is conveyed by the complete loss in the formation of a sigma bond of the lone pair of electrons on the nitrogen. Quaternization, which is the process by which this is achieved, can be accomplished via a number of different ways.
About the Lecture
The lecture Biological Interactions: Introduction by Adam Le Gresley, PhD is from the course Medical Chemistry.
Included Quiz Questions
A biological target of a pharmacologically active drug is which of the following?
- Some entity in a living organism to which the binding of a drug results in a change in its behavior or function.
- A covalent bond between water molecules.
- A hydrogen bond between water molecules.
- An ionic bond in the structure of a base molecule.
- An ionic bond in the structure of an acid molecule.
Which of the following can NOT be a target of an active drug molecule?
- Water
- Nucleic acids
- Protein molecules
- Hormone receptors
- Enzymes
What does it mean by structure-activity relationship (SAR)?
- The dependence of the biological activity of a chemical on its molecular structure.
- The dependence of the biological activity of a chemical on the molecular structure of water molecules.
- The independence of the biological activity of a chemical on the molecular structure of water molecules.
- The dependence of the biological activity of a chemical on the molecular structure of the table salt molecules.
- The independence of the biological activity of a chemical on its molecular structure.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |