Playlist
Show Playlist
Hide Playlist
Nitrogen, Nitrogen Cycle and Excretion of Nitrogen
-
Slides UreaCycle Biochemistry.pdf
-
Reference List Biochemistry.pdf
-
Download Lecture Overview
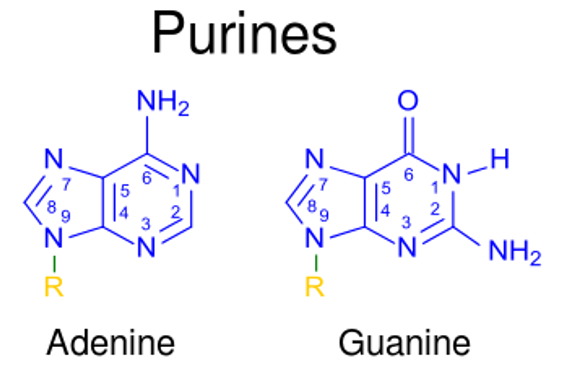
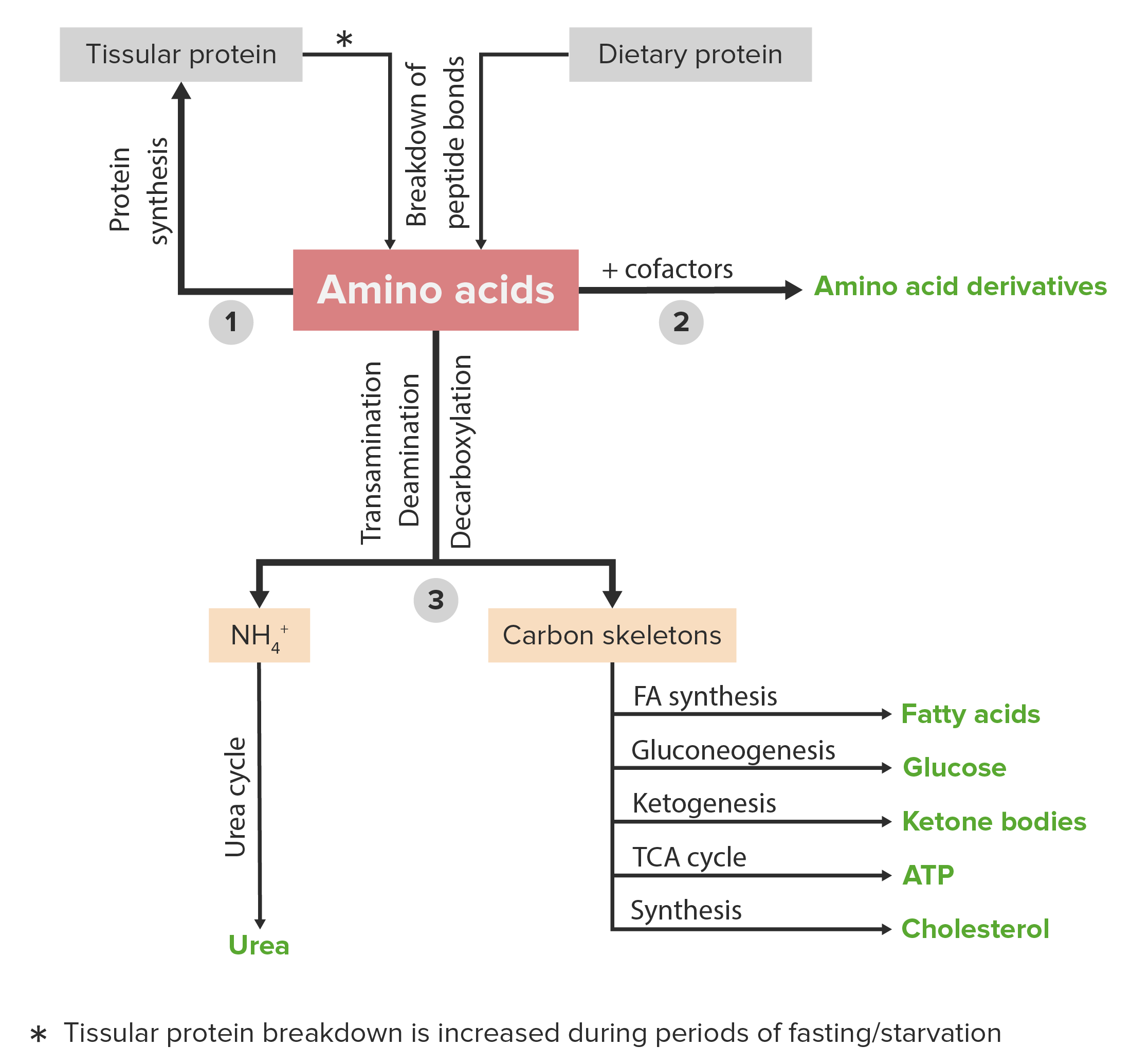
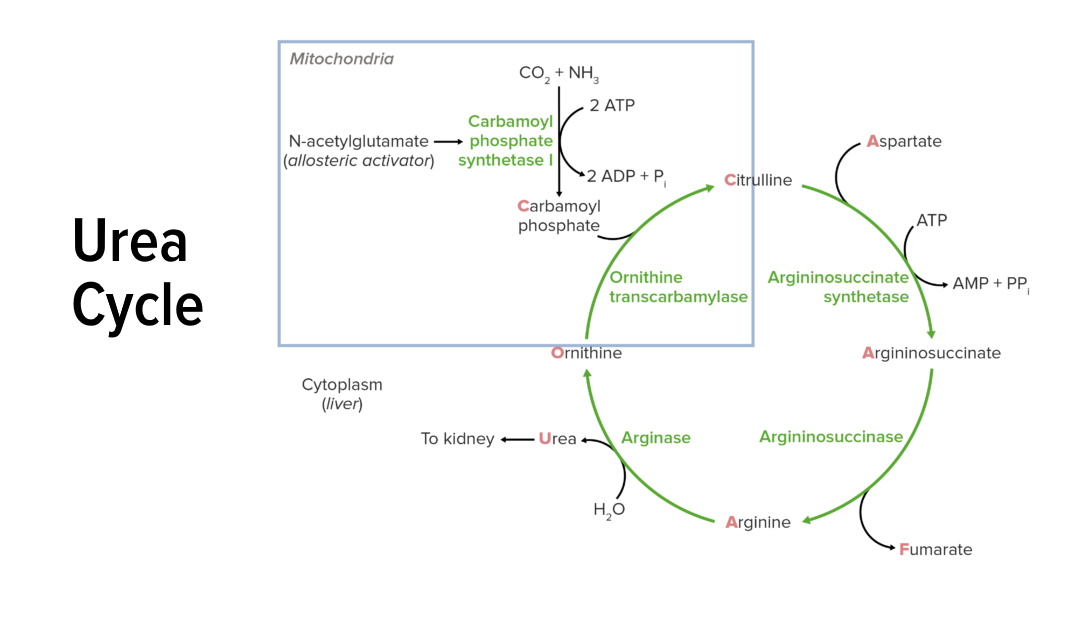
00:01 In den Vorlesungen über den Aminosäurestoffwechsel, habe ich darüber gesprochen, wie Amine von einem Molekül zum anderen bewegt werden, und wie Ammoniak ein giftiges Nebenprodukt ihres Stoffwechsels darstellt. 00:11 In diesem Vortrag werde ich darüber sprechen, wie diese Amine und der Ammoniak kombiniert und für die Ausscheidung vorbereitet werden. 00:18 Stickstoff kommt im Körper, wie wir gesehen haben, in verschiedenen Formen vor. 00:22 Das Gleichgewicht ist absolut entscheidend. 00:24 Stickstoff ist ein essentieller Nährstoff, aber es ist auch eines, das in zwei hohe Mengen giftig sein können. 00:30 Der Körper muss in der Lage sein, den Stickstoff aus dem Aminosäurestoffwechsel zu verarbeiten. 00:35 Stickstoff wird aber auch für die Synthese anderer Moleküle benötigt. 00:38 Dazu gehören die Nukleotide ATP, GTP, CTP, UTP, dATP, dCTP, dGTP und dTTP. 00:47 Stickstoff wird auch für die Herstellung von Nicht-Protein-Aminosäuren benötigt. 00:51 Dazu gehören Ornithin, Citrullin und Sarkosin, wie auf dem Bildschirm zu sehen ist. 00:56 Und es gibt noch andere stickstoffhaltige Verbindungen, die für die Synthese im Körper notwendig sind. 01:01 Dazu gehören Cholin, verschiedene Vitamine und Carnitin. 01:07 Der Stickstoffkreislauf ist ein sehr wichtiger Kreislauf in unserer Umwelt. 01:10 Der Stickstoff in unserer Atmosphäre muss reduziert werden. 01:14 Es gibt nitrifizierende Organismen in den tiefsten Teilen unseres Bodens, die Stickstoff aufnehmen und ihn in Amine umwandeln, die für höhere Organismen nützlich sind. 01:23 Dieser Gesamtprozess ist auf der Folie dargestellt. 01:28 Nun muss Stickstoff ausgeschieden werden, wenn das Gleichgewicht aufrechterhalten werden soll. 01:33 Wie ich bereits erwähnt habe, machen Aminosäuren durch Transaminierung Stickstoff mobil. 01:38 Es ist sehr einfach, den Stickstoff von einer Aminosäure zu einer Ketosäure und umgekehrt umzuwandeln. 01:45 Die Toxizität von Ammoniak bedeutet, dass der Stickstoff, der produziert wird, als Nebenprodukt des Aminosäurenstoffwechsels anfällt, exakt ausgeschieden werden muss. 01:54 Im Prozess der Ausscheidung gibt es verschiedene Strategien, die von verschiedenen Organismen angewandt werden. 02:00 Es gibt beispielsweise Organismen, die ammonotelisch sind - also Ammoniak ausscheiden. 02:04 Das gilt auch für Fische, und deshalb müssen Sie Ihr Aquarium regelmäßig reinigen. 02:08 Uricotelische Organismen scheiden Harnsäure aus, darunter auch Vögel. 02:14 Ureotelische Organismen schließen den Menschen ein. 02:16 Diese scheiden Harnstoff aus und dazu gehören auch die meisten Wirbeltiere und ein paar Wirbellose. 02:22 Wenn wir uns nun die Struktur dieser Moleküle ansehen, sehen wir natürlich, dass Ammoniak nur einen Stickstoff enthält. Dieser hat aber den Vorteil, dass er wasserlöslich ist. 02:31 Ammoniak hat natürlich den Nachteil, dass er giftig ist. 02:34 Harnsäure ist ein sehr gutes Mittel, um eine Menge Stickstoff loszuwerden. 02:37 Es hat vier Stickstoffe in sich, aber leider hat es den Nachteil, dass es nicht sehr wasserlöslich ist. 02:44 Harnstoff ist sozusagen der perfekte Kompromiss zwischen den beiden. 02:47 Er enthält zwei Stickstoffverbindungen für die Ausscheidung und ist sehr wasserlöslich. 02:52 Der Ammoniak, der beim Aminosäureabbau entsteht, wird im Harnstoffzyklus verwendet. 02:58 Und die Harnsäure, die durch den Purinabbau entsteht, ist ein Nebenprodukt des Abbaus von Nukleotiden. 03:05 Und schließlich wird Harnstoff durch den Harnstoffzyklus produziert.
About the Lecture
The lecture Nitrogen, Nitrogen Cycle and Excretion of Nitrogen by Kevin Ahern, PhD is from the course Amino Acid Metabolism.
Included Quiz Questions
Which of the following is true regarding nitrogen in the body?
- Proper balance of nitrogen is critical.
- Nitrogen is not needed for the synthesis of nucleotides.
- Nitrogen is not needed for the synthesis of ornithine.
- Nitrogen is needed for fatty acid synthesis.
- Nitrogen is only needed for protein-based amino acids.
Which of the following is true regarding nitrogen excretion?
- The ureotelic organisms include most vertebrates.
- The ammonotelic organisms are primarily birds.
- The uricotelic organisms excrete urea.
- The uricotelic organisms are mainly humans.
- Ammonotelic organisms excrete uric acid.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |