Playlist
Show Playlist
Hide Playlist
Vaccination: Subunit Vaccines, Toxoid Vaccines, Conjugate Vaccines
-
Slides Vaccine Immunology.pdf
-
Reference List Immune System.pdf
-
Download Lecture Overview
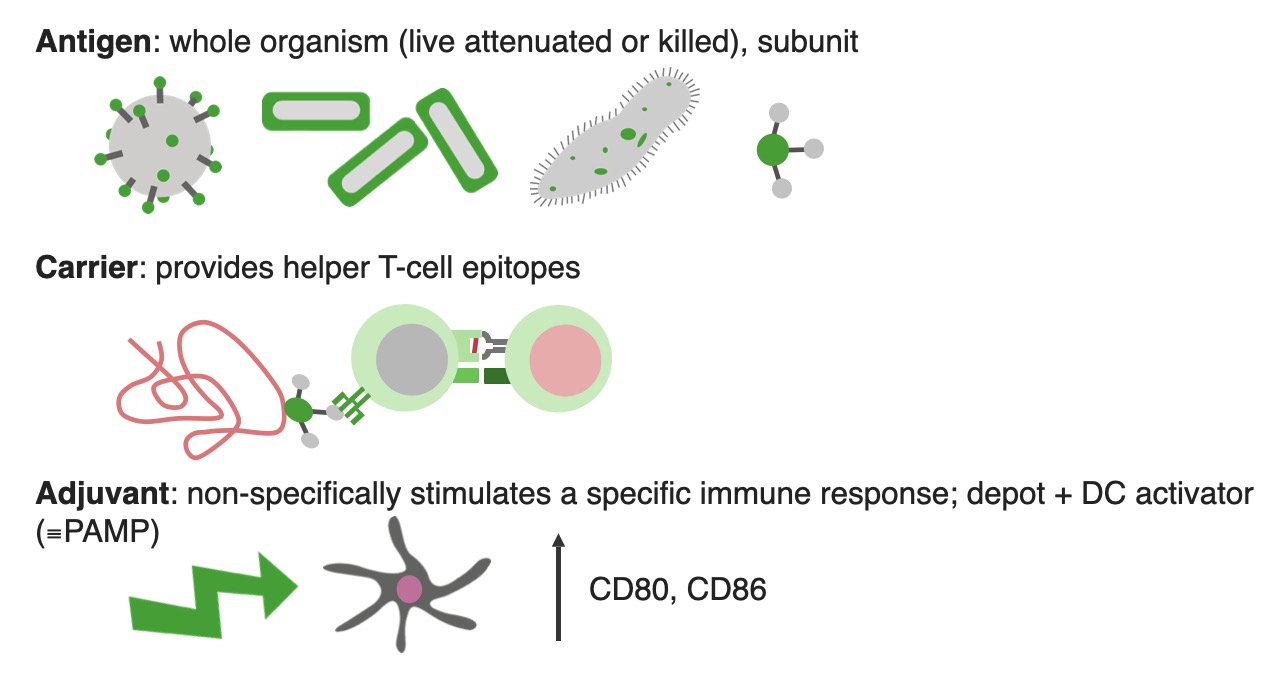
00:00 Regarding subunit vaccines, first of all looking at some diseases that are caused by viruses that we can protect using subunit vaccines - the Hepatitis B virus, there is a vaccine based upon recombinant Hepatitis B surface antigen. This is produced in Saccharomyces. 00:19 The human papillomavirus - there’s a vaccine based upon recombinant L1 major capsid proteins that self assemble into virus-like particles. 00:31 Regarding protection against bacterial diseases using subunit vaccines, there’s the DTaP, which is the acellular pertussis vaccine which is inactivated pertussis toxin and it has one or more other bacterial components. 00:50 For example, filamentous hemagglutinin or pertactin which is an outer membrane protein and fimbriae, plus bacterial toxoid and tetanus toxoid. 01:03 And then we have the meningococcal serogroup B vaccines, based upon recombinant Neisseria meningitides group B proteins. Here we have an example of a polyvalent virus like particle based vaccine, the Gardasil-9 vaccine. This is a human papillomavirus 9-valent vaccine. What that means-- it has nine different antigens in it. It uses virus-like particles derived from the major capsid, the L1 proteins of nine different human papillomavirus types. 01:40 These are types 6, 11, 16, 18, 31, 33, 45, 52 and 58. 01:49 And these self-assemble into these recombinant virus-like particles that are produced in Saccharomyces cerevisiae. 01:57 They are released from the yeast cells by cell disruption and purified using chemical and physical methods. 02:08 They are then adsorbed onto preformed aluminium-containing adjuvant called amorphous aluminium hydroxyphosphate sulfate. 02:21 They are given to females and to boys and men for the prevention of cancer and genital warts. 02:29 So looking at toxoid vaccines, these are chemically inactivated bacterial exotoxins. 02:37 They protect from disease but not from infection. 02:41 Examples include tetanus toxoid and diphtheria toxoid. 02:45 Here we can see bacteria releasing a bacterial toxin. 02:52 This is pathogenic, it can harm our cells. 02:56 It can be recognized by the antigen receptor on B-lymphocytes and stimulate a memory B-cell response. 03:04 However by chemically inactivating the toxin in the laboratory, one can produce a harmless version of it that has lost the pathogenicity. 03:16 And we refer to this form of the inactivated toxin as a toxoid. 03:22 Although it has lost the ability to cause pathology, it maintains enough of its structure so that it can still be recognized by the B-cell receptor on the surface of B-lymphocytes, and therefore still generate memory cells. 03:39 Purified bacterial capsular polysaccharides only elicit IgM antibodies due to a lack of helper T-cell epitopes. 03:50 This can be overcome by employing conjugate vaccines. 03:57 Conjugate vaccines comprise polysaccharides coupled to a protein. 04:03 Tetatus or diphtheria toxoid is often the protein of choice. 04:10 Using this strategy, it converts a T-independent response to a T-dependant response. 04:18 So here we can see recognition of the polysaccharide by the B-cell receptor on the surface of a B-lymphocyte. 04:26 But on its own, this would not recruit helper T-cells and there would be no class switching from IgM to higher affinity IgG antibodies. 04:39 However by conjugating the polysaccharide onto a protein, the complex of the polysaccharide plus protein will be taken up by the B-cell. 04:51 The protein component will be processed into peptides and those peptides will be shown to the T-cell receptor on the surface of helper T-cells. 05:02 Following interaction of the B-cell with the T-cell, the T-cell will release cytokines and the molecules CD40 on the B-cell will interact with the CD40L molecules on the surface of the helper T-cell. 05:19 And together, those interactions will cause class switching from IgM to higher affinity IgG antibody. 05:29 So this class switching produces high affinity IgG and also IgA antibody. 05:38 Examples of conjugate vaccines include the Hib vaccine, Haemophilus influenza type b capsular polysaccharide that’s conjugated to tetanus toxoid. 05:49 The meningococcal conjugate vaccine where serogroups A, C, W and Y capsular polysaccharides are conjugated to CRM197, which is a non-toxic variant of diphtheria toxin. 06:04 And the pneumococcal conjugate vaccine, PCV13, where capsular polysaccharide antigens of 13 different Streptococci pneumonia serotypes are conjugated to CRM197.
About the Lecture
The lecture Vaccination: Subunit Vaccines, Toxoid Vaccines, Conjugate Vaccines by Peter Delves, PhD is from the course Vaccine Immunology. It contains the following chapters:
- A Closer Look at Subunit Vaccines
- A Closer Look at Toxoid Vaccines
- A Closer Look at Conjugate Vaccines
Included Quiz Questions
The DTaP subunit vaccine includes?
- Acellular pertussis, diphtheria toxoid, tetanus toxoid
- Whole-cell, inactivated pertussis, diphtheria toxoid, tetanus toxoid
- Whole-cell, inactivated pertussis, attenuated diphtheria vaccine, tetanus toxoid
- Whole-cell, inactivated pertussis, attenuated diphtheria vaccine, attenuated tetanus vaccine
- Acellular pertussis, attenuated diphtheria vaccine, attenuated tetanus vaccine
Which of the following regarding toxoid vaccines is INCORRECT?
- Toxoid is the harmful version of toxin.
- They cannot cause the disease they prevent.
- They include tetanus toxoid and diphtheria toxoid.
- They do not spread to unimmunized individuals.
- The vaccine antigens do not actively multiply.
What are conjugate vaccines?
- A combination of a weak antigen and a carrier strong antigen resulting in a strong response to the weak antigen by the host immune system
- A combination of a strong antigen and a carrier weak antigen resulting in a strong response to the strong antigen by the host immune system
- A combination of a strong antigen and a carrier weak antigen resulting in a weak response to the strong antigen by the host immune system
- A combination of a weak antigen and a carrier strong antigen resulting in a weak response to the weak antigen by the host immune system
- Vaccines that normally evade the immune system
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |