Playlist
Show Playlist
Hide Playlist
Hemodialysis and Peritoneal Dialysis: Procedure
-
Slides Renal Replacement Therapy.pdf
-
Reference List Nephrology.pdf
-
Download Lecture Overview
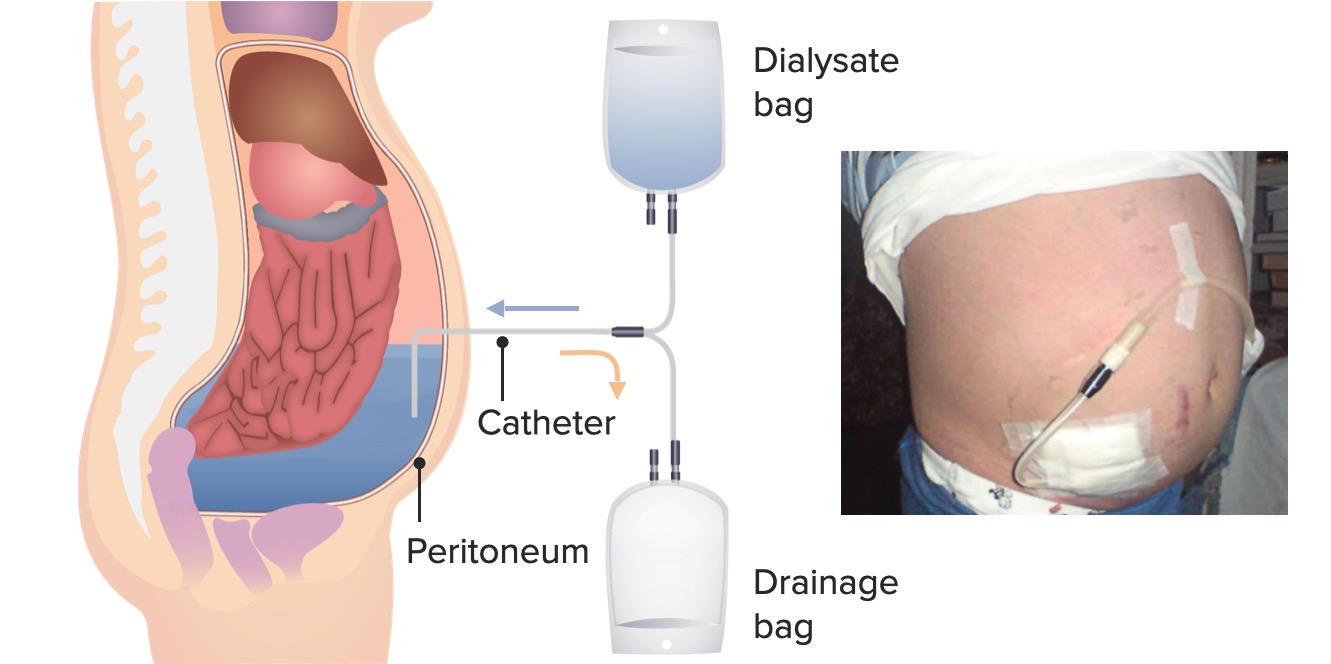
00:01 Okay, so let's turn back to hemodialysis. 00:04 So let's talk about how solutes are transported across that dialyzer. 00:09 I want you to remember what we're talking about again. 00:11 In hemodialysis, I essentially have a patient hooked up to a dialysis machine, blood is going to be coming from that patient going through an extracorporeal circuit, it goes through the dialyzer. 00:22 That dialyzer is separated by a semipermeable membrane on the other side or other compartment of that dialyzer is something called dialysate, that's dialysis solution. 00:32 And it contains, essentially, what I want my patients blood to look like. 00:37 A sodium concentration of typically around 137 mL equivalents Calcium closer to 10. 00:43 I want that potassium if they have high potassium in their blood to be low, and then bicarb to be high, because my patients remember are not being able to essentially get rid of the acid production that they make every day. 00:57 So that is then separated by that semipermeable membrane. 01:00 The blood flow and the dialysate flow go counter current to each other, the blood is then returned back to the patient through their dialysis catheter, or through their vascular access. 01:12 So the mechanism by which we do solute transport release is going to depend on diffusion. 01:18 And that again, is going to depend on that semipermeable membrane. 01:21 How porous are those holes essentially, in that semipermeable membrane, It's going to depend on the molecular weight of the solute. 01:29 How big is that solute? Something like urea, which is so tiny, can easily diffuse across that membrane. 01:35 But something bigger, might take a lot longer phosphorous or creatinine might take a little bit more time to dialyze across that membrane. 01:42 Certainly things like beta-2 microglobulin or bigger molecules like the aliphatic amines are going to be more time dependent, and will take time to go across that membrane. 01:52 Also the speed with which these are passing each other. 01:55 So if I have a very high blood flow in my patient flowing across that membrane, and a very high dialysis flow moving counter current to that, that's also really going to increase my diffusive efficacy. 02:08 I also depend a little bit when I'm talking about solute transport on ultrafiltration. 02:13 So ultra filtration is essentially applying a negative pressure, a negative hydrostatic pressure on that dialyzer. 02:21 And by doing so, I'm able to pull out water from that blood volume. 02:25 When I do that, the more ultrafiltration or the more the hydrostatic pressure, then I actually can drag out solute as well by solvent drag. 02:35 That's also the mechanism of volume removal that I'm talking about are ultrafiltration. 02:40 So again, our patients are coming in not only with the solutes that need to be essentially cleared, they also have excess volume. 02:48 They're edematous. 02:49 They have they're retaining sodium and water. 02:52 So the best way to remove that is to apply that negative pressure or hydrostatic pressure to that dialyzer thereby removing water, and then with that by solvent drag, we have sodium removal as well. 03:06 In peritoneal dialysis, I'm actually going to use that peritoneum as my dialyzer. 03:13 So that's my semipermeable membrane. 03:16 So essentially my dialysis fluid is going to be in different volumes. 03:22 So I can have either liters or five liter bags depending on my patient, whether they're going to do ambulatory dialysis, that's the manual type of dialysis or the kind where they hook up to a machine. 03:34 The sodium concentration is typically around 132. 03:38 Potassium, oftentimes is zero in my dialysis solution for peritoneal dialysis. 03:42 So I have to be really cognizant of that. 03:45 My patients on PD or peritoneal dialysis oftentimes can become hypokalemic and it's not uncommon to see those patients taking potassium supplements. 03:55 We also have a glucose or dextrose concentration in our peritoneal dialysis solution. 04:00 Now, reason being is that we're actually going to take advantage of the osmotic gradient that can build to cause ultrafiltration. 04:08 We've got a calcium that's typical to the blood calcium, magnesium, and we're going to use lactate as our source of essential base. 04:16 in order to provide the base needed from the acid production that these patients make every day. 04:22 So the mechanism of solute transport is the same, it's diffusion, just as we talked about, and ultrafiltration, depending on how much ultrafiltration we get. 04:30 You get a little bit of convective transport with that as well. 04:34 The mechanism of volume removal is going to be through ultrafiltration, but instead of applying a negative hydrostatic pressure, I actually can use that osmotic gradient that I'm creating by putting a dextrose solution in that peritoneal dialysis solution. 04:49 So if I have a very high dextrose of 4.25% or 2.5%, that's going to stimulate water movement from the blood side to that cavity and with that I will have solvent drag including sodium. 05:03 And that's the way I can remove volume. 05:06 So when we're taking care of these patients, there's a few aspects that are really important to pay attention because it's going to influence the morbidity and mortality of this patient population. 05:15 Number one, when I'm doing these therapies, I have to make sure that I'm actually adequately removing solute. 05:21 So there's mechanisms that we can do that by looking at urea and the clearance of urea, we can see how adequately we're moving solute across those membranes that we're talking about. 05:31 Now remember, urea isn't one of those uremic toxins, but it's something that we can use to look at the transport of how efficaciously we're transporting solute across membranes. 05:42 We also want to look at the adequacy of blood pressure control and in particular, the patient's volume status. 05:49 We really try to maintain a patient's dry weight, that means the weight at which they don't have extra volume on their body. 05:55 So it's critical in order to make sure that you have appropriate ultrafiltration, removal of salt and water, that will absolutely influence the patient's morbidity and mortality. 06:04 We want to manage the patient's anemia. 06:07 Remember, patients who have chronic kidney disease and end-stage renal disease, lose those specialized fibroblasts, which make a erythropoietin. 06:14 That's responsible for generating red blood cells in the bone marrow. 06:18 Without that, they become extremely anemic. 06:20 So most of our patients on dialysis are on Erythrocyte Stimulating Agent (ESA) in order to correct their anemia. 06:28 Our patient also, it's very important to pay attention to their protein nutrition. 06:34 We actually encourage our patients to eat very high protein meals. 06:37 And I want to make a point about this because in our chronic kidney disease lecture, we actually talked about how we want patients to actually restrict protein, that high protein can actually progress their disease. 06:48 But once the patient is on dialysis, they're so catabolic. 06:52 They have protein energy wasting, and it's critical for them to replete that. 06:56 So they must have high protein levels. 06:58 We actually tell patients to eat somewhere around 1.5 grams per kilogram of protein per day. 07:04 So that's quite a bit. 07:06 We want to control their hyperkalemia. 07:08 Again, these patients don't have a mechanism of getting rid of their potassium so it's important to dialyze them against low-potassium bass and ensure that they're eating correctly in low potassium foods. 07:18 And we want to manage the renal bone disease. 07:20 This is things like hyperparathyroidism and vitamin D.
About the Lecture
The lecture Hemodialysis and Peritoneal Dialysis: Procedure by Amy Sussman, MD is from the course Renal Replacement Therapy.
Included Quiz Questions
Which of the following increases the rate of creatinine removal during hemodialysis?
- Increasing blood flow through the dialyzer
- Decreasing the dialysate flow
- Decreasing the size of pores in the semipermeable membrane
- Applying positive hydrostatic pressure on the dialyzer
What is the mechanism by which hemodialysis removes excess fluid from the bloodstream?
- Negative hydrostatic pressure
- Osmotic pressure
- Positive hydrostatic pressure
- Stagnation pressure
What is the purpose of the lactate in the standard peritoneal dialysis solution used in patients with chronic kidney disease?
- It corrects the metabolic acidosis.
- It prevents bone and mineral disorders.
- It corrects hypocalcemia.
- It corrects the nitrogen imbalance.
- It slows the progression of CKD.
Which of the following statements is true regarding peritoneal dialysis?
- Patients on peritoneal dialysis can become hypokalemic.
- It uses negative hydrostatic pressure to improve the efficiency of filtration.
- The high concentration of sodium in the dialysis solution helps with excess fluid removal.
- It is essential for patients on peritoneal dialysis to be on a low-protein diet (less than 1 g/kg/day).
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |