Playlist
Show Playlist
Hide Playlist
Hypertrophic Cardiomyopathy: Pathophysiology and Clinical Manifestations
-
Slides Cardiomyopathy.pdf
-
Reference List Pathology.pdf
-
Download Lecture Overview
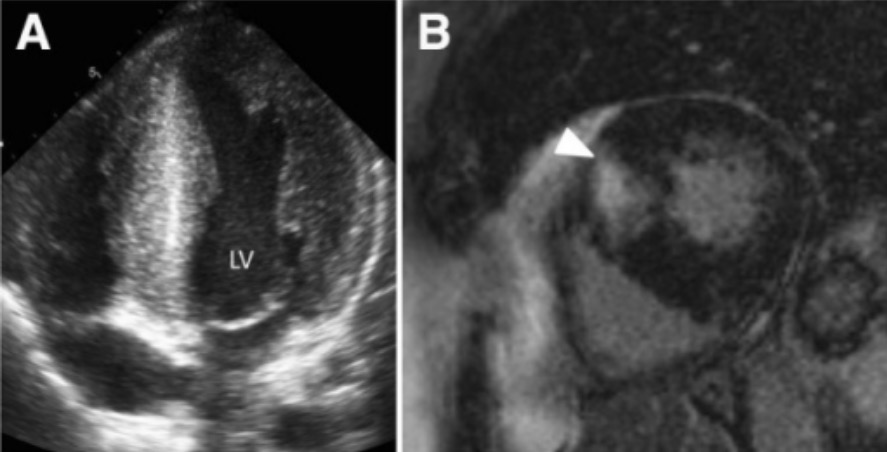
00:01 So hypertrophic cardiomyopathy has 100% genetic basis. 00:06 We now understand this and the vast majority are autosomal dominant. 00:10 So if mom or dad has it, 50% of the kids will have it as well. 00:15 The main effect of proteins where wetalked about, 60-70% of the time involves beta-myosin heavy chain mutations. 00:24 But there are also other proteins that regulate the interaction of actin and myosin. 00:29 So myosin binding protein C, troponin T tropomyosin, and others may be involved. 00:37 What happens? We get hypercontractility, the sarcomeres squeeze really well. 00:42 They don't relax, they don't release myosin heavy chain from the accent quite as well as they should, so we tend to have a very muscular heart that is hypercontractile but poorly relaxing. 00:55 The phenotype is myocyte hypertrophy, we classically see myocyte or myofiber disarray, those are the classic buzzwords, and I'll show you what that looks like in a moment. 01:07 That's the classic appearance on histology. 01:10 And it's basically an exaggeration of the normal cardiac myocyte appearance. 01:16 So as I've already mentioned previously, in other talks, the cardiac myocyte is not a boxcar, it is not a rectangle. 01:24 It's actually a branched cell that interacts in multiple ways with adjacent cardiac myocytes. 01:31 As the individual cells undergo prominent hypertrophy, we've just seen exaggeration of that normal kind of branching architecture, but then we can recognize it as myofiber disarray. 01:41 There will be fibrosis, so in the same way that the cardiac myocytes are squeezing and becoming hypertrophic. 01:49 They also tug very vigorously on the associated cardiac fibroblast. 01:55 And those fibroblasts when they feel increased pressure and squeezing like that, they elaborate more connective tissue so you will see interstitial fibrosis. 02:06 You can have left ventricular outflow tract obstruction, and that is going to be the HOCM, H-O-C-M, hypertrophic obstructive cardiomyopathy phenotype. 02:16 And one of the other manifestations that we can see on histology are thickened septal vessels. 02:22 A way to think about this is that the cardiac myocytes are squeezing so vigorously, the poor blood vessels that are permeating into the muscle of the myocardium are trying to keep themselves open and not be squeezed shut. 02:35 So they get a very thickened wall. 02:37 I'm not sure that that's the accurate description of why that happens, but it does happen, make sense to me. 02:44 You end up with poor left ventricular relaxation, poor filling, diastolic dysfunction. 02:50 As a result, you don't fill the ventricle. 02:54 And so you get reduced systolic output volume. 02:56 And particularly if you have HOCM where you get that premature closure of the left ventricular outflow tract, you will have a markedly reduced systolic output. 03:06 So even with a heart that's squeezing really well, you may not have the same volume of blood perfusing the peripheral tissues, so you have reduced peripheral perfusion. 03:16 And as a result of reduced myocardial perfusion, you may actually now worsen the entire process by also imposing an ischemic component. 03:28 Overall, these patients are going to be very much predisposed to cardiac arrhythmias and/or heart failure. 03:35 The cardiac arrhythmias are happening because you have markedly enlarged cardiac myocytes and all the cells are individually larger, the number of capillaries perfusing those do not increase. 03:46 So you have a relative intracellular ischemia, with a propensity to have arrythmias. 03:56 Signs and symptoms, so we don't typically see hypertrophic cardiomyopathy, until individuals go through their pubertal growth spurt. 04:07 And during that time, there are profound changes in the expression of various genes, including some of the cardiac sarcomeric proteins, and that's when we start, beginning to see the manifestations of hypertrophic cardiomyopathy. 04:22 Importantly and tragically, hypertrophic cardiomyopathy is a cause of almost a third of sudden cardiac deaths and athletes younger than 35 years of age. 04:31 And again, this is because they have an hypertrophied heart that is prone to ischemia, they then exercise and they may get a sudden, arrhythmic death What are the other signs and symptoms? Well, basically, this is usually frequently asymptomatic, particularly the non-obstructive type, So if you have okay outflow, and you don't get a syncopal episode or you don't have arrhythmias, you may not have much in the way symptomatology. 05:01 We typically pick up on kindreds who are affected by the disease. 05:08 When we have an initial individual who develops a sudden cardiac death or has some sort of episode, we identify hypertrophic cardiomyopathy, and because it's autosomal dominant dominant, we go back and look at all of the related family members. 05:22 And in that case, we can pick up those individuals. 05:25 and we will try to prevent the consequences. 05:30 So we will give them antiarrhythmics, we will give them drugs that will reduce myocardial contractility. 05:35 We'll put in cardiac pacers, so they don't have a fatal arrhythmia. 05:41 So the patients are frequently asymptomatic. 05:44 and if they do have symptoms, this is going to be due to either regurgitant flow, because we have we have moved the entry leaf of the mitral valve out of the way and we have now insufficiency of the mitral valve with pressure and volume going into the lungs. 06:01 So exertional dyspnea. 06:03 You can also have diminished forward flow, and so patients will present with systemic hypotension, which we manifested as dizziness, or lightheadedness or frank syncope. 06:14 If you have diminished perfusion to the cardiac muscle cells, we can have angina, and in fact, can be a cause of a myocardial infarct. 06:26 Patients can present with arrhythmias, which to them will feel like palpitations, the heart is just beating out of their chest or is beating irregularly. 06:36 And because of the tight left ventricular outflow, you can have a very harsh systolic ejection murmur. 06:43 So signs and symptoms of hypertrophic cardiomyopathy.
About the Lecture
The lecture Hypertrophic Cardiomyopathy: Pathophysiology and Clinical Manifestations by Richard Mitchell, MD, PhD is from the course Cardiomyopathy.
Included Quiz Questions
During which period of life does hypertrophic cardiomyopathy become clinically significant?
- Postpubertal growth spurt
- Neonatal period
- Fourth or fifth decade of life
- Old age (after age 65)
- Pregnancy
What mechanism leads to poor peripheral perfusion among patients with hypertrophic cardiomyopathy?
- Reduced systolic output
- Overfilling of the right ventricle
- Electrical conduction abnormality
- Overfilling of the left ventricle
What is a common symptom of hypertrophic cardiomyopathy?
- Exertional dyspnea
- Weight loss
- Well-demarcated subcutaneous masses
- Cluster headaches
- Excessive thirst
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |