Playlist
Show Playlist
Hide Playlist
IgA Nephropathy – Nephritic Syndrome
-
Slides Nephritic Syndrome.pdf
-
Reference List Nephrology.pdf
-
Download Lecture Overview
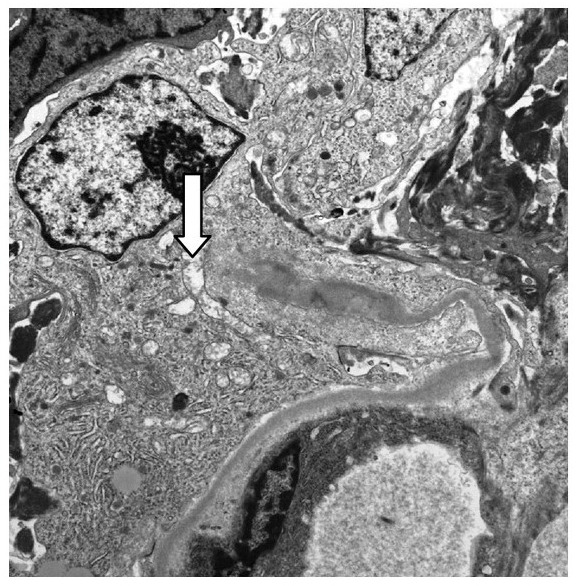
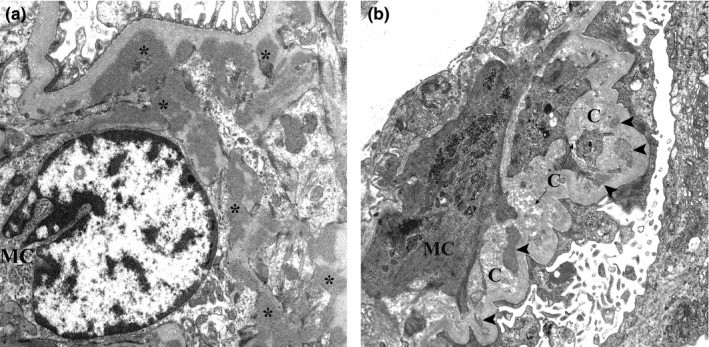
00:01 So, let's talk about IgA. It is the most common cause of glomerulonephritis worldwide. 00:07 The peak incidence is in the 2nd and 3rd decade of life, and the highest incidence is actually seen in Asian and Caucasian populations. 00:16 When we think about the etiology of IgA nephropathy, it's really triggered by environmental antigens that drive the generation of pathogenic IgA immune complexes. 00:28 Those antigens can be viral, it could bacterial, or it could be food. 00:33 And that makes sense because think about where IgA is. 00:36 It is that protector of the mucosal host defense system. 00:40 So, it's natural that these particular antigens are going to trigger pathogenesis of those abnormal IgA immune complexes. 00:48 There's a genetic predisposition and that it is polygenic, many genes are involved, and like almost any glomerular disease that we've talked about, I just wanna take a moment to remind you that when we're talking about glomerular diseases, patients will often have a genetic predisposition, an environmental trigger, and a tendency towards autoimmunity and that's exactly what's happening here. 01:12 Now, there are some familial IgAs so it can affect the multiple family members in certain clusters. 01:18 So, when we think about the pathogenesis, again, this is a disorder of the mucosal IgA synthesis. 01:24 Under normal conditions, IgA is gonna form an immune complex with a mucosal pathogen or antigen. 01:33 Once it does that, the galactose residue on that IgA will allow that immune complex to be cleared from the circulation by the spleen. 01:41 But in IgA, what ends up happening is we have -- or the patient will have an increase in synthesis of abnormal IgA. 01:51 So, in particular, there's an abnormal area of glycosylation at the hinge region that's produced in those particular patients. 01:59 Now, because of that abnormal glycosylation at that hinge region, then a patient with IgA will have another antibody, an IgG antibody, that's gonna be targeted against that hinge region because it recognizes that as foreign. 02:16 Patients then have this large macromolecular complex that's circulating within the serum and instead of getting picked off by the spleen, those macromolecular complexes then deposit preferentially in the mesangium of the kidney. 02:30 And when you have that, remember, we've got immune complex formation here, then you have complement activation. It is seen in the mesangium, it's at a local level, so we're not gonna see serum complement getting used up. 02:45 So, again, if I'm looking for a diagnosis and I'm suspecting that my patient has IgA, there's nothing serologically that I can get that's necessarily going to help me. 02:55 There's no serum complement abnormalities. 02:57 We talked about complement being activated but it's very local. It's where the mesangium is. 03:01 Now, I do have to keep in mind some differentials on my list. 03:05 I need to think about other people who might have isolated persistent glomerular hematuria. 03:10 This is gonna be somebody who might have thin basement membrane disease and what that means is that their glomerular basement membranes are thinner than normal and therefore, can lead to hematuria. 03:21 And even that hematuria can have dysmorphic features but they have a relatively benign course. 03:26 The other one that I wanna think about is hereditary nephritis or Alport's syndrome. 03:31 An Alport's syndrome is essentially a genetic disease that manifest with sensorineural hearing loss and ocular abnormalities, and that's due to mutations in the alpha 3 through 5 chains of type 4 collagen. 03:45 Okay, so, if my patient has IgA nephropathy and I really wanna clinch that diagnosis and there's nothing serologically that's gonna help me, then a definitive biopsy or a definitive diagnosis can be made with biopsy. 03:57 And what am I gonna see on biopsy? So, our first image shows light microscopy. 04:03 This is a hematoxylin and eosin stain which is going to highlight our cells, and what you can see and what the arrow is pointing to is our mesangial region. 04:11 That region has exploded a little bit. It's a little bit larger than normal. 04:16 Why? Because we have mesangial hypercellularity. 04:20 On immunofluorescence, my heavy chain IgA is going to stain in those mesangial regions. 04:26 And on electron microscopy, I'm going to see these denser deposits or these darkening which represents her immune complex and deposits within the mesangial region. 04:39 So, in terms of how people present over time and how they manifest clinically, about 40% of patients will actually manifest with gross hematuria and often times, it's synpharyngitic with an upper respiratory infection. 04:52 About 30% or more can present with just microscopic hematuria and mild proteinuria. 04:58 So, it may be incidentally detected on a urine analysis, often times people will be at the physician or at the emergency department seen for something else and they notice that they have hematuria and proteinuria, and that stimulates or precipitates a visit to the nephrologist where they ultimately come to the diagnosis. 05:16 And then, there is about 5-10% of people who can present with acute nephritic syndrome more like a rapidly progressive glomerularonephritis and they lose their function very quickly. 05:28 So, patients may have some associated abnormalities of the skin and GI tract, and we should pay attention to that because if that patient is presenting in front of you and they have any of these associations, it really has to get you thinking about IgA nephropathy. 05:41 So, for skin lesions, it's been associated with dermatitis herpetiformis. 05:45 So, that's an uncommon autoimmune cutaneous eruption of the skin that manifest often times with intensely pruritic inflammatory papules and vesicles on the forearms, knees, and scalps. 05:57 That also has been associated with gluten sensitivity. 06:01 From a GI perspective, patients may have celiac sprue that makes sense, we just talked about gluten sensitivity, or cirrhotic liver disease. 06:08 Patients can also manifest with a vasculitic presentation or IgA vasculitis. 06:16 That's also called Henoch-Schonlein purpura, a systemic vasculitis that involves IgA nephropathy in the kidneys but they also have purpuric skin lesions as well as GI manifestations with pain. 06:29 So, how do patients do over time? Because it's such a wide spectrum of disease, it's really variable. 06:36 There are patients who have IgA that would never know it but on post-mortem, it's discovered. 06:41 And there are other people who are diagnosed and essentially progress to the need of end-stage renal disease renal replacement therapy within a 5-year period of time. 06:50 If we take all comers together, it's a relatively slow progression to end-stage renal disease, somewhere on the order of 15-40%. 06:58 So, how do we treat our patient? If they're sitting here before us, we biopsy them, we understand that they have IgA nephropathy, there's a couple of things that we keep in mind when it comes to treatment. 07:09 Number one, we don't have to treat patients at all. If they have normal renal function and they have minimal proteinuria, there's nothing that we can do at this point in time from an immune targeted therapy or other therapy that will help that patient in terms of overall renal survival. 07:27 They already have risk factors that are very favorable for not progressing so no treatment is warranted in that particular group. 07:34 Now, for patients who do manifest with proteinuria somewhere around 500 mg to 1 g/day, it's going to be essential to use a non-specific therapy like ACE inhibition or angiotensin receptor blockers. 07:48 Remember how these medications work. 07:50 These suppress the RAAS system, that renin-angio-aldo system, that ang-II gets antagonized, that's working directly at the efferent arteriole, it dilates it, we drop our glomerular capillary pressure, and we reduce proteinuria. 08:03 So, that's essential for patients who have IgA and some protein in their urine. 08:07 SGLT2 inhibitors are often used for diabetic kidney disease with proteinuria, and studies are demonstrating a benefit for nondiabetic kidney disease with proteinuria, including IgA nephropathy. An SGLT2 inhibitor can be used in addition to angiotensin inhibition for these patients. 08:28 Now, you might hear about fish oil. 08:30 Fish oil has been discussed probably for the last 3 decades and initially, when taking their randomized control trial that showed taking 12 g of fish oil per day may have a benefit in patients who have IgA nephropathy. 08:45 Now, this has been repeated and there have been several meta-analyses out there that show that fish oil probably does not really alter the course of IgA nephropathy. 08:54 So, the studies really are equivocal and if you were to take fish oil in what those randomized control trials actually designated, 12 g of fish oil is quite a bit to take and a lot of patients may not be able to tolerate that. 09:07 So, when it comes to treating a patient, there is no data behind it but some patients might read that and they often times will volunteer to take fish oil and there is no problem if they would like to do so. 09:20 Finally, there's prednisone and prednisone is really reserved for patients who have proteinuria greater than 1 g/day despite ACE inhibition or ARB therapy. 09:31 So, if you've maximized your ACE inhibitor, you've maximized your angiotensin receptor blocker, blood pressure's well-controlled, yet they still have proteinuria and they have a relatively preserved renal function, then prednisone may be indicated. 09:46 The data does show that it can decrease proteinuria but in terms of overall renal survival, the jury is still out.
About the Lecture
The lecture IgA Nephropathy – Nephritic Syndrome by Amy Sussman, MD is from the course Nephritic Syndrome.
Included Quiz Questions
Which of the following extrarenal manifestations is associated with IgA nephropathy?
- Skin lesions
- Meningitis
- Whipple disease
- Peptic ulcer disease
- Malignancy
Which of the following is characteristic of IgA nephropathy?
- Synpharyngitic hematuria
- Low complement levels
- Onset in the fourth decade of life
- Ocular abnormalities
Which of the following histopathologic changes is associated with IgA nephropathy?
- IgA deposition in the mesangium
- Spikes and domes in the basement membrane
- Nodular glomerulosclerosis
- Duplication of the glomerular basement membrane
- C3 deposition in a ribbon-like distribution
Which of the following is involved in the pathogenesis of IgA nephropathy?
- The production of abnormally glycosylated immunoglobulins
- Autoantibodies that stabilize C3 convertase
- Mutations in genes encoding type IV collagen
- Deposition of abnormally folded proteins with a beta-pleated sheet structure in the kidney
- Nonenzymatic glycosylation of the basement membrane
A patient with IgA nephropathy presents with minimal proteinuria and normal renal function. Which of the following is the most appropriate treatment option?
- No treatment is indicated.
- ACEI/ARBs
- Prednisolone
- Fish oil
- Statin
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
I couldnt understand the pathology for so long. I am so glad for the clear diagrams and interesting talk.