Playlist
Show Playlist
Hide Playlist
Gastritis
-
Slides GIP Gastritis.pdf
-
Reference List Pathology.pdf
-
Download Lecture Overview
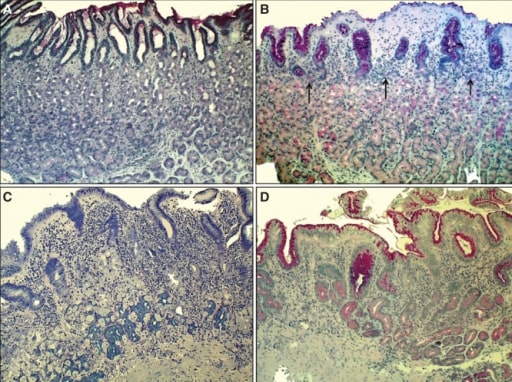
00:01 Welcome. 00:01 In this talk, we're going to cover a very common entity gastritis, inflammation of the gastric mucosa. 00:10 Let's get into epidemiology first. 00:12 As he said, it's a very common entity overall. 00:17 The prevalence is probably higher but approximately 6 per 100,000 population. 00:21 And it's higher in developing countries where there may be a greater infectious agent burden. 00:29 When we talk about infectious etiologies, most commonly we're talking about Helicobacter pylori. 00:35 So aspire keep, and there will be, as I've already indicated, a higher prevalence of this particular infection in developing countries, but also occurs in the pediatric population. 00:46 Another form of gastritis is autoimmune gastritis. 00:50 So an autoimmune disease more commonly associated with women, and it does the incidence does increase with age. 00:58 Let's talk about the pathophysiology. 01:00 There are various ways that one can get gastritis. 01:05 Infection is probably the most common and again, Helicobacter pylori is the most common of the infectious causes. 01:12 That is about the third time when I've said it, so probably is important. 01:15 But there are other other bacterial and viral agents that can do this. 01:19 So mycobacterium avium intracellulare particularly within the immunocompromised population can be a cause of an infectious gastritis. 01:28 Enterococcus, another very common GI bacterium. 01:32 Herpes simplex and cytomegalovirus are viral etiologies for gastritis. 01:38 I've already mentioned autoimmune gastritis, typically in women and in the older population. 01:45 And it's associated specifically with biomarkers that we can measure anti-parietal cell autoantibodies, and anti-intrinsic factor autoantibodies. 01:54 And these will be causes of additional pathologies that we'll cover very shortly. 02:00 There are other less common causes. 02:02 I wouldn't say they're exactly rare, but less common causes of gastritis. 02:06 This includes Crohn's disease, so an inflammatory bowel disorder that, in fact, can affect any part of the GI tract and can cause a gastritis within the stomach. 02:16 Sarcoidosis, certain to the vasculitis, And then other forms less common of gastritis, that are either collagenous gastritis, eosinophilic gastritis, or ischemic gastritis, often caused by certain medications that we'll get into. 02:32 The stomach is just not a uniform bag. 02:35 In fact, there are different areas within it where the esophagus joins, there's the cardia, the very dome of the stomach is the fundus. 02:42 The corpus or the main body of the stomach has a particularly large surface area, and is involved with majority of what the stomach does. 02:52 And then in the antrum, near the pyloric sphincter is a separate area. 02:58 In all of these areas. there's different histology. 03:02 And for the most part, when we talk about gastritis, although it can affect any part of the stomach, we're mostly going to be talking about the antrum. 03:11 So why does this have any impact or why do we care? What I'm showing you here within the main corpus of the stomach is the typical organization. 03:22 It is a simple columnar epithelium that's thrown up into very prominent villi with deep deep pits. 03:33 If we look at this in a different kind of organization, you can see that near the top where there's a lot of kind of cleared out cells, that's where we're going to have mucus, and that's going to be protective to the epithelium throughout the stomach. 03:47 The middle zone, which looks a little bit pinker on the left hand side are going to be the parietal cells. 03:52 Those are going to be the cells responsible for the most part for making the gastric acid, the hydrochloric acid, they're also going to make intrinsic factor. 04:02 And then the chief cells, they're going to be making a zymogen pepsinogen at the base they're going to be making a zymogen pepsinogen at the base that is going to be eventually processed to make pepsin which is going to be part of the digestive enzymes necessary for getting food turned into smaller metabolites that can be absorbed. 04:19 That's up in the main corpus. 04:21 As we look down into the antrum, we have a slight modification of the overall architecture. 04:27 We have many, many more mucus cells. 04:29 Well, this makes sense because we've been accumulating throughout the corpus, all that gastric acidity. 04:34 So we're going to need to protect within the pylorus. 04:35 So we're going to need to protect within the pylorus. 04:37 The cells that are there, the epithelium that's there, so we're going to need to make more mucus. 04:42 In this zone as well, we don't need them have as many parietal cells, but we will have more stem cells that are going to turn into the parietal cells and other elements within the GI epithelium. 04:54 And then deep within that structure, there is gonna be more mucus cells. 04:58 So this is going to be the area where inflammation is going to have a major impact. 05:04 And we're not going to have as much mucus and this is where we're going to get the most damage overall. 05:09 Looking at this histologically, it doesn't have that same kind of light pink and then bluer organization that we saw on the corpus. 05:17 It's more mostly mucus glands. 05:20 And then within the lamina propria around the individual glands, we're gonna see more inflammatory cells. 05:26 And that's going to be very characteristic when we talk about the gastritis. 05:31 So shown here on the right hand side now is a schematic where you can see the various elements, the mucous neck cells at the top, the stem and the preparietal cells in the middle in the deeper mucus cells. 05:43 And in the lamina propria, we've expanded it, and in there are lymphocytes and macrophages and some cases, neutrophils that are going to be part of an inflammatory infiltrate causing damage that appearance where we've dropped out cells. 05:57 We have lost epithelium and that's going to be part and parcel of what goes on with the inflammatory infiltrate. 06:03 So how is this happening, that kind of a global higher level look? Well, you could have increased acid production. 06:11 So you may have more gastrin secretion that is driving increased acidity. 06:18 So the parietal cells up in the corpus are going to be cranking out more hydrochloric acid. 06:23 You may have diminished mucus production. 06:25 There a variety of reasons for this, some of it can be genetic and intrinsic. 06:30 Others can be driven by certain drugs that cause a diminished vascular supply to the GI tract into the stomach. 06:39 You can have direct injury to the epithelial cells or effects on the vascular supply. 06:44 And this is what I was talking about the nonsteroidal anti-inflammatory drugs, although you think they would be anti-inflammatory, they also cause constriction of the micro vasculature. 06:55 So you do not get the normal blood flow to the gastric mucosa. 07:00 And therefore, you're not making the normal levels of mucus, and therefore you're not getting appropriate protection. 07:07 And alcohol. 07:08 An alcoholic gastritis can cause a direct injury to the epithelium that's making the mucus. 07:14 So those cells go, "I give up." And you don't have enough mucus to coat the rest of the stomach. 07:20 In autoimmune disease, you have specifically targeted lymphocytes that recognize typically parietal cells, and they will in turn drive the production of autoantibodies. 07:32 So you have other antibodies and T cells directed against the parietal cells. 07:36 So, you are influencing the production of gastric acid, but you also have some secondary injury due to cytokines and the activation of macrophages that will cause destruction of other parts of the epithelium, in particular, the mucous cells. 07:51 And then infection. 07:53 Back to Helicobacter pylori, our favorite little spirakeet here. 07:58 So it is a flagellated microorganism. 08:00 It really does have kind of a spiral shape, that's kind of cool. 08:03 And it has the ability to spin and to move quite well within the mucous that lines the stomach. 08:12 And it can chemotax to colonize underneath the mucosa. 08:16 So it's a clever little devil. 08:19 It makes a number of things that are going to be particularly toxic to the epithelium. 08:23 So one is urease, this in some cases, in many cases will neutralize the gastric acidity, but it causes direct mucosal injury by increasing the production of ammonia. 08:36 On its surface, it has a number of lipopolysaccharides. 08:39 And those are going to be very activating for the innate immune system in particular macrophages, so that's going to drive the inflammatory response. 08:46 And then, just for the heck of it, it secretes a variety of exotoxins. 08:52 So these have various names. 08:53 I don't think that those are particularly important for you to remember. 08:57 But the vacuolating toxin causes gastric mucosal injury. 09:01 In addition to secretory enzymes, things that break down the mucin. 09:05 Oh, wow. If your mucous layer has been degraded, you don't have as good of protection. 09:09 They make proteases that can cause injury to the epithelial cells. 09:14 They will cause direct gastric mucosal injury through the elaboration of lipases. 09:20 They do have an additional structure that allows them to inject various effectors. 09:26 So a Pilli-like structure. 09:29 So basically a little probe that can sidle up to cells, and inject things directly into them. 09:37 And then as part of the exotoxins and the other effectors, there are others, the cagA complex that will cause actin remodeling within cells so that allows the organism to move around within the cell. 09:53 They will also induce the production of interleukin 8 which is a potent chemotactic agents specifically for neutrophils. 10:00 They can induce the proliferation and also the apoptosis inhibition of the epithelial cells. 10:09 So while they're damaging them and eliciting an inflammatory response, they're also potentially driving proliferation. 10:16 So, long term chronic infection with Helicobacter pylori can also be something that drives the development of malignancy. 10:25 So in acute gastritis is an antral-predominant inflammation. 10:30 So remember I said most of the inflammation is going to be happening down in the antrum. 10:34 Increased gastrin production and diminish the amount of statin production will lead to increased acid secretion, and that increased acid secretion exacerbates mucosal injury and associated inflammation. 10:49 With chronic gastritis, so going on for months and even years, with injury, the glands will become progressively atrophic. 10:58 And there is a response of the epithelium to try to combat to push back on all of the damage that's going on. 11:06 And one of the changes is the conversion of metaplasia to a more intestinal sort of epithelium, not like the normal gastric epithelium, but intestinal, which theoretically can better tolerate the conditions that are there. 11:19 As we injure the parietal cells, we'll have actually a diminished acid production. 11:25 And that hypochlorhydria means that we're not going to absorb iron. 11:29 So we will eventually develop an iron deficiency anemia when we have chronic gastritis. 11:34 As we damage the parietal cells as well, we're not making intrinsic factors much. 11:39 And without intrinsic factor, we can't absorb ultimately B12. 11:43 And so we end up with a B12 deficiency. 11:47 I've already indicated that with now proliferation, diminished apoptosis, ongoing inflammation with all those evil, inflammatory mediators, we're increasing the risk for developing gastric adenocarcinoma. 12:00 And with all that inflammation, we can actually develop an increased risk for a lymphoma, a mucosa-associated lymphoid tissue or MALT lymphoma. 12:12 Clinical presentation for gastritis. 12:14 And you may have already experienced some of this yourself, but it's predictable in many ways. 12:20 So early on, it may be completely asymptomatic, that there may be some dyspepsia of so there may be heartburn, or feelings of reflux. 12:31 There may be abdominal pain with bleeding, because of damage to the epithelium, you may have hematemesis. 12:39 So meaning vomiting blood, or you may have dark, tarry stools, so blood within the stomach. 12:45 By the time it goes through the entire GI tract, you have black, sticky, tarry stools. 12:52 And clearly you're losing blood, so you would develop an iron deficiency anemia. 12:56 But that's compounded by the fact that you are not making enough hydrochloric acid to allow normal iron absorption. 13:05 Because we are injuring the parietal cells, you may also develop the B12 deficiency because you're not making an intrinsic factor. 13:13 And so all of the normal symptoms associated with B12 deficiency, fatigue, inflammation of the tongue and oropharynx, a cognitive decline other neurologic symptoms, all B12 deficiency-associated symptoms may also soon pervane. 13:29 Making the diagnosis then of gastritis. 13:31 So we're going to want to do a complete blood count. 13:35 Clearly, if you have a microcytic anemia, that means you probably have reduced iron, makes sense. 13:40 If you have a macrocytic anemia that suggests that you have a B12 deficiency. 13:44 You want to look in the stool for blood. 13:47 You would definitely want to do endoscopy and biopsy and that would help you to establish the etiology. 13:54 Is it Helicobacter? Is it something else? Is it eosinophilic gastritis, one of those other entities? If you're thinking and the vast majority of these gastroenteritis are going to be due to Helicobacter pylori, you can do certain other tests. 14:11 So there's a urea breath test. 14:12 Remember that the Helicobacter makes urease, which will allow us to break down urea. 14:22 You can actually measure those metabolites in the breath. 14:25 You can do a fecal antigen test looking for Helicobacter pylori antigens within the stool and you can do serologies. 14:33 The serologies would may only indicate you've had a prior Helicobacter pylori infection. 14:39 It may not necessarily mean you have one now, but a positive breath test and/or a positive fecal antigen test means you've got ongoing Helicobacter pylori infection. 14:51 You may look for autoantibodies in that autoimmune gastritis and look for autoantibodies directed against parietal cells or intrinsic factor that will allow you to make that particular diagnosis. 15:01 And you would treat these quite differently. 15:03 With an infection, we're going to treat the infection. 15:06 With autoimmune disease, we're actually going to give immunosuppression. 15:09 So both sides of the coin. 15:12 How do we manage this? Well, it depends on your final diagnosis. 15:16 In general, we want to avoid any additional toxicity. 15:19 So tell your patients to avoid alcohol which causes direct mucosal injury, and will also dissolve the mucin. 15:28 Over the surface, you want to stay away from nonsteroidal anti-inflammatory drugs, which will cause spasm in the microvasculature, so you're not getting good blood supply. 15:36 You would want to get proton pump inhibitors to limit the amount of damage happening due to the gastric acidity, the hydrochloric acid. 15:43 Clearly, you want to replace B12. 15:45 You want to replace iron so that you can treat those particular problems associated with the anemia, but B12 so you don't develop chronic long term neurologic deficits. 15:57 And then, if you make the diagnosis of Helicobacter pylori, you go after it. 16:02 Lot of therapies, we're pretty good at treating this now we can clear it. 16:07 In many areas in developing countries where sanitary conditions are not optimal, it may recur over and over again. 16:15 But the therapies that you want to give are basically proton pump inhibitors to limit gastric acid secretion, and then a combination of various antibiotics. 16:26 The triple therapy are three different antibiotics, a quadruple therapy as in bismuth. 16:31 And it turns out that bismuth containing compounds are bactericidal for the Helicobacter. 16:40 And with that, a very long talk about a very common entity, gastritis.
About the Lecture
The lecture Gastritis by Richard Mitchell, MD, PhD is from the course Disorders of the Stomach.
Included Quiz Questions
What is the most common cause of gastritis?
- Helicobacter pylori
- Autoimmune gastritis
- Epstein-Barr virus
- Human papillomavirus
- Varicella zoster virus
How does Helicobacter pylori harm the gastric mucosa?
- Increased acid production
- Increased mucus production
- Indirect injury to epithelial cells
- Deactivation of cytokines
- Deactivation of macrophages
What product of Helicobacter pylori damages the gastric mucosa?
- Urease
- Ammoniase
- Proteins
- Endotoxins
- Mucousase
What is NOT an effect of chronic gastritis?
- Reduced phosphate
- B12 deficiency
- Cancer
- Intestinal metaplasia
- Reduced iron absorption
What is an early symptom of gastritis?
- Dyspepsia
- Iron deficiency anemia
- B12 deficiency
- Neurologic decline
- Cognitive decline
What is the preferred diagnostic test for gastritis to determine its etiology?
- Endoscopy and biopsy
- CBC
- CMP
- Colonoscopy and biopsy
- Flexible sigmoidoscopy and biopsy
Which of the following is a treatment for gastritis?
- Proton pump inhibitor
- NSAID
- Cholecystectomy
- Increasing alcohol use
- Avoiding bismuth medications
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
Estos videos estan increibles para repasar de una forma que no sea tediosa, ya con las bases y esto para repasar uno mismo puede ir comentando el video junto con el expositor lo cual está excelente