Playlist
Show Playlist
Hide Playlist
Viral Culture and Virus Quantification
-
Slides 04 Viruses MicrobiologyAdvanced.pdf
-
Download Lecture Overview
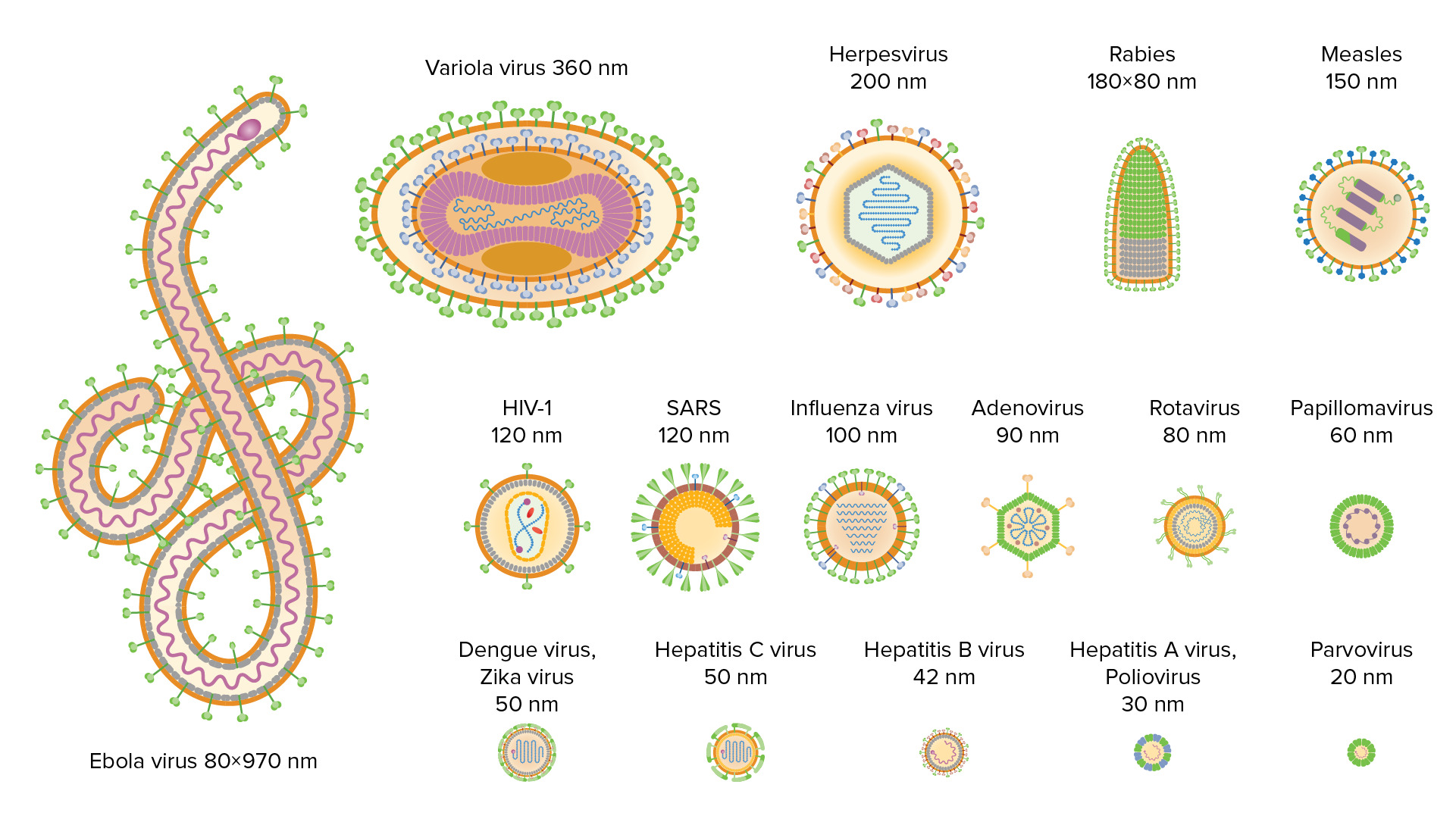
00:00 In my laboratory, where we study viruses, we grow lots and lots of cells, you can grow cells in two different ways. On the left side of this slide is a photograph of what we call a spinner culture. These are suspension cultures of cells. The flask, these glass flasks that you see here have a magnet suspending in them and that is turned by a magnet in the structure underneath them and so these are constantly spinning and the cells are growing in a liquid medium. On the right we can also grow cells in what we call monolayer cultures. Here they're growing in plastic dishes or flasks, covered with mediu. And I think you can see at the back of this incubator me actually taking this photograph. This is a true selfie, CELLFIE, all the other ones are fake. When you grow viruses in cells, it can be very easy to detect the growth, because many viruses kill the cells that they replicated in. And this is called cytopathic effects. And this is a series of four photographs where we were tracking the cytopathic effects of poliovirus in an infected culture. On the upper left is a monolayer of uninfected cells, these cells were then infected with poliovirus. And then four hours later, the picture on the upper right shows you the effects of the virus very early in replication, the cells are becoming rounded and detaching from the monolayer. At eight hours post infection, the lower left-hand part of the slide, you can see most of the cells are now detached. And by 12 hours, on the lower right, all of the cells are detached and many of them have broken up. So this is called cytopathic effects. It is an easy way to tell that your virus is replicating or growing in cells. 01:50 Another important aspect of studying viruses is to detect the virus in the sample, to know how much virus is present. And I want to tell you two different ways that we do this; one is we can measure infectivity of the viruses, we can do that by a plaque assay which is shown here and we can also measure physical virus particles, or their components. This photograph is an electron micrograph of virus particles and you could count them for example, and know how many particles are in a preparation. Infectivity assays actually tell you how many infectious viruses are present, physical particle measurements only tell you the particles, because not all virus particles are infectious. Let's explore the plaque assay a bit. In this assay, we take a virus stock and we assume there's a lot of virus in this virus stock. 02:46 And then we have to make tenfold serial dilutions of the stock in order to obtain a dilution which has a reasonable number of particles in it. So here we're making tenfold serial dilutions and we end up with -1, 2, 3, -4, etc dilutions of the virus. We then take a small amount of each dilution and we add it to a monolayer of cells in a plastic dish, we cover the monolayer with an agar overlay and we incubate it. The viruses will infect cells and they will destroy the cells in a small area. They are restrained in their diffusion by the agar overlay and they form what we call plaques. We can then stain the plaques and count them. So earlier I said we need to make dilutions because we need to find a suitable number of viruses and I think the three plates shown here illustrate that. The one on the left part of the slide, there are too many plaques, we can't count these with any accuracy. The one in the middle has 17 plaques, which is a reasonable number. Anything between 10 and 1 or 200, depending on the size of the dish will work. And then on the far right, only two plaques. This does not provide a statistically rigorous number. From the 17 plaques, we know the dilution that was used to produce that plate, we can calculate the titer of this virus stock at 1,7×10 to the eighth plaque forming units per milliliter. 04:10 It's very important, this is plaque forming units, we can't say virus particles because we don't know for this particular virus how many viruses form a plaque. So that's an example of a plaque assay, this is used in many laboratories, it was developed in the 50s and it is still used today because it's incredibly powerful. In fact in my laboratory we do them all the time and I've built in my office what I call the wall of polio. This is a stack of plates used to do plaque essays. I've glued them all together myself. This is what I do for relaxation and when people visit my lab, I take their picture in front of the wall of polio, so if you come to visit me, please ask and I will take your picture as well. 04:52 Physical measurements of viral particles can be done in a variety of ways. 04:56 We can use a technique called hemagglutination. 04:59 Some viruses bind red blood cells and we can use that to make a physical measurement of particles. 05:05 We can look at the virus particles by electron microscope as I showed you earlier. 05:11 This is not so convenient because not everyone has an electron microscope. 05:15 We can measure viral enzymes in the virus particle. 05:20 Many viruses package into the particle and enzyme, and many assays have been devised to take advantage of that. 05:26 We can use serologic techniques to measure virus particles and their components. 05:32 We can use antibodies against viruses and their proteins to measure how much of each one is there. 05:38 And we can also measure nucleic acids now by a variety of techniques.
About the Lecture
The lecture Viral Culture and Virus Quantification by Vincent Racaniello, PhD is from the course Viruses.
Included Quiz Questions
Which of the following assays measures infectious viruses?
- Plaque assay
- Serological methods
- Hemagglutination assay
- Electron microscopy
- Measuring viral enzymes
What do scientists look at when attempting to detect the cytopathic effect of viruses?
- Cell detachment and destruction
- Visible change on light microscope
- Speed of cell replication
- Cell swelling with transformation into a phagocyte
- Formation of cell's nuclear envelope, decreased cell cytoplasm
What is the difference between infectivity assays and viral assays?
- Viral assays measure how many viral particles are present.
- Viral assays measure how many particles can infect the host.
- Infectivity assays measure how many viral particles are present.
- Infectivity assays measure the viral load.
- Viral assays measure the ability to produce disease in the host.
Which of the following cultures are used to grow viruses in laboratories?
- Spinner culture
- Sabouraud culture
- Blood culture
- Dextrose culture
- TSA culture
Which effect causes most cell breakdown during viral replication?
- Cytopathic effect
- Warburg effect
- Von Willebrand effect
- Mitochondrial effect
- Virotic effect
How can viruses in a sample be measured?
- By measuring the infectivity of the virus
- By spinner culture
- By monolayer culture
- By dextrose culture
- By TSA culture
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |