Playlist
Show Playlist
Hide Playlist
Glycolysis: Dhap –> Ga3-P – Glycolysis and Pyruvate Metabolism
-
04 Advanced CarbohydrateMetabolism.pdf
-
Reference List Biochemistry.pdf
-
Download Lecture Overview
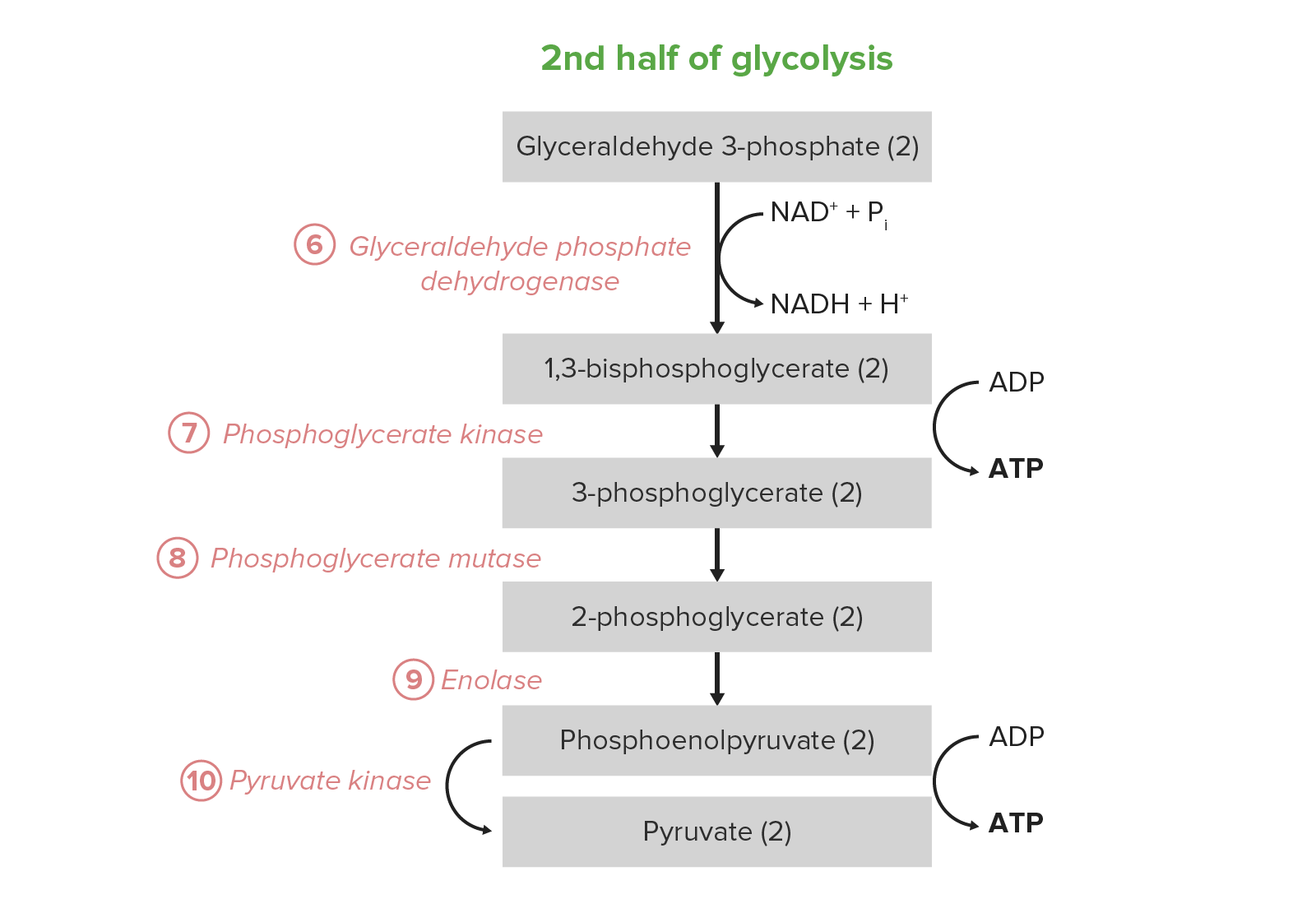
00:00 This is a reaction that students of biochemistry frequently like a lot and the reason that they like this reaction is because dihydroxyacetone phosphate is becoming converted into the other product of the previous reaction glyceraldehyde-3-phosphate. When this happens we then have 2 of every molecule that continues down the pathway of glycolysis. 00:21 This way students don't have to learn one pathway for the dihydroxyacetone phosphate. 00:26 and a different pathway for the glyceraldehyde-3-phosphate. We simply have two glyceraldehyde-3-phosphates to follow. 00:32 The enzyme that catalyzes this reaction is called triose phosphate isomerase and as an example of what we call a perfect enzyme. Now in one of the other lectures that I have given, I have described what perfect enzymes are. But perfect enzymes are enzymes that catalyze reactions amazingly efficiently, amazingly rapidly with amazingly little material. 00:56 The reason of this enzyme does this here is because there is an intermediate its in the process of making the glyceraldehyde-3-phosphate that is extraordinary unstable. 01:08 If that intermediate sits around for any period of time it will fall apart and this reaction will not occur. So to keep that from happening, this enzyme has evolved to be very very rapid in its turnover. 01:20 So this reaction is also one of these readily reversible reactions. It doesn't matter too much to the cell which way it goes and the direction that it goes is gonna be a function of the concentration of the intermediates. 01:31 As you might imagine, this reaction can be reversed in the process of gluconeogenesis. 01:37 So the delta G zero prime is a little positive but it is not excessive for cell to overcome. 01:43 This as I noted is a diffusion limited enzyme and I will remind you that the diffusion limited enzyme is one who's rate of reaction relies only on the rate with which the substrates can diffuse into the active site.
About the Lecture
The lecture Glycolysis: Dhap –> Ga3-P – Glycolysis and Pyruvate Metabolism by Kevin Ahern, PhD is from the course Carbohydrate Metabolism.
Included Quiz Questions
Which of the following is true about Triosephosphate isomerase (TPI)?
- It converts the ketose dihydroxyacetone phosphate to the aldose glyceraldehyde-3-phosphate.
- It catalyzes an irreversible reaction.
- It is the rate limiting enzyme of gluconeogenesis.
- It requires lactate as a cofactor.
- It breaks down Glyceraldehyde-3-phosphate into three phosphates.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |