Playlist
Show Playlist
Hide Playlist
Type III Hypersensitivity: Manifestations in Different Diseases
-
Slides Immune-mediated Diseases Type III.pdf
-
Reference List Pathology.pdf
-
Download Lecture Overview
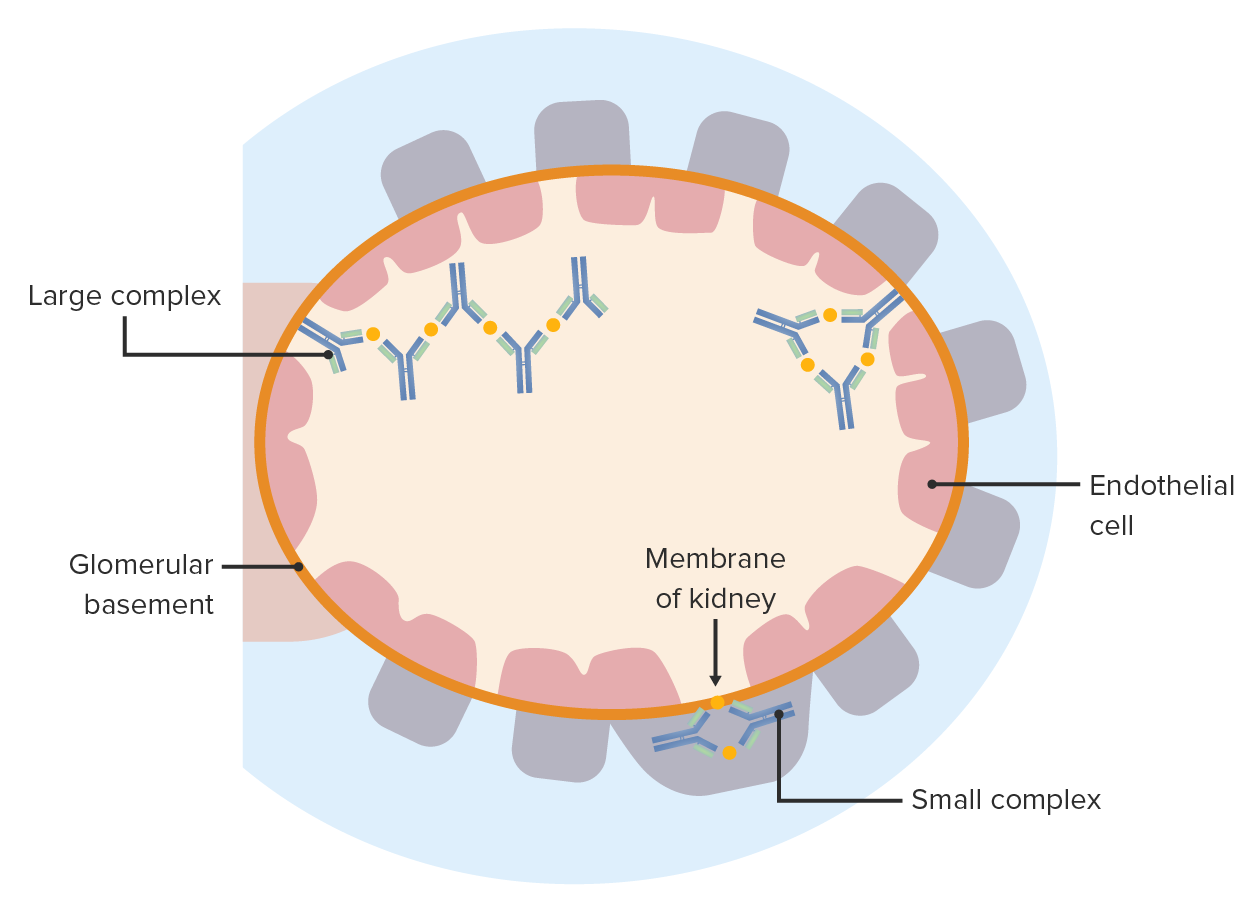
00:01 What does this look like and where does it go? So, think about this, and again it's predictable, I have antibody with antigen in a complex in a vessel wall. 00:14 That Fc, the constant region of the antibody has undergone its conformational change. 00:19 So we'll recruit Fc receptor varying cells and complement it will be activated, which will be causing local damage. 00:26 Between the inflammatory cells that are recruited and the local damage, we are going to get endothelial cell damage, vessel wall damage, so we're going to get thrombosis. 00:36 And if there's thrombosis in a vessel, all the tissue downstream, not going to have a blood supply. 00:41 So you will see infarction. 00:44 With time as we organize the damage that's happened with these, you will get vascular fibrosis, the vessels will become scarred and we're just seeing kind of a low power of a cutaneous vasculitis in the middle panel, and what that looks like up close and personal in the middle panel. 01:04 The characteristic sites for immune complexes will typically go are the kidney so you get glomerulonephritis. 01:11 You can get arthritis because of sluggish flow through the synovial tissue and you can get a pneumonitis again, the capillary beds and the lungs are a good place to get immune complex deposition. 01:23 So those are kind of the characteristic places in addition to the skin for immune complex vasculitis to occur. 01:32 There are two kind of buzz words or buzz word phrases that we use to describe the characteristic changes in immune complex vasculitis. 01:41 On the left hand side, it's fibrinoid necrosis, and it's pointing to kind of a glassy pink hylan-looking thing in the middle of the media of that small bore vessel. 01:53 That is because we've damaged endothelium and we've got leakiness of proteins including fibrinogen and other proteins into the vessel wall which will give it that kind of hyalinized pink look. 02:05 On the right hand side is the other buzzword necrotizing vasculitis. 02:09 So we have inflammatory cells that are marching right through neutrophils and macrophages, marching right through the vessel wall and destroying everything in their path, the smooth muscle cells. 02:19 So fibrinoid necrosis, necrotizing vasculitis are the typical things that pathologists use to describe immune complex mediated damage in the vessel wall. 02:32 So, let's look at a specific example. 02:35 This is post-streptococcal glomerulonephritis. 02:40 Post-streptococcal. 02:41 So, you remember that antibodies generated against the strep pharyngitis microorganism can form cross reactive antibodies that recognize heart. 02:53 Well, if you also have immune complexes that formed because of a poorly treated strep throat, then those immune complexes can be deposited in vascular beds. 03:06 And this particular vascular bed is one of the more common ones, it's the kidney glomerulus, again, because of a fenestrated endothelium and access to the underlying charged basement membrane. 03:18 And what we see is that the glomerulus is jam packed with cells, there are many, many more nuclei than should normally be in that glomerulus. 03:27 That's because we've got immune complexes that have deposited and that are expressing Fc that's rearrange, that can bind Fc receptors on neutrophils and monocyte macrophages that are being recruited into this glomerulus. 03:44 So the hypercellular glomerulus is not because we have more mesangial cells or anything else, it's because we've recruited inflammatory cells. 03:52 And as we damage them, then we're gonna get fibrin thrombi, we're going to clog off the entire glomerulus. 03:59 As a result, these poor patients are going to lose filtration capacity will have acute renal failure. 04:07 On immunohistochemistry, there's a characteristic look and again, this is for those of you who will be doing board exams to distinguish between Goodpasture's disease, which is a good example of Type 2 hypersensitivity and Post-streptococcal glomerulonephritis, which is Type 3. 04:23 Remember, in Goodpasture's, we have antibodies, acquired antibodies that recognize a component of the basement membrane in the glomerulus. 04:32 And so we're getting a diffuse linear staining of all of the capillaries. 04:38 That's Goodpasture disease on the left hand side. 04:40 On the right hand side, we're getting immune complex deposition. 04:43 So little balls of immune complex antigen antibody complex are binding in the basement membrane, so it's a granular deposition. 04:52 And if you see these two on your board exam, you'll be able to recognize one is type 2, Goodpasture's is type 3 Post-streptococcal. 05:00 A classic immune complex mediated disease is systemic lupus erythematosus. 05:05 So what is SLE? What is lupus? And basically it is a polyclonal B cell stimulation. 05:12 Somehow we've had lost peripheral tolerance, and we're turning on a whole variety of B cells that are making a whole variety of antibodies. 05:21 But there are some characteristic antibodies that drive that disease. 05:25 So there's multiple auto antibodies include antibodies that are against nuclear material, anti-double stranded DNA, all of us have antibodies against single stranded DNA, but not against double stranded or against ribonucleoprotein or RNP or against other components of the nucleus. 05:45 Characteristically, that anti-nuclear anti-double stranded DNA is a major drive. 05:52 And if there's damage anywhere, so you get sun exposure and you get UV damage, we have cells that are dying, releasing their double-stranded DNA, circulating autoantibodies against a double-stranded DNA bind to all of that release DNA and now we have immune complex deposition in that sun exposed vascular bed. 06:14 That's how you get type III hypersensitivity lesions in a patient who has lupus. 06:21 In addition to the anti-double stranded DNA, which is classic in characteristic. 06:26 Patients with lupus also have antibodies against their red cells. 06:29 So they can have an autoimmune hemolytic anaemia. 06:32 They have anti-platelet antibodies in many, many cases, so that they can get abnormal thrombosis and/or thrombocytopenia. 06:41 They also have antibodies that will bind to a variety of phospholipid surfaces, which will cause endothelial cell activation, which will cause thrombosis. 06:49 So these poor patients because of the various natures of their antibodies can have a variety of diseases, but it's the immune complex deposition that's going to cause the most profound pathology. 07:03 What's happening? So in patients who have lupus, they are said to have protein manifestations. 07:08 Well, yeah, that's happening because wherever the immune complexes deposit is where we're going to get complement activation, recruitment of FC receptor bearing cells. 07:18 And it could be for whatever reason that they deposit in the brain, in which case you will have neuro psychological problems. 07:26 It could be that they deposit in the kidney, and you will have the Maryland nephritis. 07:31 It can be the deposit in the lungs or the skin. 07:34 It depends on where they deposit. 07:36 So the manifestations of lupus can be multiple protein, because of the vagaries of where the immune complexes deposit. 07:46 Wherever they do deposit, there's going to be thrombosis and tissue ischemia. 07:50 And again, this final bullet point that I've been hammering to death, wherever the immune complexes deposit, that will determine the presentation. 07:59 So you can have patients with lupus who just have joint disease, or just have cutaneous disease, or just have renal disease, or they may have a combination of all of the above or something completely different, again depending on the site of deposition. 08:14 One final entity and you may never see one of these patients in your entire life. 08:19 However, this disease, the antineutrophil cytoplasmic antibody (ANCA) associated vasculitis is something that they like to put on the boards with some regularity just because it's a rare disease and they don't, they don't expect you to know it. 08:36 I'm going to mention this, you'll review it when you take your boards. 08:39 And you may never see a patient who has this, it's relatively rare. 08:44 So we are starting with inflammatory trigger, and/or neutrophils that are expressing a variety of things, including Fc receptors, as you see on the right hand side, but also the neutrophils have intracytoplasmic granules that contain myeloperoxidase and proteinase 3, that's a PR3 is. 09:07 And if they get activated, they released that material, the cytoplasmic material to their cell surface, they can also dump it onto associated tissues. 09:17 So the little red balls or myeloperoxidase, the little yellow balls or proteinase 3, they are derived from cytoplasmic granules, and that's why this is called ANCA (antineutrophil cytoplasmic antibody) -associated vasculitis. 09:32 Activated neutrophils, put these various molecules on their surface or on to adjacent tissues. 09:39 And in a patient, if they have ANCA antibodies, antineutrophil cytoplasmic antibodies, they can then bind either to the PR3 or myeloperoxidase that are present on the neutrophil surface. 09:52 Or they can bind to the underlying tissues that have also deposited these various granule contents. 10:00 Regardless, having those antibodies that are directed against PR3 or myeloperoxidase will then lead to the recruitment and activation, complement activation, etc. 10:11 But it tends to be relatively antibody poor. 10:14 It doesn't take very many antibodies to drive this process and that's why it's called pauci-immune vasculitis. 10:23 If we crosslink the molecules on the surface of the neutrophil, we can get auto activation, but we're also going to get neutrophils that are recruited and damaging wherever the antibodies have deposited in the tissues. 10:36 So this is ANCA-associated vasculitis. 10:38 You've seen it now. 10:40 And hopefully you'll never have a patient who has it. 10:44 And with that, we've completed type III hypersensitivity responses.
About the Lecture
The lecture Type III Hypersensitivity: Manifestations in Different Diseases by Richard Mitchell, MD, PhD is from the course Immune-mediated Diseases.
Included Quiz Questions
Which of the following is not a typical anatomic site of immune complex deposition in a type III hypersensitivity reaction?
- Liver
- Kidneys
- Lungs
- Joints
- Skin
Which of the following is not a typical histologic manifestation of a type III hypersensitivity reaction?
- T-helper cell infiltration
- Thrombosis
- Infarction
- Fibrosis
- Endothelial cell damage
Which of the following autoantibodies are not characteristic of systemic lupus erythematosus?
- Anti-single-stranded DNA antibodies
- Anti-double-stranded DNA antibodies
- Anti-ribonucleoprotein antibodies
- Anti-erythrocyte antibodies
- Anti-platelet antibodies
Which of the following is not a typical anatomic site of involvement in systemic lupus erythematosus?
- Ears
- Skin
- Kidneys
- Lungs
- Brain
Antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis is characterized by autoantibodies against which of the following?
- Myeloperoxidase
- Ribosomes
- DNA
- RNA
- Phospholipids
ANCA-associated vasculitis typically involves autoantibodies against which of the following?
- Proteinase 3
- Tissue necrosis factor
- Vascular endothelium
- Phospholipase C
- Endorphins
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |