Playlist
Show Playlist
Hide Playlist
Traumatic Brain Injury (TBI)
-
Slides TraumaticBrainInjury Surgery.pdf
-
Download Lecture Overview
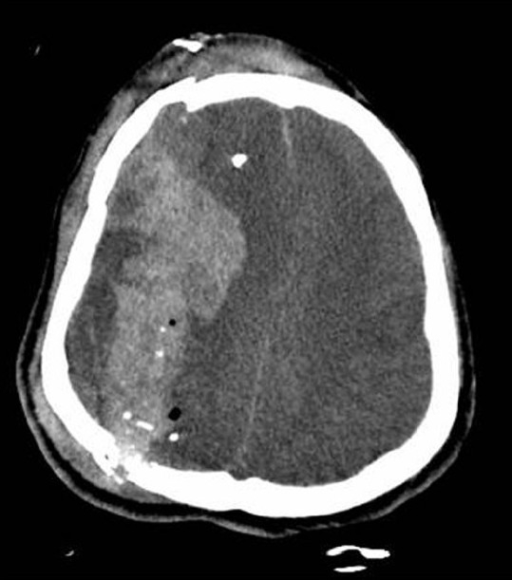
00:01 Welcome back. 00:02 Thanks for joining me on this discussion of traumatic brain injury under the section of trauma. 00:08 Let’s start with a clinical scenario. 00:10 Take a look at this picture. 00:12 The driver, a 25-year-old man, sustained a high-speed roll-over motor vehicle accident who now arrives in your trauma bay, is not responsive. 00:22 Do you know what to do next? I’ll give you a second to think about it. 00:28 Remember, back to our previous initial assessment and management discussion, always begin with the ABCs. 00:35 Now that you’ve ascertained an intact airway, a breathing patient and a patient who has intact circulation, we move to the D of disability where we ascertain a Glasgow Coma Scale. 00:46 The Glasgow Coma Scale has three components. 00:49 It’s a global assessment of the patient’s neurological status. 00:53 Take a look at this table. 00:55 You’ll notice that there are columns for eye, verbal, and motor examinations. 01:00 The coordinated effort of the patient who is able to open their eyes, speak to you, and move their extremities is one that is an oriented patient. 01:10 Glasgow Coma Scales are scored from a minimum of 1 to a maximum of 6 points. 01:16 We assign the best exam finding. 01:19 Remember, the best exam finding. 01:22 When presented on an examination of what is the GCS score, always look for the best exam finding. 01:28 The eye exam is listed from 1 to 4. 01:31 The verbal examination is listed from 1 to 5. 01:34 And the motor exam is listed from 1 to 6. 01:38 There is no precedence or importance of one exam over another, although it appears that motor is heavily weighted. 01:46 Important to remember that you just have to spend a few minutes looking at the slide and memorizing. 01:51 Unfortunately, I don’t have any simple ways of memorizing the GCS EVM scoring. 02:00 The patient does not open his eyes, respond verbally or move. 02:06 My question to you is, “What is this patient’s Glasgow Coma Scale?” Again, the patient doesn’t open eyes, has absolutely no verbal and is not moving at all. 02:19 This is a common trick question. 02:21 The minimum GCS is 3 because the lowest score is 1 on a 1 to 6 scale. 02:27 So, the minimum score GCS is 3 in this completely comatose patient. 02:33 As a side note, the placement of the ET tube, if that’s the only limiting factor for patient’s ability to speak, we assign a T for endotracheal tube at the end of the GCS score. 02:44 Let’s transition to a discussion of traumatic brain injury but first let’s discuss the anatomy. 02:52 On this image, you see the first layer as the skull and right underneath it is the superior sagittal sinus containing venous drainage blood. 02:59 The yellow outline suggests a subarachnoid space between 2 arachnoid and pia maters. 03:07 I’d like to start the discussion of specific traumatic brain injuries with a discussion of subdural hematomas. 03:13 Classically, subdural hematomas or any intracranial bleeding is associated with high impact to the skull. 03:20 They are traditionally associated with tearing of bridging veins between the brain surface to the dura sinus. 03:27 As you can see in this image, bleeding in the subdural space results in a semilunar or often called moon-shaped crescent hematoma. 03:37 The reason is because the blood separates the arachnoid away from the dura. 03:41 It is not, however, bound by the sagittal sinuses. 03:46 There may or may not be medial deviation or elevation of the intracranial pressure. 03:53 Remember, any space-occupying lesion in the brain may lead to elevations of intracranial pressure. 04:00 We will come back to this concept. 04:02 How do we manage subdurals? Based on the clinical status and the severity of the subdural, management is guided by midline shift, intracranial hypertension, clinical picture such as a comatose or worsening GCS patient or particularly large subdural hematomas as defined by bleeding hematomas greater than one centimeter in thickness. 04:26 If these findings are present, consider calling a neurosurgical colleague for surgical decompression of the hematoma. 04:33 Of course if the patient doesn’t have any of these significant findings, then our job as trauma surgeons is to prevent secondary injury. 04:41 What does it mean to prevent secondary injury? With any trauma, just like when you bang your knee, some swelling is bound to happen. 04:48 We want to decrease swelling and also prevent ischemia or low perfusion of your brain. 04:54 Let’s move on and discuss epidural hematomas. 04:59 Once again, epidural hematomas are associated with high impact to the skull. 05:04 Unlike subdurals which are associated with bridging veins, the epidurals are associated with middle meningeal artery. 05:13 In a clinical scenario, this often describe classic lucid interval where the patient immediately following the trauma may actually be lucid and after about 30 minutes to an hour has a second comatose episode. 05:26 I caution the viewer though that the classic lucid interval is much more discussed and seen in real life. 05:33 And seen on this CAT scan, you notice that the lenticular shape or biconvex shape which is limited by the suture line. 05:41 How do we manage an epidural hematoma? Most epidural hematomas, unlike subdural hematomas, require a surgical decompression. 05:53 If a clinical scenario is presented to you where a patient sustains high velocity or a high-impact brain trauma and they demonstrate a lucid interval, I encourage you to hold epidural hematomas high on your differential list and a list of surgical decompression. 06:11 Once again, like any traumatic brain injury, we want to try to prevent secondary injury. 06:17 Now, let's have a short view on tumor types bleeding in the brain. 06:22 First, there's a traumatic subarachnoid hemorrhage. 06:25 This is type, there is blood in the subarachoid space, so between the pia and the arachnoid mater. 06:31 A head trauma is the most common cause. 06:32 Possible symptoms are headache, nuchal rigidity, photophobia, focal neurologic deficits, and loss of consciousness. 06:39 For a diagnosis, you can use a head CT without contrast. 06:43 To treat such a hemorrhage, you can do a surgical clipping or an intravascular coiling. 06:48 On this slide, you can see two types of intracranial hemorrhage. 06:51 The CT scan on the left depects a case of intracerebral hemorrhage, whereas the slide on the right, shows a case of intraventricular hemorrhage. 06:59 Treatment is mainly supportive. 07:00 In severe cases, an external ventricular drain maybe required to evacuate the blood or even craniotomy. 07:06 Let’s discuss skull fractures. 07:11 Once again, skull fractures are associated with high-impact mechanisms such as an assault with a weapon. 07:18 It’s a classic scenario to have a patient undergo assault with a bat or a metal instrument direct blow to the head. 07:29 Remember that skull fractures are often associated with cervical spine injuries. 07:34 This likely has to be dissociation due to the high impact injury. 07:39 Some very important classic signs include the raccoon’s eyes which is a bruising around the eyes. 07:46 Remember, raccoon eyes and battle’s signs are signs of a basilar skull fracture. 07:52 Battle’s sign is bruising around the mastoid process. 07:55 Just to review, raccoon’s eyes is bleeding or bruising around the eyes and battle’s sign is bruising of the mastoid process just behind the ear. 08:04 And depending on the severity, the patient can have a CSF, cerebrospinal fluid leak. 08:11 Most skull fractures are largely managed non-operatively unless couple of caveat exists. 08:17 One, significant depression of the skull. 08:20 This may require elevation of the skull. 08:22 And most importantly, an open skull fracture uniformly requires exploration and elevation. 08:29 Earlier, I mentioned that one of the clinical signs to evaluate the patient for is intracranial pressure monitoring and to try to prevent intracranial hypertension for high ICPs. 08:42 A very useful and practical equation to remember is the cerebral perfusion pressure. 08:48 The cerebral perfusion pressure is a difference between the mean arterial pressure. 08:53 That’s the systemic circulation minus the intracranial pressure. 08:57 Again, the cerebral perfusion pressure is a difference between your MAPs and your ICPs. 09:04 In severe TBI patients where their GCS is less than 9 were considered comatose. 09:10 Intracranial pressure monitoring may help with diagnosis of further deterioration. 09:16 Remember, comatose patients will not likely participate in your neurological examination. 09:21 Patients in whom we cannot follow the examination, for example, a comatose patient or one who is on severe sedation or anesthesia may also require intracranial monitoring.
About the Lecture
The lecture Traumatic Brain Injury (TBI) by Kevin Pei, MD is from the course Surgery: Trauma. It contains the following chapters:
- Traumatic Brain Injury
- Subdural Hematoma
- Epidural Hematoma
- Skull Fractures
Included Quiz Questions
Which of the following motor responses is considered a 3 on the GCS?
- Abnormal flexion to painful stimuli (decorticate response)
- Localizes pain
- Withdraws from pain
- Extensor posturing (decerebrate response)
- Obeys commands
A trauma patient in the emergency department opens his eyes to pain, speaks inappropriate words, and has no motor responses. What is his GCS?
- 6
- 7
- 5
- 3
- 4
What is the minimum GCS score that a patient can have?
- 3
- 4
- 2
- 1
- 0
Which of the following is NOT seen in a case of subdural hematoma?
- A convex pattern of blood seen on a non-contrast CT scan of the head
- Crescent-shaped hematoma
- High impact to the skull
- A tear of bridging veins
- Elevated intracranial pressure
A mastoid fracture has which of the following signs?
- Battle’s sign
- Lemon sign
- Halo sign
- Guerin sign
- Dural tail sign
Customer reviews
4,0 of 5 stars
| 5 Stars |
|
0 |
| 4 Stars |
|
1 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
I would have love to learn further about the criteria for performing a head CT eg Canadian Head CT Criteria or others that may be appropriate to guide decision for head CT