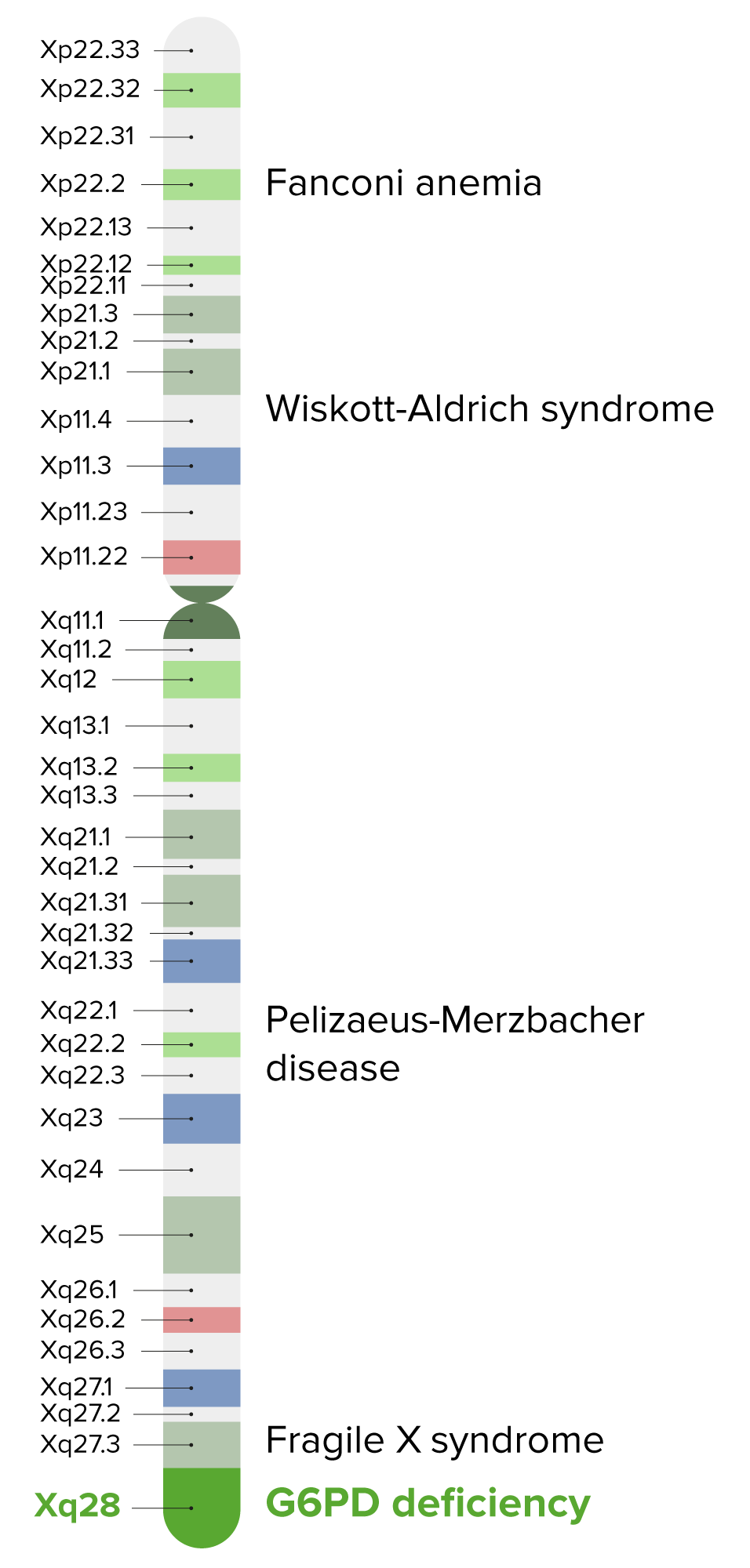Playlist
Show Playlist
Hide Playlist
Pediatric G6PD Deficiency
-
Slides G6PD Pediatrics.pdf
-
Download Lecture Overview
00:01 In this lecture, we're going to discuss glucose six phosphate dehydrogenase deficiency or G6PD. 00:08 G6PD happens because of a metabolic problem. Here's a glucose molecule. 00:14 Remember glucose is now broken down into pyruvate and that goes into the Krebs cycle to make ATP. 00:21 There's also gluconeogenesis in the event the liver wants to make more sugar. 00:25 Glucose has another enzyme from the glucose six phosphate molecule in that pathway in -- which is glucose six phosphate dehydrogenase. 00:37 That feeds into the pentose phosphate shunt whose job it is to make NADPH out of NADP. 00:45 NADPH is then used to make glutathione. 00:49 And the glutathione is responsible for maintaining our body's vigilant fight against oxygen free radicals. 00:57 If we have a deficiency of G6PD we can make less NADPH and that can result in particular for -- from our red cells from being incapable of defending themselves against oxidative stress. 01:12 So here's an example of a slide of a patient with G6PD. 01:17 What you can see is that the red blood cells have become a little bit more fragile and they're being cleared by the spleen, having this bitten out appearance that you can see in that cell right in the center of the slide. 01:29 When a special stain is used for their RBCs, patients may also be found to have Heinz bodies as shown in the slide here. 01:37 Heinz bodies are also seen in some thalassemias. 01:43 So, patients with G6PD are generally males. 01:48 That's because it's an X linked disorder. 01:51 And so it's almost exclusively in male patients. This is a prevalent disease. 01:56 It's present in about 1 to 2% of the US population and is more common in certain populations, in particular patients of African or Middle Eastern descent. 02:07 So, what happens to these patients? Generally, this can result in a male infant with a prolonged jaundice or even Frank kernicterus because of that red cell breakdown and increased indirect bilirubin. 02:22 And, because infants have a harder time clearing that bilirubin they can develop a prolonged jaundice. 02:28 Otherwise, development is pretty much normal and their CBC will look normal until the patient has an event of oxidative stress. 02:38 This happens because if for example an ingestion of a food product that has much oxidative stress within it. 02:46 And then these patients will develop an acute hemolytic anemia. 02:50 In severe cases, the hemolysis is chronic and there are no periods of relief. 02:57 But in most cases this is an episodic thing as a result of a stressful period. 03:02 So, what are common sources of oxidative stress in children? These are things that patients with this condition must avoid for their entire lives. 03:12 The classic one is Trimethoprim/Sulfamethoxazole or any sulfur containing medication. 03:19 Quinolones are also oxidative stresses. 03:24 And so children going to malaria prone areas or being treated with malaria drugs must avoid the quinolones. 03:30 Remember, if it ends in a quine, such as chloroquine, that's a quinolone. 03:35 Nitrofurantion can provide oxidative stress. 03:39 So we must avoid this drug especially for treatment of urine tract infections and simple cystitis. 03:45 Aspirin might cause problems so we should avoid aspirin. 03:48 And although it's not used very commonly, methylene blue should be avoided as well. 03:54 More commonly though, foods are the thing that get these patients in trouble and we have to avoid certain foods specifically fava beans or these broad based beans are a source of oxidative stress. 04:07 Blue food coloring can cause problems. 04:09 Tonic water because it has quinine in it can also cause problems. 04:13 And interestingly the gluten-free diet can cause problems. 04:17 This is because in some gluten-free foods, people insert, fava beans or some of those broad-based beans as a way to supplement protein. 04:26 So, package reading has to be carefully done. 04:30 There are randomly other things that can cause it. 04:33 Any infection can cause an oxidative stress. 04:35 In particular, mothballs if present can cause oxidative stress. 04:39 And diabetic ketoacidosis can cause oxidative stress in a patient who's unfortunate to have both G6PD and type I diabetes. 04:50 So, let's look at how these patients present. 04:55 Usually, during an oxidative stress moment when they have acute hemolytic anemia they'll have a period of acute irritability or fatigue. 05:04 These children will be tired and they'll be fuzzy. 05:07 And, you'll see signs of infection if that's their trigger but most importantly patients complain that they have Coca-Cola or cola contained -- colored urine. 05:17 That's from the hemoglobinuria as a result of the cell breakdown. 05:22 Patients on exam will be found to have pallor. 05:25 They may have tachycardia and they may have a flow murmur heard best at the upper sternal borders. 05:30 These patients may be jaundiced because they suddenly can't clear their cell breakdown. 05:35 They may have an acute Hepatosplenomegaly and in severe cases they might go into frank shock. 05:41 A CBC shows acute anemia and evidence of hemolysis and we may get some other labs as well. 05:49 An indirect bilirubin will be elevated as well as an LDH. 05:54 We can get a serum haptoglobin which will be abnormally low. 05:59 Haptoglobin binds to free hemoglobin that's why it's low in intravascular hemolysis. 06:08 So, some important key things about G6PD. 06:13 First off, always remember it's always about avoidance. 06:16 We have to counsel them in terms of things they need to avoid and medications they need to avoid. 06:21 If there is an acute hemolysis, we may well need to transfuse. 06:25 And for rare cases, especially those cases where there's no period during which they're not getting a break, we may pursue splenectomy as a way to mitigate some of that cell breakdown. 06:37 So that's a brief summary of G6PD. Thanks for your time.
About the Lecture
The lecture Pediatric G6PD Deficiency by Brian Alverson, MD is from the course Pediatric Hematology. It contains the following chapters:
- G6PD – Metabolic Pathways
- G6PD – Clinical Presentation
- G6PD – Diagnosis
Included Quiz Questions
Which of the following drugs is not appropriate in a G6PD deficient patient?
- Trimethoprim-Sulfamethoxazole
- Ibuprofen
- Tylenol
- ACE inhibitors
- Beta-blockers
Deficiency of which of the following can lead to less NADPH and less glutathione leading to unstable RBCs?
- Glucose 6 phosphate dehydrogenase
- Hexokinase
- Glucokinase
- Pyruvate kinase
- Phosphokinase
In G6PD, which of the following is seen in the blood peripheral film?
- Bite cells and heinz bodies
- Burr cells
- Target cells
- Sickle shaped cells
- Increased reticulocytes
When counseling a mother whose child has G6PD, you ask the mother to avoid all of the following, except?
- Penicillin
- Sulfa drugs
- Aspirin and quinolones
- Blue food coloring
- Fava beans
A neonate is suspected to have G6PD if he has...
- …prolonged jaundice
- …prolonged cyanosis
- …persistent hypoglycemia
- …polycythemia
- …splenomegaly
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
Excellent lecture. Crystal clear: easy to understand, easy to remember. Thanks!