Playlist
Show Playlist
Hide Playlist
Aromatic Compounds – Aromatic Compounds and Eas
-
Slides 06 Chemistry Advanced Le Gresley.pdf
-
Download Lecture Overview
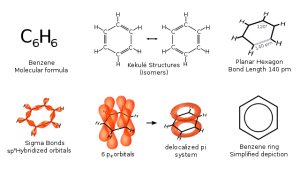
00:01 So, how do we know if the compound we are looking at which has more than one double bond or two or three is actually aromatic? And here we turn to the Huckel rule which is shown in the middle of the board, 4n+2 pi electrons. So, we have to look at the number of pi electrons in our polyalkene structure. 00:24 Let’s have a look, for example, at benzene, where we take n to be the ring size and we see that we have one ring, 4 x 1 is 4 + 2 is 6. Do we have 6 pi electrons? Yes, we do. 00:39 So, we can say that this is aromatic. 00:41 Let’s apply the same for naphthalene. Two ring systems. 4 x 2 is 8 + 2 is 10. Do we have 10 pi electrons? Count the bonds. Yes, we do. Therefore, this is aromatic and so on and so forth. And sometimes it’s actually difficult to see when you are dealing with a very large molecule. But, as long as within the ring system you can see that 4n +2 and they are adjacent to each other, you can usually be confident that they are aromatic. 01:10 There are some other examples of aromaticity that you see in, for example, a deproteinated pyran ring. This is beyond the terms of reference of this lecture, but it’s something that should be borne in mind that aromaticity is not just restricted to carbon only containing species. 01:24 Right. Okay. So, characteristics of aromatic compounds that have a high degree of unsaturation, we have already said this, they give substitution rather than addition reactions. They don’t react with bromine water. They are highly stable in comparison and they have a planar structure. 01:43 Let’s have a look and see what that actually means. All the four bases contain aromatic rings of one description or another. And therefore, all of them are planar. Because of this not only they do form specific hydrogen bonds between the pairs that hold those individual bases together, but they also contribute to the double helix structure, as you can see in the rotating diagram on the board. 02:09 You can see here how this effectively shows a Watson-Crick or duplex based pairing in a stacked planar array. And indeed, what’s just as important, is this stacking of the aromatic groups is not just a feature of duplex DNA, but also of quadruplex DNA. And it’s worthy of note that quadruplex DNA is proving highly interesting target in anti-cancer treatment and you should be looking that out as well. 02:39 Let’s have a quick look at what I mean by the aromaticity of the individual bases. Here we have guanine bonding via hydrogen bonds to cytosine, adenine bonding via hydrogen bonds to thymine. Note what I said before. You may think, “Well, hang on a second. 02:54 It’s got nitrogen groups in it.” But, the reality is this is still an aromatic. 03:00 If you look, for example, at the cytosine, it is possible for that to tautomerase and for the double bond to form between a carbon and nitrogen, thus creating your 6 pi electrons in a cyclic state. Adenine and guanine are known as the purine bases. This is because of that structure as you can see with the two nitrogens on what appears to be a cyclopentene ring whereas cytosine and thymine are known as the pyrimidines because the thing is they do of a benzene ring with carbons in two places, one free to each other, have been substituted by nitrogen.
About the Lecture
The lecture Aromatic Compounds – Aromatic Compounds and Eas by Adam Le Gresley, PhD is from the course Organic Chemistry.
Included Quiz Questions
Which of the following is an INCORRECT statement about a molecule exhibiting an aromatic character?
- Hückel’s rule does not stand true for the aromatic compound.
- The aromatic molecule has a planar structure.
- The aromatic molecule follows Hückel’s rule (4n+2).
- The aromatic molecule is cyclic.
- Every atom in the ring participates in delocalization of electrons by having an unhybridized p-orbital or an unshared pair of electrons.
Which of the following is not an aromatic compound according to Hückel’s rule?
- 1,3,5,7-cyclooctatetraene
- Naphthalene
- Anthracene
- Benzene
- Phenanthrene
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |