Playlist
Show Playlist
Hide Playlist
Future Needs for Vaccines: Influenza, COVID, Malaria, Tuberculosis, HIV
-
Slides Vaccine Immunology.pdf
-
Reference List Immune System.pdf
-
Download Lecture Overview
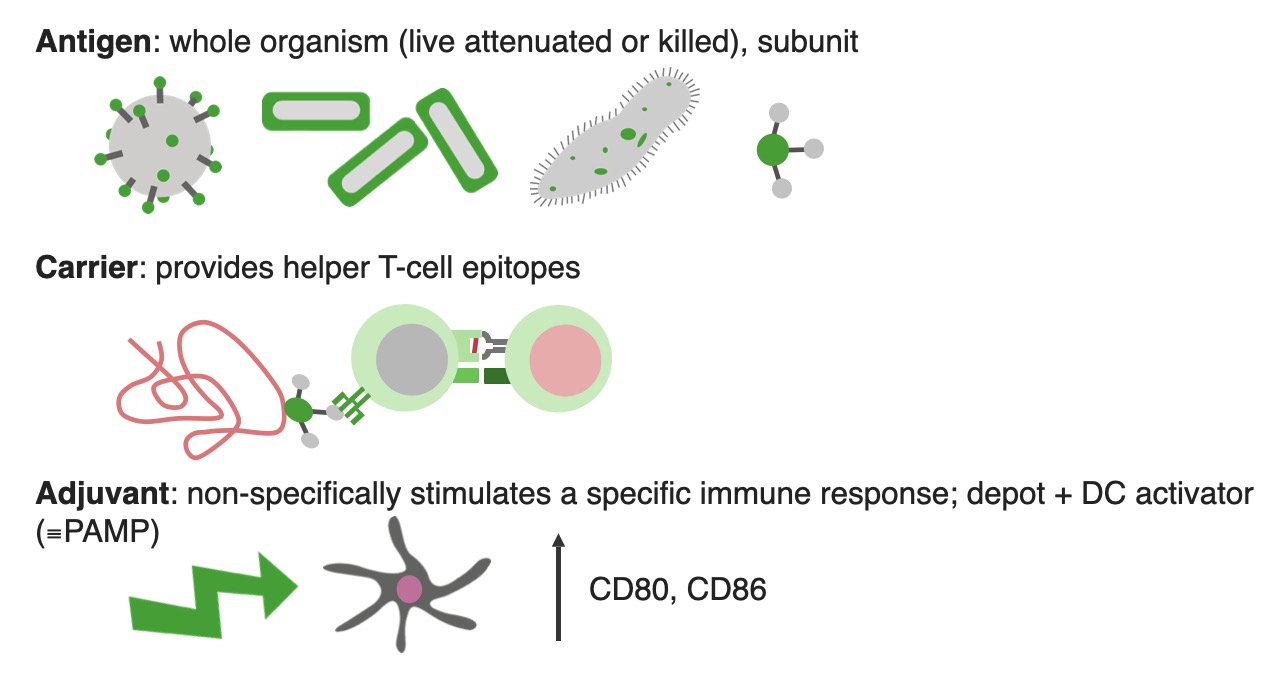
00:01 There is a need for both new vaccines and improved versions of existing vaccines. 00:09 For example, better vaccines against influenza, COVID, malaria, TB, and we still desperately need a vaccine against HIV. 00:23 The vaccines against influenza are perfectly good. 00:26 The problem is that the influenza virus is changing all the time by mutations. 00:33 And therefore the vaccine needs to be administered every year to provide protection against new variants of the virus. 00:42 The influenza virus can undergo two types of genetic change. 00:46 We call them antigenic drift and antigenic shift. 00:51 Antigenic drift refers to point mutation. 00:55 The neuraminidase and hemaglutinin antigens in the influenza virus can undergo point mutations. 01:05 And as you can see in this diagram with the little red dot; if you look carefully there you’ll see a little red dot has appeared. 01:13 This is to indicate a point mutation that will lead to a slight change in one or other of those two antigens - the neuraminidase or the hemaglutinin antigens. 01:24 This will mean that people exposed to the previous version will have partial immunity but not full immunity to this new variant. 01:34 In contrast, antigenic shift involves the exchange of genetic material. 01:41 So for example, there may be exchange between a human influenza virus and an avian influenza virus. 01:49 This exchange of genetic material creates a completely new strain of human influenza virus to which hardly anybody is immune. These new variants of the virus can then sweep through the world and cause pandemics. With the current annual vaccine for influenza, there is a global surveillance for emerging strains. 02:18 The FDA advisory panel selects four strains, that is thought will be the dominant strains for the coming winter season. These are manufactured and tested. 02:33 They’re distributed and then individuals are vaccinated. 02:39 However because this has to occur every year, the time scale means that there is insufficient vaccine to vaccinate everybody. 02:48 There is ongoing research based on a variety of different strategies aimed at developing a universal influenza vaccine. 02:59 So the hope is in the future, we will have a influenza vaccine that is able to protect against a much wider number of variants of influenza. But at the moment, because of the time scale involved and the limited amount of vaccine that can be produced prior to the winter season, only a particular at risk groups are usually vaccinated. So the elderly, people with diabetes and other conditions that make them more prone to infection. There is a real need for a malaria There are a number of different COVID vaccines available, either utilizing the whole virus in an inactivated form of words and inactivated SARS-CoV-2 virus, or employing the spike protein or s protein that contains the receptor binding components of the virus. 03:53 This can be the S protein in a non replicating had no virus vector. 04:00 The S protein coated onto synthetic nanoparticles or lipid nanoparticle encapsulated messenger RNA that encodes the S protein. 04:14 There is a real need for a malaria vaccine and some progress is being made. Malaria is caused by the Plasmodium protozoan parasite, and there is a vaccine that is called RTSS/AS01. This comprises a portion of the circumsporozoite protein of Plasmodium falciparum fused to a Hepatitis B surface antigen. 04:51 It targets the pre-erythrocyte stage of the parasite. 04:56 So by no means a perfect vaccine, but at least some progress in towards the production of a effective malarial vaccine. The BCG vaccine for tuberculosis is an attenuated live Bacille Calmette Guerin strain of Mycobacterium bovis. 05:18 It is used in countries with a high prevalence of TB to prevent childhood tuberculous meningitis and miliary disease. 05:28 It is not generally recommended in the USA due to a low risk of infection with Mycobacterium tuberculosis and a variable effectiveness against adult pulmonary TB and a potential interference with the tuberculin skin test. 05:44 It is considered only for children who have a negative tuberculin skin test and a high risk from exposure. 05:53 For example, if they are continually exposed to isoniazid and to rifampin resistant M. TB. 06:03 Healthcare workers who are routinely exposed to drug resistant M. TB are also vaccinated. 06:14 Regarding HIV vaccines, success requires identification of immunogens and an immunization strategy that induces broad and long lasting cytotoxic T-lymphocyte immunity together with broadly neutralizing antibodies. 06:34 Broadly neutralizing antibodies have been identified in a very small number of people living with HIV. 06:41 And intravenous infusion of such antibodies is entering clinical trials. 06:48 There’s no vaccine tested in clinical trials so far that has been sufficiently successful. 06:56 The only hint of any success whatsoever was a trial carried out in Thailand in 2009. 07:03 This involved giving four priming injections of a recombinant canarypox vector containing HIV gag, pol and env genes. Two booster injections were given with a recombinant gp120. There was a limited protective effect with an efficacy of 25-30%. 07:28 The future needs for vaccination are first of all, the eradication of polio which has seemed so close for a number of years now, and hopefully will be achieved in the not too distant future. 07:41 Effective vaccines for HIV, TB and malaria. 07:47 A broadly specific influenza vaccine so that individuals don’t need to be vaccinated every single year. 07:54 Therapeutic vaccines for those already exposed to pathogens; so a hepatitis vaccine, HIV vaccine and vaccines against cancer. 08:05 And also there is a need for vaccines against a number of parasitic diseases.
About the Lecture
The lecture Future Needs for Vaccines: Influenza, COVID, Malaria, Tuberculosis, HIV by Peter Delves, PhD is from the course Vaccine Immunology. It contains the following chapters:
- Future Needs for Vaccines
- Influenza Vaccines
- Malaria Vaccines
- Tuberculosis Vaccines
- HIV Vaccines
Included Quiz Questions
Which of the following vaccines require annual or biannual administration in some populations?
- Influenza
- Tetanus
- Pertusis
- BCG
- Varicella zoster
Which of the following terms refers to the accumulation of point mutations in virus genes that result in changes in virus-surface proteins that host antibodies recognize?
- Antigenic drift
- Antigenic shift
- Genetic drift
- Epigenetic modification
In the Unites States, the BCG vaccine for tuberculosis (TB) is recommended for which of the following populations?
- TB exposed health care workers in high risk settings
- All children
- All exposed individuals
- TB exposed tuberculin skin test-positive infants and children
- TB exposed tuberculin skin test-positive adults
What is Bacillus Calmette–Guérin vaccine?
- An attenuated strain of Mycobacterium bovis
- An inactive strain of Mycobacterium bovis
- An attenuated strain of Mycobacterium bacteria
- An inactive strain of Mycobacterium tuberculosis
- A Mycobacterium tuberculosis antigen
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |