Playlist
Show Playlist
Hide Playlist
Benzene – Aromatic Compounds and Eas
-
Slides 06 Chemistry Advanced Le Gresley.pdf
-
Download Lecture Overview
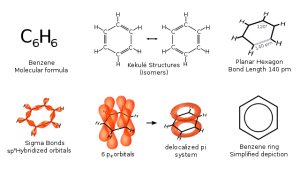
00:00 So, all carbon-carbon bonds are actually an intermediate between a single and a double bond. If you recall when we were talking about mesomeric effects, right back at the first module, we were talking about the ability of a double bond to resonate throughout a particular structure where more than one double bond exists. 00:20 As you can see from the diagram shown here at the bottom, you can see that we… that it is possible for the double bonds to move around in almost snake-like fashion. This is due to the fact that the real structure of benzene is a hybrid between these two resonant forms; a standard carbon-carbon double bond is shown here as 1.34 angstroms, a standard carbon-carbon single bond is shown as having a bond length of 1.5 angstroms. However, the half way house length in benzene is 1.39 angstroms, somewhere between the two, suggesting it is not quite as long as a carbon-carbon alkane, but not nearly as short as the conventional carbon-carbon double bond. 01:05 So, the problem is that once these two structures are effectively degenerate, you may have recalled that phrase when we were talking about orbitals because you can superimpose them, they are also equivalent to make equal contributions to the hybrid. However, the benzene structure is badly represented in fairness by either of these two examples structure one or two. 01:27 It actually has a structure which is an intermediate between the two being more stable than both of them. And so, what you will sometimes see is a benzene ring… benzene ring being abbreviated as effectively a hexagon with a circle in the center such as that shown here on the board. 01:43 The reality when we actually look at the orbital system for benzene shouldn’t come as a huge surprise. As you can recall from hybridisation, you should be aware that SP2 hybridisation correlates to the hybridisation of 1S and 2P orbitals. This results in a single P orbital being unhybridised. And as you can see here, each carbon on our benzene ring has that P orbital which has been unhybridised… not been hybridised. 02:14 And what this effectively allows to happen is the electron in each of those is able to delocalise around the entire ring structure such as that which is shown on the diagram in the top corner. As all of the carbon atoms are SP2 hybridised, you effectively end up with something which is a planar model… molecule with bond angles of 120 degrees. 02:42 Contrast that, if you will, with the actual structure and geometry of a cyclohexyl ring. 02:49 We haven’t necessarily covered that geometry, but look it up. You will see it exists as a chair or boat or somewhere in between, something that forces the carbons and hydrogens to be as far apart as possible to minimise steric interference. Benzene rings, on the other hand, are planar.
About the Lecture
The lecture Benzene – Aromatic Compounds and Eas by Adam Le Gresley, PhD is from the course Organic Chemistry.
Included Quiz Questions
In the molecular orbital description of bonding in benzene (C₆H₆), how many electrons occupy the delocalized molecular orbitals?
- 6
- 5
- 2
- 3
- 0
Why does benzene not give a positive qualitative test when treated with bromine water?
- Because the electrophile is not able to attack extensively delocalized pi-electrons over the six carbon nuclei structure of benzene.
- Because the six hydrogen atoms present in benzene cause a steric hindrance.
- Because the sp3 hybridized carbon atoms create a spatial barrier.
- Because the number of pi electrons on the benzene ring is insufficient.
- Because the pi electrons are not properly delocalized over the benzene ring.
What is the correct carbon-carbon bond length in benzene?
- 1.39 Å
- 1.09 Å
- 1.54 Å
- 1.34 Å
- 1.22 Å
Which of the following is INCORRECT about benzene?
- Benzene is an energetically highly unstable structure.
- Benzene exhibits a high degree of unsaturation with six pi electrons.
- Benzene gives substitution reactions rather than addition reactions.
- Benzene gives a negative bromine water test and Baeyer test for unsaturation.
- Benzene is a highly stable molecule with a planar structure.
Complete the following statement. All carbon atoms in the benzene molecule exhibit …
- … sp² hybridization.
- … sp hybridization.
- … sp³ hybridization.
- … sp³d hybridization.
- … sp³d2 hybridization.
What is the cause of the excellent chemical and thermodynamic stability of benzene?
- Capabilities of pi electrons to delocalize around the entire ring.
- A high degree of unsaturation.
- A low carbon to hydrogen ratio.
- A chair shaped ring geometry to avoid steric hindrance.
- A boat shaped ring geometry to prevent steric hindrance.
Complete the following statement. The term resonance is used to define ...
- … the delocalization of electrons within a molecule having alternating single and double bonds.
- … a permanent state of polarization of a bond between two atoms with variable electronegativity values.
- … an intramolecular movement of pi electrons from a double bond to another atom under the effect of an attacking reagent.
- … the delocalization of sigma bond electrons of an alkyl group to an attached atom with an unshared orbital.
- … the delocalization of electrons within a molecule having alternating double and triple bonds.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |