Playlist
Show Playlist
Hide Playlist
Termination of Translation
-
Slides 08 TranslationAndSummary Genetics.pdf
-
Reference List Molecular and Cell Biology.pdf
-
Download Lecture Overview
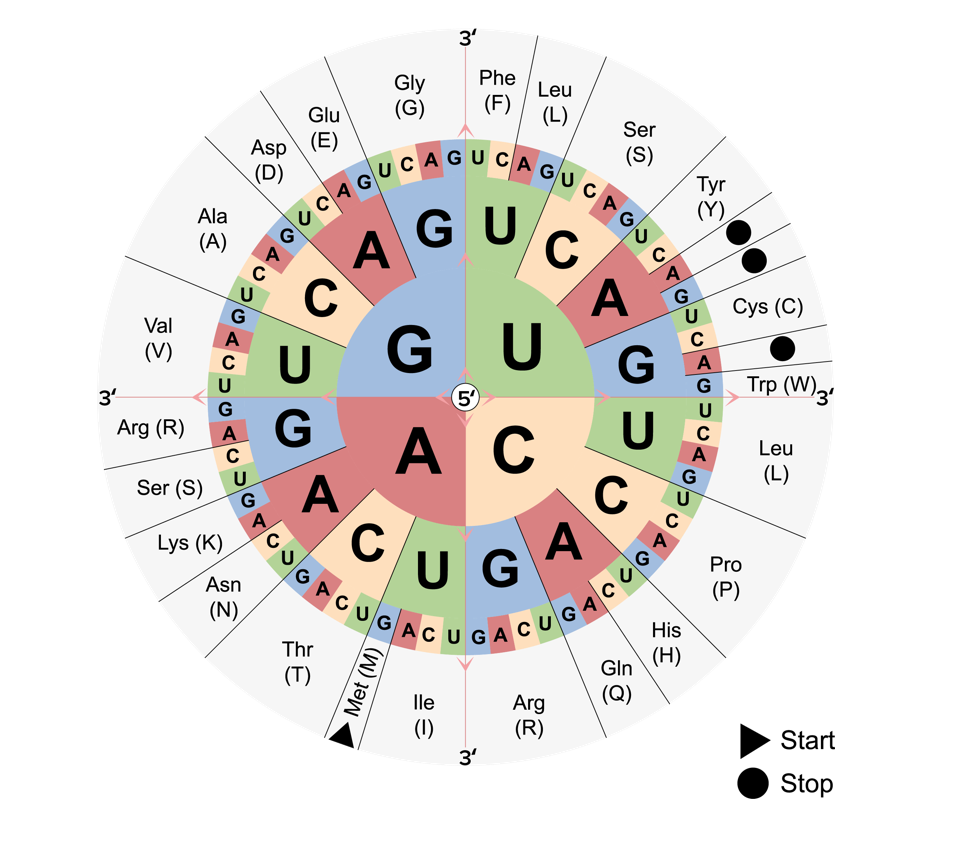
00:01 So now let’s take a look at how the whole thing ends. 00:04 We’ve gone along around the cycle, then we’ve switched from P to A and translocate it and so on and so forth. 00:10 We’re growing the polypeptide chain. 00:13 It’s getting longer and longer. 00:14 And now, we have reached the end of the coding sequence on the messenger RNA. 00:19 Now, just as there is a 5’ untranslated region, that helps the ribosome associate and helps the messenger RNA find its way, there’s also a 3’ untranslated region, so we will get to the stop codon. 00:36 There will still be a little tail of RNA. 00:38 We’re not necessarily completely sure how those come into play at this point in time, but we know they have some sort of role. 00:47 But anyway, the point here is we come to a stop codon and the stop codon does not code for a tRNA. 00:55 It codes for a release factor. 00:58 I think of release factors as if they are bombs. 01:01 Once that the release factor binds into that site, we’ll see that the whole thing falls apart. 01:07 So it’s not a tRNA, binds into the site, and it causes the whole thing to blow apart. 01:14 We release the polypeptide chain and then the large subunit dissociates from the small subunit and we end up having both large and small subunit dissociated. 01:28 So let’s say we are dealing with proteins that are going to be used inside the cell versus proteins, polypeptides, that are going to be used outside the cell. 01:43 You recall from cell structure, there was rough endoplasmic reticulum, which had ribosomes studded all over it. 01:49 And there is smooth endoplasmic reticulum, which does not. 01:53 But there are also free-floating ribosomes. 01:56 What’s the difference? So some proteins are to go and some proteins are to stay. 02:03 You’ll recall that we use the endoplasmic reticulum for proteins to go. 02:08 We package them, send them to the Golgi, who then puts them in the right transport vehicles. 02:13 That’s another lecture. 02:13 So what happens if we are to go? We need to get associated with the endoplasmic reticulum. 02:21 First of all though, will be a signal sequence on the beginning of that polypeptide chain. 02:28 All sorts of things happen to these polypeptide chains once they’ve been formed. 02:32 But the signal sequence will trigger a certain set of reactions and it involves a signal recognition particle, which is going to help dock this ribosome on the endoplasmic reticulum. 02:48 So first of all, they’re not associated and once it’s recognized that there’s this signal sequence, the particle will bind on and help the ribosome find the endoplasmic reticulum. 03:03 So the signal recognition particle binds to the signal peptide and that’s going to arrest the elongation cycle. 03:13 So the elongation stops, then we go all the way over to the endoplasmic reticulum and we’re going to dock the ribosome on the endoplasmic reticulum and associate it with a pore so that it can then restart protein synthesis and filter its polypeptide into the endoplasmic reticulum. 03:37 So it’s going to – The polypeptide enters through a pore and synthesis is going to resume so the ribosome hanging out on the outside of the endoplasmic reticulum continues to grow the polypeptide chain using that whole elongation cycle business. 03:54 And now the polypeptide is inside the endoplasmic reticulum and it’s ready to go. 03:59 It’s ready for export. 04:02 So now, let’s take a quick look at a summary of all of this gene expression
About the Lecture
The lecture Termination of Translation by Georgina Cornwall, PhD is from the course Gene Expression.
Included Quiz Questions
How do ribosomes get attached to the endoplasmic reticulum?
- They associate with the rough endoplasmic reticulum with the help of the signal-recognition particle that binds to the signal sequence present on the elongating polypeptide chain.
- They associate with the smooth endoplasmic reticulum with the help of the signal-recognition particle that binds to the signal sequence present on the elongating polypeptide chain.
- They associate with the Golgi apparatus with the help of the signal-recognition particle and are transported to the endoplasmic reticulum through vesicles.
- They associate with the rough endoplasmic reticulum with the help of the signal-recognition particle that binds to the signal sequence present on the small subunit of the ribosome.
- They associate with the rough endoplasmic reticulum through phagocytosis.
Which of the following stops the translation process?
- Stop codons will bind to the release factors, instead of tRNA, to stop the translation process.
- Stop codons will bind to elongation factors to switch to the termination phase of translation.
- Stop codons will bind to initiation factors to stop the translation process.
- Stop codons will bind to transfer factors to stop the translation process.
- Stop codons will bind to transfer factors to transfer the elongating polypeptide chain out of the E site.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |