Playlist
Show Playlist
Hide Playlist
Aspartate Family and Asparagine Metabolism
-
Slides AminoAcidMetabolism Biochemistry.pdf
-
Reference List Biochemistry.pdf
-
Download Lecture Overview
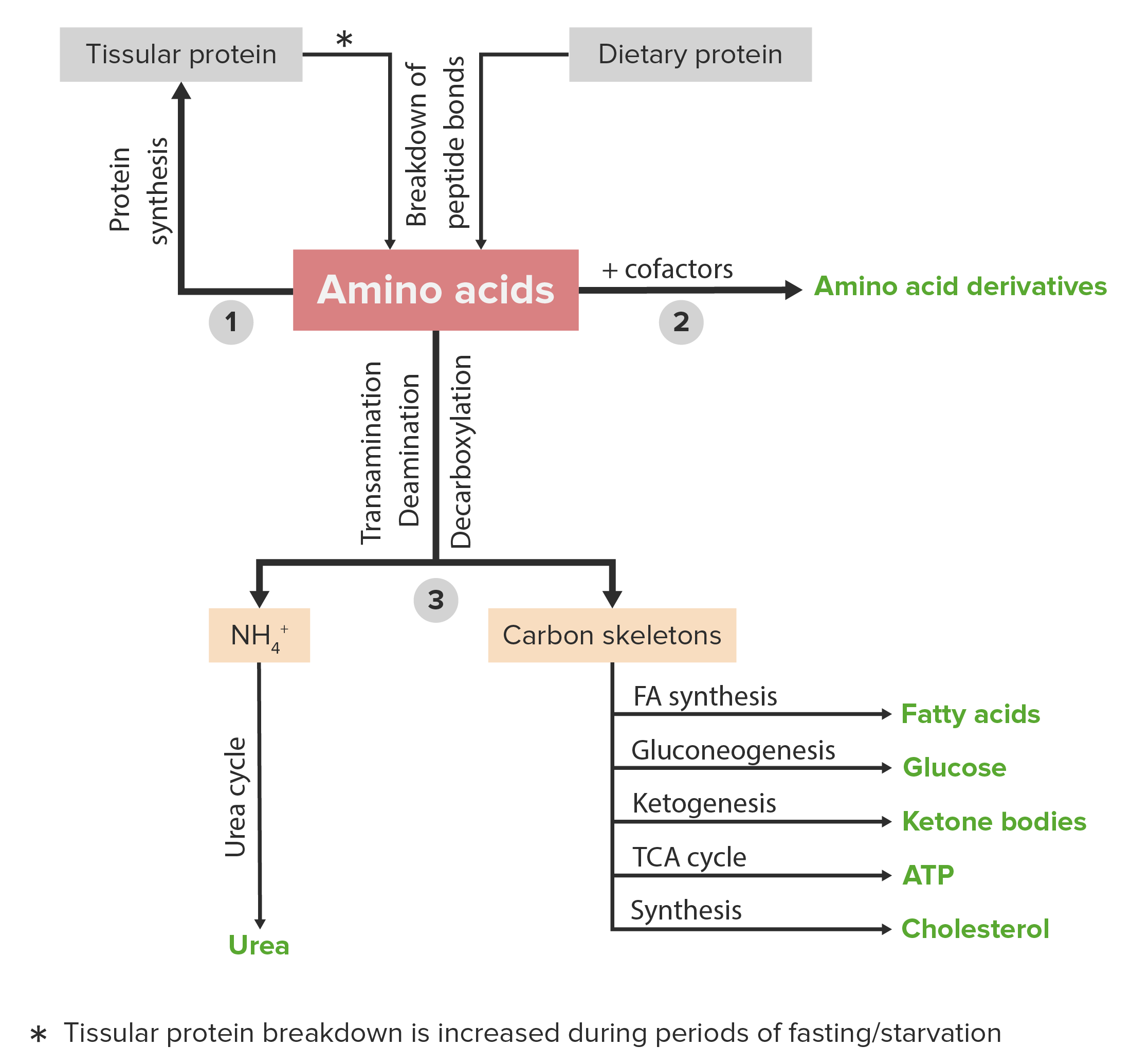
00:01 The next stem we'll be considering our reactions is that of the aspartate family. 00:05 The aspartate family, as its name suggests, all involve aspartate. 00:10 And aspartate can actually trace its roots back one further molecule and that's oxaloacetate as you can see here. 00:17 All family members in this metabolic pathway come from aspartate. 00:22 Aspartate can actually also be made from one of them. 00:24 So there's reversed reaction that can produce aspartate as we will see. 00:29 There are numerous ways that lead to aspartate. 00:32 So I'm only going to talk about a couple of them. 00:35 One of the ways and one of the more common ways in which aspartate can be made is by the process of transamination that we've already talked about. 00:43 Here's one sample transamination and there are many different transaminations that can produce aspartate. 00:51 One starts with glutamate and oxaloacetate. 00:53 Glutamate being the amino acid that is the source of the amine. 00:56 Oxaloacetate being the alpha keto acid which is the source of the oxygen. 01:01 The product of that transamination, the glutamate becomes alpha-ketoglutarate, and the oxaleacetate becomes aspartate. 01:10 Amine has come from the glutamate, onto the aspartate, the oxygen has come from the oxaloacetating, becomes the alpha-ketoglutarate. 01:17 Another way of making aspartate is by hydrolysis. 01:20 This is a reversal of the synthesis of asparagine in a way. 01:25 Now, we'll see later, it's not the exact reversal of the synthesis of asparagine. 01:31 Asparagine can be cleaved using water in a hydrolysis reaction to produce aspartate and there's that ammonium ion again. 01:39 Ammonia is a toxic compound. 01:42 The cell meets the aspartate, they're going to do this reaction. 01:45 If the cell was breaking down asparagine, it's got too much asparagine, he'll do this reaction. 01:50 But that ammonium ion has to be gobbled up somewhere or it's going to cost a problem. 01:56 The third way of making aspartate involves the urea cycle. 01:59 The Urea cycle involves the cleavage of argininosuccinate with AMP to make aspartate and citrullyl AMP. 02:08 Now this is kind of a complicated reaction that I won't go into here, but will be discussed later in another one of these lectures in the urea cycle. 02:17 As I note, it's a reversal of a reaction that occurs normally in the urea cycle. 02:21 So normally the reaction, the forward direction of the urea cycle goes upwards. 02:26 The synthesis of asparagine occurs from aspartic acid in a fairly simple fashion. 02:32 We can see here that if we start with aspartate and glutamine and we add ATP, we can create asparagine and glutamate plus AMP. 02:42 Now this is a transamination but this is a transamination that's a little bit different in the sense that it's requiring additional energy to do that. 02:51 Why does that happen? Well, the reason that that happens is we're not putting the amine onto the alpha keto group. 02:58 We're actually putting the amine onto the carboxamide group at the end of asparagine. 03:03 That requires energy. 03:05 And because that requires so much energy, we see ATP becoming AMP. 03:10 When we see that happen, we know there's a lot of energy involved. 03:13 When that happens, it means that this reaction, even though it's drawn in a reversible fashion, isn't very practical in the reversed direction. 03:22 The enzyme catalyzing this reaction is asparagine synthetase. 03:26 It's energetically costly and essentially not reversible. 03:29 So the other way of course of making aspartate and the much more common way of making aspartate from asparagine was by the breakdown of asparagine that involves the use of water, production of aspartate and the enzyme catalyzing that is known as asparaginase. 03:42 This is the most common way that asparagine is broken down and the most common way that aspartate is produced from asparagine. 03:49 We produce the ammonium ion in this process. 03:52 We remember that's toxic and it will have to be gobbled up in another reaction.
About the Lecture
The lecture Aspartate Family and Asparagine Metabolism by Kevin Ahern, PhD is from the course Amino Acid Metabolism. It contains the following chapters:
- Aspartate Family
- Aspargine Metabolism
Included Quiz Questions
Which amino acid is NOT part of the aspartate family?
- Cysteine
- Lysine
- Threonine
- Methionine
- Asparagine
Which is true about the reaction catalyzed by asparagine synthetase? Select all that apply.
- There is a high energetic cost.
- It involves pyruvate and alanine for transamination.
- It produces ATP.
- It produces AMP.
Which of the following reactions give rise to aspartate? (Select all that apply).
- Transamination of oxaloacetate and glutamate
- Asparagine hydrolysis
- Folate metabolism
- Transamination of selenocysteine
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |