Playlist
Show Playlist
Hide Playlist
Pneumonia: Treatment
-
Slides 01 URTIBronchitisPneumonia RespiratoryAdvanced.pdf
-
Download Lecture Overview
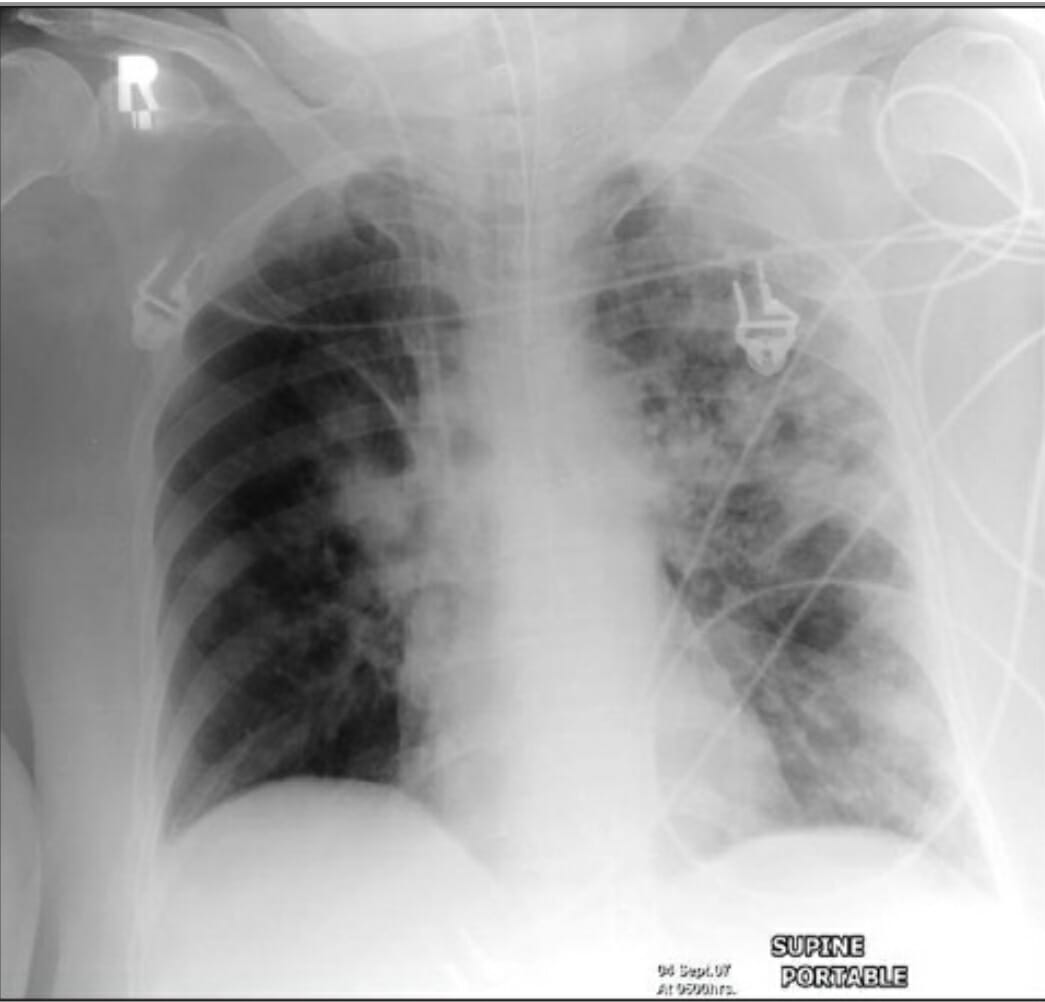
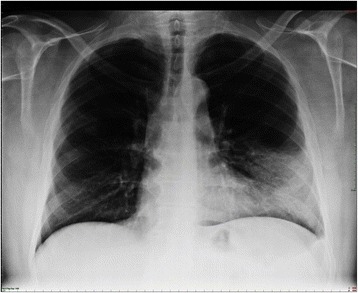
00:01 The other aspect of management is this correction of oxygenation. If you have consolidation, you are placing air in the alveoli with inflammatory fluid. And that causes hypoxia because it generates what we call a right to left shunt. What I mean by that is that blood coming up the pulmonary artery, which is deoxygenated, will reach the area with the consolidation in it, it will go through that area but because there's no air in the alveoli, that blood will not be oxygenated, and therefore, it will reach the pulmonary veins in a deoxygenated state, and you end up mixing the deoxygenated blood coming from the consolidated lung with the oxygenated blood coming from the remaining normal lung. The consequence of that is that you have a mixture of hypoxic blood and normoxic blood, and the combination of the two will dictate how hypoxic the patient will be. So in this example, for example we have a patient who has quite extensive consolidation and therefore, the deoxygenated blood shunting through that consolidated lung has a significant effect on the arterial oxygen concentration, reducing it down to about 80%. So oxygen needs to be corrected and we do that using masks and if necessary mechanical ventilation. Another important physiological upset that occurs in patients with pneumonia is due to the sepsis, due to the septic shock potentially, that can develop in a minority of patients with acute bacterial pneumonia. Before that, actually many patients because they’ve been breathing fast, unable to drink and eat, they are dehydrated. So dehydration is common, and beyond that some patients will develop septic shock with persisting hypotension even after they’ve been rehydrated. An acute kidney injury is very common in these patients as a consequence of these problems. So patients with pneumonia will require intravenous fluids to rehydrate them if they have got a low blood pressure or clinically, they are dehydrated. And if the low blood pressure persist despite rehydration, then you need to consider inotropes and admission to intensive care to treat the septic shock, and to avoid the downstream consequences of having hypotension, increasing kidney injury, etc. etc. Killing the pathogen. Well, when the patient first presents to you with pneumonia, you don't know what the bug is causing that pneumonia, so we treat the patient initially with what we call empirical antibiotics, ones which will cover the common causes of pneumonia. To do that we need to know what the common microbial causes of pneumonia are. And for the industrialized world, these are relatively similar across different countries, and they split down into about 20-25% of cases are just due to viruses, influenza being the commonest one, but occasionally you get other viruses rhinovirus, RSV, parainfluenza, etc. and adenovirus. And then, three quarters of cases are due to bacteria. Those bacteria can be divided into two categories: the pyogenic bacteria the ones that cause a higher inflammatory response, focal consolidation generally speaking. 03:15 The ones that will cause more acute and severe disease, and that's mainly Streptococcus pneumonia, which is responsible for about half of all cases of community acquired pneumonia. 03:25 But there are a couple of other bacteria that occasionally come through Haemophilus influenza, Stapylococcus aureus, being the commonest. Fortunately, gram-negative pneumonia is relatively rare. Atypical organisms cause a less aggressive disease, and those are mycoplasma and chlamydophila, and occasionally Legionella, which is an exception and that it can actually cause severe disease despite being part of the atypical category of organisms. An important point is that many patients will have a mixed infection, so they will have a viral infection that allows the bacteria in, so they have a combination of influenza A and say pneumococcal pneumonia. 04:00 And the same thing happens with mycoplasma and chlamydophila, that allows the bacteria in to cause an infection, so you get a combination of an atypical organism plus Haemophilus, Staphylococcus aerus or Steptococcus pneumonia. A very important point here is that actually, when it comes to the treatment of pneumonia we don't normally treat the viruses, the exception being influenza A which we’ll treat with a neuraminidase inhibitors because there are no good antiviral agents for most of these viruses, but we do have to treat the bacteria. 04:32 So it's a question of which antibiotic to use. 04:35 Microbiologic testing and the site of care are usually determined by the severity of the illness, with most of the testing taking place in the hospital setting. 04:43 In mild Severity, with a CURB-65 score of zero, or a PSI score of 1-2, patients are usually treated as an outpatient. 04:52 Tests are usually minimal but can include covid 19 testing or influenza testing depending on local incidence levels. 04:58 In moderate severity with CURB-65 scores of 1-2 or PSI scores of 3 to 4, patients are usually usually hospitalized on a general medicine floor. 05:07 Tests would include covid 19, rapid PCR or culture for MRSA, blood cultures (which are taken prior to administration of antibiotics), sputum gram stains or cultures, urine strep antigen, legionella testing, respiratory virus panels during flu season, and HIV screening. 05:25 Finally, in very severy cases with a CURB-65 score of three or more, and a PSI of 4-5, The reason why is that to actually get a positive result for serology, you need a matched sample, these patients are usually treated in the ICU. Testing is similar to that of moderate severity cases plus the possible addition of a bronchoscopy if the benefits outweigh the risks. 05:45 This allows for specimen collection for gram stain and cultures, fungal stains and cultures, and molecular testing. 05:52 The Empiric antibiotic guidelines is based on several factors, but for patients without comorbidities, penicillin allergy, or suspicion of MRSA pr pseudomonas infections, the guidelines are as follows. 06:04 antibiotics should be started within 4 hours of presentation or within 1 hour if the patient is in septic shock. 06:11 The regimine selected is dictated by both the pathogen and the case severity. 06:15 It is usually guided by local susceptibility patterns, drug formularies, and individual patient characteristics. 06:22 If a pathogen is subsequently identified, the susceptibility testing should be used to guide that regime adjustments. 06:28 The duration of therapy varies, with Mild CAP cases receiving 5 days or oral antibiotics as an outpatient, and moderate or severe cases receive 5-7 days of IV antibiotics in the hospital. 06:40 Clinical improvement, trending procalcitonin levels (which is a biomarker for active infection), pathogen, and presence of clinical complications all must be taken into consideration when determening the lengh of therapy. 06:52 Right, how do we monitor the progress of the disease? Well normally patient with community acquired pneumonia presenting to hospital, coming in, receiving their antibiotics, their IV rehydration, they actually get better within a couple days, from 2 to 3 days. 07:07 Normally, the pyrexia will go away within 72 hours. The pulse rate and their blood pressure will come back down to normal, and the oxygenation will improve. And, as well we can do a blood test to see whether there's an improvement in whatever parameters that have been abnormal on the initial presentation. So for example, the C-reactive protein, the inflammatory marker which is normally quite high on presentation should usually fall by 50% every 24 hours or so once the stimulation of the inflammation has gone. So once the infecting organisms have been killed by the antibiotics then the CRP will fall by 50% every day or so. So by the day 3 or 4, we expect remitting CRP level to have fallen by over 50%. And the things like the hyponatremia, the abnormal liver function test, the high white cell count, the high urea and creatinine, they should all normalize over the first couple of days of admission. If somebody is not getting better, if the blood tests are persistently abnormal, if they are still hypoxic, there is still pyrexia, you need to think about various things. One is that you need a chest X ray to look for the complications, and if there is a pleural effusion present then you need to tap that pleural effusion to make sure it’s not infected. You need to think about re-culturing to see whether the bacteria that is present might be a resistant organism and I've mentioned pseudomonas already as one of the key organisms we need to consider. And then, we just have to think about the diagnosis. Was this the right diagnosis? Did they have pneumonia or do they have one of the other conditions that I mentioned earlier? Is this a pulmonary embolus for example? Is there a pulmonary oedema complicating this? Is there a cancer that makes the consolidation difficult to treat, as there is no drainage of the area of the lung. Or is this an inflammatory lung disease which is not infective at all, a cryptogenic organizing pneumonia etc. etc.
About the Lecture
The lecture Pneumonia: Treatment by Jeremy Brown, PhD, MRCP(UK), MBBS is from the course Infections of the Respiratory Tract.
Included Quiz Questions
Of the following, which pneumonia-causing pathogen is NOT paired with the appropriate curative antimicrobial?
- Amoxicillin for Chlamydophila pneumoniae
- Neuraminidase inhibitor for influenza
- Amoxicillin for Streptococcus pneumoniae
- Clarithromycin for Mycoplasma pneumoniae
A patient presenting to the emergency department with pneumonia is noted to be hypotensive with dry oral mucosae. The patient is appropriately volume resuscitated with IV fluids and broad-spectrum antibiotics are initiated. However, the patient's blood pressure remains low and the patient begins to exhibit signs of confusion. What is the next best step in the management of the patient?
- Start inotropes and refer to the intensive care unit.
- Start IV hydrocortisone and admit to the general medicine floor.
- Increase the fluid flow rate through the IV line and admit to the intensive care unit.
- Discharge without intervention.
Which of the following is a preferred diagnostic test for Legionella pneumophila?
- Sputum PCR
- Nasal bone cartilage biopsy
- Rapid nasal PCR
- Sputum culture
Which type of pneumonia is common in patients with active HIV infection?
- Community-acquired pneumonia
- Aspiration pneumonia
- Hospital-acquired pneumonia
- Interstitial pneumonia
- Atypical pneumonia
Which organism is most likely to cause pneumonia in an HIV-positive patient?
- Streptococcus pneumoniae
- Legionella pneumophila
- Mycoplasma pneumoniae
- Haemophilus influenzae
- Cytomegalovirus
According to UK guidelines, which of the following is the appropriate empirical therapy for a patient with moderate (CURB-65 score of 2) community-acquired pneumonia?
- Amoxicillin and clarithromycin
- Amoxicillin or ceftriaxone
- Amoxicillin or clarithromycin
- Co-amoxiclav and clarithromycin
- Ceftriaxone
A 66-year-old man is diagnosed with pneumonia. The patient's respiratory rate is 24 breaths per minute, blood pressure is 120/88 mm Hg, oxygen saturation is 95%, and urea level is 4 mmol. The patient appears confused. What is the preferred empirical treatment?
- Two or more IV antibiotics for 5–7 days
- An oral antibiotic for 5 days
- Wait several days and treat with an oral antibiotic if the symptoms remain.
- Treat immediately with an oral antibiotic for up to 5 days.
- Combination of oral and IM antibiotics for over 10 days
A patient diagnosed with pneumonia was hospitalized for 10 days. In spite of initial improvement with administration of amoxicillin/clavulanic acid and clarithromycin, the patient's condition began to deteriorate by day 6. Ticarcillin was added with good response and eventual resolution of symptoms. What was the probable causative organism in the patient?
- Pseudomonas aeruginosa
- Streptococcus pneumoniae
- Legionella pneumophila
- Mycoplasma pneumoniae
- Haemophilus influenzae
How much is CRP expected to fall in a patient with pneumonia responding to antibiotic therapy by day 3-4?
- 55%
- 30%
- 10%
- 40%
- 5%
Which of the following methods are useful to evaluate a patient with pneumonia not responding to treatment? Select all that apply.
- Perform a chest X-ray
- Perform MRI
- Wait for improvement
- Re-culture and check for resistant organisms
- Perform paracentesis
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |