Playlist
Show Playlist
Hide Playlist
G-Protein Coupled Receptors (GPCRs)
-
Slides HormonesSignalTransduction Biochemistry.pdf
-
Reference List Biochemistry.pdf
-
Download Lecture Overview
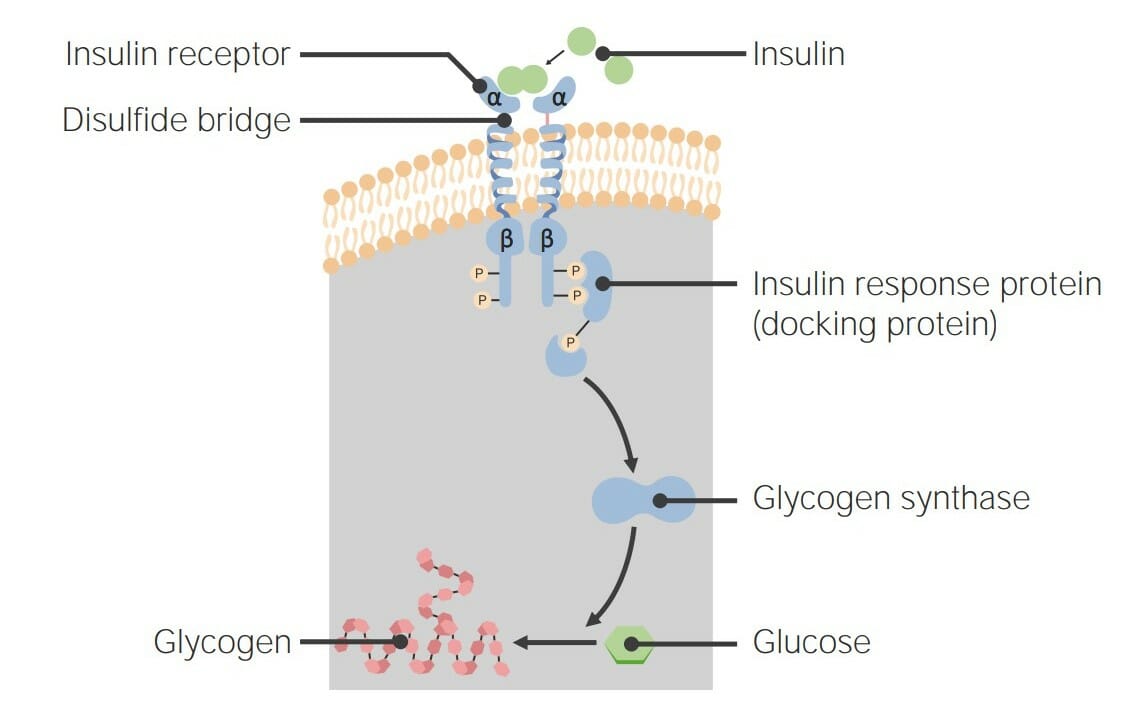
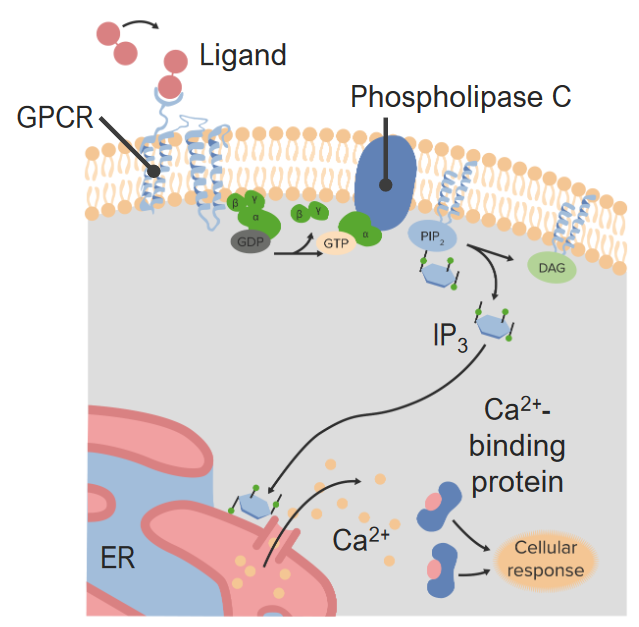
00:01 Bis jetzt habe ich über Rezeptorsysteme gesprochen, die in recht allgemeiner Form funktionieren. 00:07 Schauen wir uns nun ein einzelnes Rezeptorsystem aus der Nähe an, die so genannten G-Protein-gekoppelten Rezeptoren (Englisches Akronym:GPCRs). 00:17 Die GPCRs, G-Protein-gekoppelte Rezeptoren, sind eine sehr häufig vorkommende Klasse von Rezeptorproteinen. 00:24 Es gibt fast 800 Gene im menschlichen Genom, die spezifisch in der Form von G-Protein- gekoppelten Rezeptorsequenzen vorliegen. Erstaunlicherweise sind fast 460 davon olfaktorisch, das heißt, dass sie am Prozess des Riechens beteiligt sind. Der hier abgebildete GPCR ist also eingebettet in eine Lipid-Doppelschicht. Und wir haben Farbe genutzt, um die verschiedenen Regionen des Proteins, die durch die Lipiddoppelschicht ragen, zu kennzeichnen. Obwohl die Doppelschicht natürlich in grau dargestellt ist. Das darin eingebettete Protein hat unterschiedliche Farben von Blau, Grün, Gelb und Rot. Wir sehen Zahlen, die mit diesen verbunden sind und wir sehen verschiedene Farben, die mit diesen verbunden sind. Jede Farbänderung steht also für einen anderen Teil des Proteins, der entweder von unten nach oben oder von oben nach unten, oder von unten nach oben und von oben nach unten, usw. geht. Dieses Protein durchquert die Lipiddoppelschicht, indem es sich siebenmal auf und ab und auf und ab bewegt. Diese sind als sieben Transmembrandomänen bekannt und daher kommt auch der Name 7TM. Wir können sehen, dass das Protein zwei verschiedene Endungen hat. Eine Endung an der Spitze, die ein NH3+ enthält, das ist das Amin-Ende des Proteins. Und ein Carboxyl-Ende des Proteins in rot am unteren Rand. Diese Ausrichtung von Protein und Lipid- Doppelschicht ist spezifisch für jedes einzelne Protein. Nun, der β-adrenerge Rezeptor über den ich hier sprechen möchte ist ein Beispiel für eine der sieben TMs und beteiligt an der Steuerung des Glykogen-Stoffwechsel in der Zelle. GPCRs haben also ihren Namen von der Tatsache, dass sie G-Protein-gekoppelte Rezeptoren sind. Es ist also angebracht, dass ich ein wenig darüber sagen sollte, was G-Proteine sind und wie sie funktionieren. G-Proteine sind kleine Proteine, die an Guanin Nukleotide binden, das ist der G-Teil ihres Namens. 02:08 Die beiden verschiedenen Nukleotide, die sie binden können, sind bekannt als GDP oder Guanosin-Diphosphat oder Guanosin-Triphosphat, GTP. 02:17 Wir sehen in dem Beispiel auf dem Bildschirm, dass das G-Protein sich als drei Einheiten darstellt - eine Alpha (α)-Einheit, eine Beta (β)-Einheit und eine Gamma (γ)-Einheit. 02:26 Dieses Bild zeigt ein sogenanntes heterotrimeres G-Protein. 02:30 Und alle G-Proteine sind im Wesentlichen heterotrimer, das heißt, sie haben drei verschiedene Untereinheiten, die in ihrer Gesamtstruktur nicht identisch sind; dies hat α, β und γ. 02:41 Diese Proteine assoziieren mit GPCRs. 02:44 Das G-Protein assoziiert also mit diesem membrangebundenen Protein. 02:49 Und sie assoziieren mit ihm im inneren Teil der Zelle. 02:52 Denken Sie daran, dass der äußere Teil der Zelle der Ort ist, an dem sich das Hormon bindet und der innere Teil der Zelle der Ort, an dem sich das G-Protein befindet. 03:00 Die G-Proteine werden tatsächlich durch die Bindung des Hormons an die GPCRs verändert. 03:06 Sie erinnern sich an die Folie, die ich Ihnen gezeigt habe, dass die Bindung des Hormons die Form des GPCRs im Inneren der Zelle verändert. 03:14 Die Veränderung der Form des GPCR führt dazu, dass das G-Protein, das mit ihm verbunden ist, sich verändert. 03:21 Und das sehen wir auf dieser Folie hier. 03:24 Das G-Protein ist also assoziiert, wie wir sehen können und das ist das, was wir den Ruhezustand nennen. 03:28 Das Hormon hat nicht gebunden. 03:31 Epinephrin kommt in diesem Fall und bindet an den G-Protein-gekoppelten Rezeptor. 03:35 Und wenn es das tut, verändert es tatsächlich die Form des G-Protein-gekoppelten Rezeptors. 03:39 Und ich habe das hier etwas übertrieben, indem ich ein Fünfeck verwendet habe. 03:43 Die Veränderung der Form des GPCR verursacht die Wechselwirkung zwischen der α Untereinheit des G-Proteins und der β und γ-Untereinheiten, die sich verändern. 03:54 Die β- und γ-Untereinheiten werden freigesetzt und zusätzlich ersetzt die α-Untereinheit das GDP, das sich in ihr befand, durch ein GTP. 04:03 Das erleichtert nun die weitere Freisetzung der γ- und β-Untereinheiten. 04:08 Nun, eine der Funktionen der γ-Untereinheit besteht darin, der α-Untereinheit zu helfen, sich mit dem GPCR zu verbinden. 04:15 Wenn die γ-Untereinheit verschwunden ist, kann die α-Untereinheit nun auch gehen und sie transportiert GTP. 04:23 Nun, GTP ist ein Aktivierungssignal, es bedeutet, dass diese einzelne α-Untereinheit Informationen von außerhalb der Zelle erhalten hat, die durch den GPCR übermittelt wurden und diese α-Untereinheit wird dann mit anderen Proteinen interagieren, ihr Verhalten ändern und das Signal innerhalb der Zelle weitergeben. 04:47 Wir sehen, dass das hier passiert. 04:49 Was nun passieren wird ist, dass die α-Untereinheit mit einem anderen Protein interagieren wird und dieses andere Protein dazu veranlassen wird, einen zweiten Botenstoff zu erzeugen. 04:57 Hier ist also unsere aktivierte α-Untereinheit mit einem daran gebundenen GTP. 05:02 Das Protein, mit dem die α-Untereinheit interagiert, ist ein anderes Membranprotein namens Adenylatzyklase. 05:09 Wenn die α-Untereinheit mit dem GTP an die Adenylatcyclase bindet, wird auch die Adenylatcyclase verändert. 05:16 Was ist nun die Veränderung? Nun, die blaue Darstellung der Adenylatcyclase ist die inaktive Form des Enzyms, das heißt, es katalysiert keine Reaktion. 05:25 Aber wenn sich das Aussehen verändert hat, wie wir hier sehen, bedeutet das, dass sich die Aktivität des Enzyms verändert hat. 05:33 Die Veränderung der Aktivität bei der Bindung der α-Untereinheit verursacht die Produktion eines Moleküls, das als zyklisches AMP bekannt ist. 05:41 Und wir können die Reaktion sehen, die hier auf dem Bildschirm dargestellt ist. 05:44 ATP wird umgewandelt in zyklisches AMP. 05:47 Und zyklisches AMP, wie Sie sich vielleicht von einer früheren Folie erinnern, ist ein zweiter Botenstoff. 05:51 Dieses kleine Molekül wird nun zu einem anderen Teil der Zelle gelangen und dort die zu beobachtende Wirkung hervorrufen.
About the Lecture
The lecture G-Protein Coupled Receptors (GPCRs) by Kevin Ahern, PhD is from the course Hormones and Signal Transduction. It contains the following chapters:
- G-Protein Coupled Receptors
- GPCRs and G-Proteins
- β-adrenergic Receptor Signaling – Creation of the Second Messenger
Included Quiz Questions
Which statement regarding G-protein coupled receptors (GPCRs) is true?
- They have 7 transmembrane domains.
- They bind hormones with their intracellular domain.
- They are rare in the human genome.
- They bind to ATP molecules.
What does the binding of epinephrine by a G-protein coupled receptor (GPCR) do?
- Results in the production of cAMP by adenylate cyclase.
- Causes the α-subunit of a G-protein to become activated with GDP.
- Stimulates the phosphatase activity of protein kinase A.
- Decreases the levels of cAMP.
The beta-adrenergic receptors are classified as which of the following?
- G-protein coupled receptors
- Intracellular receptors
- Second messengers
- Nuclear messengers
- Cytoplasmic messengers
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
The video helped me understanding this topic with visual representation