Playlist
Show Playlist
Hide Playlist
Transmissible Spongiform Encephalopathy (TSEs) of Humans
-
Slides 15 Prions MicrobiologyAdvanced.pdf
-
Download Lecture Overview
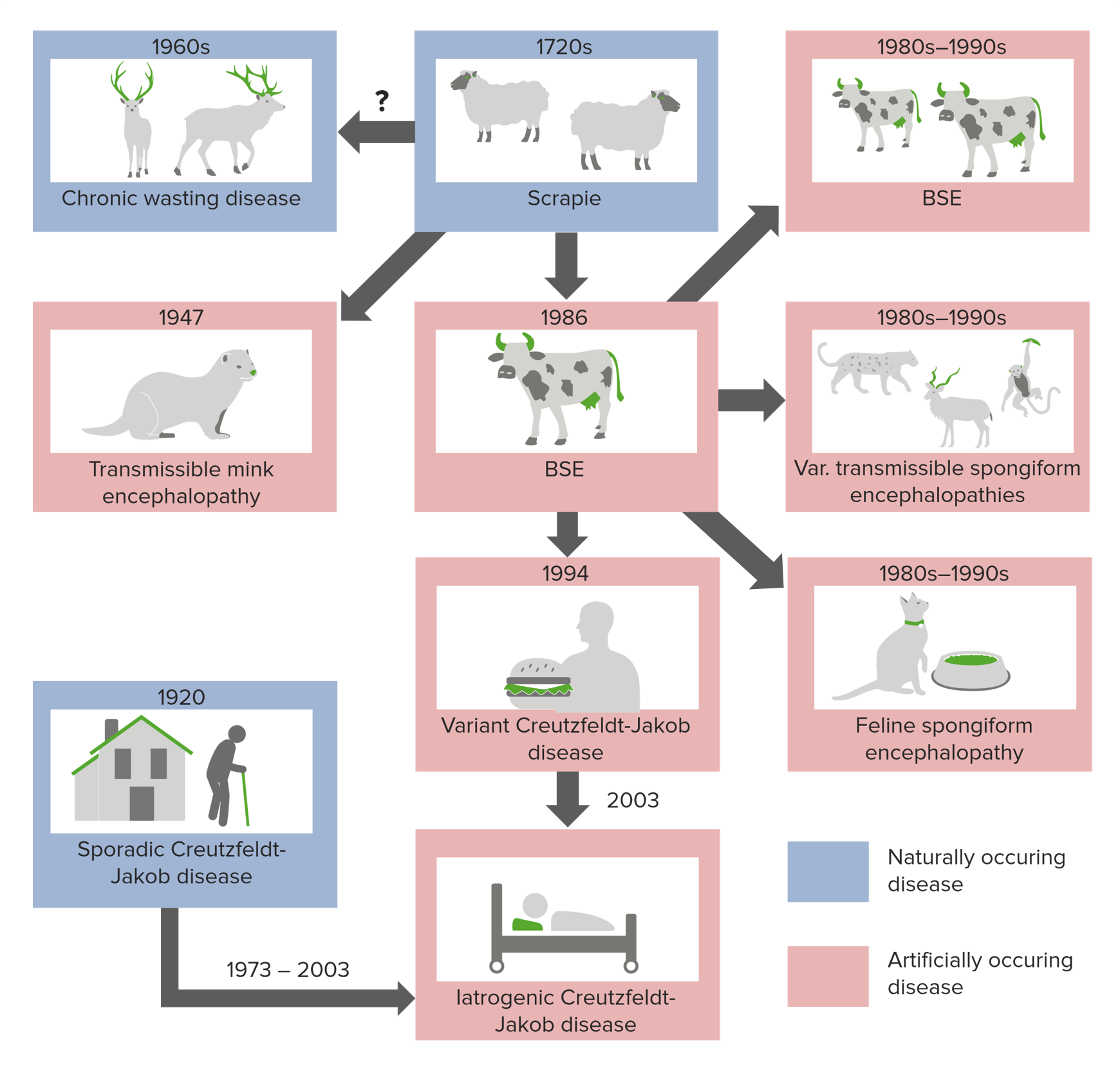
00:01 Hello and welcome to Prions. We are going to be diving a little deeper into this topic. 00:07 And after you've listened to this video, I hope you'll understand two assays for detecting prions. You'll know why Mad Cow Disease and Chronic Wasting Disease may be a threat to the food chain for humans. You’ll comprehend the species barrier to prion infections, and you'll be able to trace the possible origin of prion diseases. 00:36 Prions cause diseases that we call transmissible spongiform encephalopathies, which I'll now refer to as TSEs, and here are some of the human TSEs: Creutzfeldt-Jacob disease (CJD), Fatal Familial Insomnia (FFI), Gerstmann Straussler syndrome (GSS), Kuru, and variant CJD (vCJD). 01:02 These are human TSEs, they are also TSEs of animals. 01:07 TSEs or prion diseases are protein misfolding diseases. They involve the misfolding of a normal cell protein. So let's explore exactly what that means. The normal cell protein involved in TSEs is called the prion protein PrP with the superscript C, which means it's the cellular version. And shown here on the left is the structure of the PrPc protein. It's the normal version. You can see lots of swirls in this protein, those are alpha helices. This protein has high alpha helical content and very low beta sheet content. The pathogenic version of PRP, which is associated with TSEs as shown on the right, is called PrPsc, SC stands for scrapie, because that was the first TSE discovered, and the one where the role of the prion protein was figured it out. This protein is abnormally folded. Now we don't have the structure of this solved, but we think it has a lot of beta sheets and you can see those four long sheets going up and down in green and in yellow, those are beta sheets. So this is a conformational alteration of the normal protein. We have the normal protein and a conformationally altered PrPsc, and that PrPsc is pathogenic, when you accumulate that in your brain, you develop a TSE. It’s a very unusual disease. So pathogenic prion, PrPsc, is a conformational isoform of a normal host protein PrPc. Prion, the word prion stands for proteinaceous infectious particle, and you might hear me say “prion” or “prion”. I really don't know what's right, when you have a word invented by people, what's the difference on how you pronounce it? As long as you say “prion” or “prion”. 03:14 Prions are normal proteins on the outer surface of neurons. They are found in a few other places, but mainly on the outer surface of neurons. As in this slide, you can see at the bottom, a lovely neuron with the cell body, and then the axons and dendrites, and there on the surface is the normal cell, prion protein PrPc. So it has functions on the outside of a neuron, it is GPI linked. So it is not a transmembrane protein, but it is linked by a chemical linkage. When you acquire the pathogenic form of prion, PrPsc, let's say you ingest some meat that has bovine prions in it, bovine pathogenic prions. You'll eat that beef, it will go into your intestines, and shown here on this side, is a layer of mucosal cells, and on one of those mucosal cells, there are some normal PrPc, and you should be able to tell me which one it is, because you know it doesn't have a lot of beta sheets, alright. And then coming in just to the left of it, is a little bit of the abnormal protein PrPsc. It has beta sheets. You've just eaten this, so that's coming into your intestines. What we think happens, it comes in and causes the normal protein to convert to the abnormal form. It causes it to convert. This is really incredible. 04:39 The abnormal protein finds a normal version of the protein and it makes it fold, like it is folded, from PrPc into PrPsc. So that’s shown here, the pathogenic protein is next to the normal protein, and then the normal protein is now converted into a pathogenic one. It now has a lot of beta sheet structure. You can see those two proteins are now PrPsc. 05:02 And the idea is, this happens in the intestine, you then ingest, you then take up those proteins and they travel via various pathways, to neurons in your central nervous system and there they convert additional PrPc to PrPsc and when you have enough or more PrPsc, then you start to develop the symptoms of a TSE. And eventually you die, you will always die when you start to develop a TSE. We don’t yet have any way to stop it. Fortunately these are very rare. 05:37 So there are three ways that you can acquire or develop a TSE, also known as a spongiform encephalopathy, because of the spongy appearance of the brain when it accumulates a lot of these PrPsc proteins. So again on the left we have our normal protein PrPc, on the right, the abnormal PrPsc. There are three ways to get from normal to abnormal. You can acquire some PrPsc, you can eat it in beef or it can be introduced into you by a corneal transplant, if you get a cornea from someone who has PrPsc, maybe they don't know about it at the time when they die and donate the cornea, you can acquire PrPsc. You can get it from human hormones or other blood products. There are ways to get it. So this is the infectious form. You get that PrPsc and it converts your PrPc into more PrPsc and you develop the disease. 06:36 A second way you can acquire this is by genetics. The luck of the draw. You get a mutation from your parents in the prpn gene, the gene encoding PrPc. Single amino acid changes enough to, at some point in your life make PrPc, start to misfold, and once there's one PrPsc somewhere in you, it then makes all your others misfold. So it’s a genetic disease. So far we’ve got an infectious disease, we have a genetic disease, and finally there is what's called a spontaneous disease, where you don't eat any contaminated beef, you don't get a corneal transplant, you don't have a genetic mutation that predisposes you to misfolding, but for some reason at some point, one day some of your PrPc misfolds and becomes PrPsc and you go on to develop a TSE. Alright, three ways that you can get this, in the end they are all fatal. 07:32 They all can be transmitted. 07:35 So here is a graph showing you the number of cases of Creutzfeldt-Jakob disease deaths in the United States from like 1979 to 2011. You have deaths in the bars and the death rate is in green. So what you can see is that the rate is low, it's 150 to 200, and now maybe 400 per million population. It's rare but not unheard of, people die of this regularly. 08:05 And the incidence, this is Creutzfeldt-Jakob, a certain kind of human TSE, incidence is slowly rising with time. We don’t know why this is the case, but it's puzzling and the more people with Creutzfeldt-Jakob, the more people are likely to get it, because they can transmit it to them. So this is a combination of mostly sporadic or spontaneous TSEs and genetic. Mostly spontaneous, very, very little from eating contaminated material. 08:36 Nevertheless it's a substantial burden and it causes a 100% fatality, you never can recover from this. Hopefully one day we’ll have drugs or some way of reversing it, but we don't at the moment.
About the Lecture
The lecture Transmissible Spongiform Encephalopathy (TSEs) of Humans by Vincent Racaniello, PhD is from the course Prions.
Included Quiz Questions
Transmissible spongiform encephalopathies (TSEs) primarily affect which of the following processes?
- Protein folding
- Carbohydrate breakdown
- Gluconeogenesis
- Adenine–adenine bonding
- NAD conversion
Which of the following is NOT a human TSE?
- Bovine spongiform encephalopathy
- Creutzfeldt-Jakob disease (CJD)
- Fatal familial insomnia (FFI)
- Gerstmann-Sträussler-Scheinker disease (GSS)
- Kuru
Absence of which linkage will result in detachment of the normal prion protein PrPC from the neuronal surface?
- Glycosylphosphatidylinositol (GPI) linkage
- Glycosidic linkage
- Hydrogen linkage
- 5' to 3' phosphate linkage
- Peptide linkage
How is protein misfolding in prion diseases initiated?
- Misfolding occurs by interaction with abnormal proteins.
- Misfolding occurs by enzymatic conversion of alpha chain to zeta chain.
- Misfolding occurs by the accumulation of apoferritin.
- Misfolding occurs by lysosomal degradation.
- Misfolding occurs by cytochrome C activity.
Which of the following is NOT a mode of human-to-human transmission for prion diseases?
- By being scratched by a person who is infected with the disease
- By receiving a blood transfusion
- By receiving a corneal transplant
- Spontaneously through a novel genetic mutation that is passed to offspring
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
es un excelente vídeo, y esta muy bien explicado; justo el tema de mi próxima evaluación