Playlist
Show Playlist
Hide Playlist
Fate of Pyruvate – Glycolysis and Pyruvate Metabolism
-
04 Advanced CarbohydrateMetabolism.pdf
-
Reference List Biochemistry.pdf
-
Download Lecture Overview
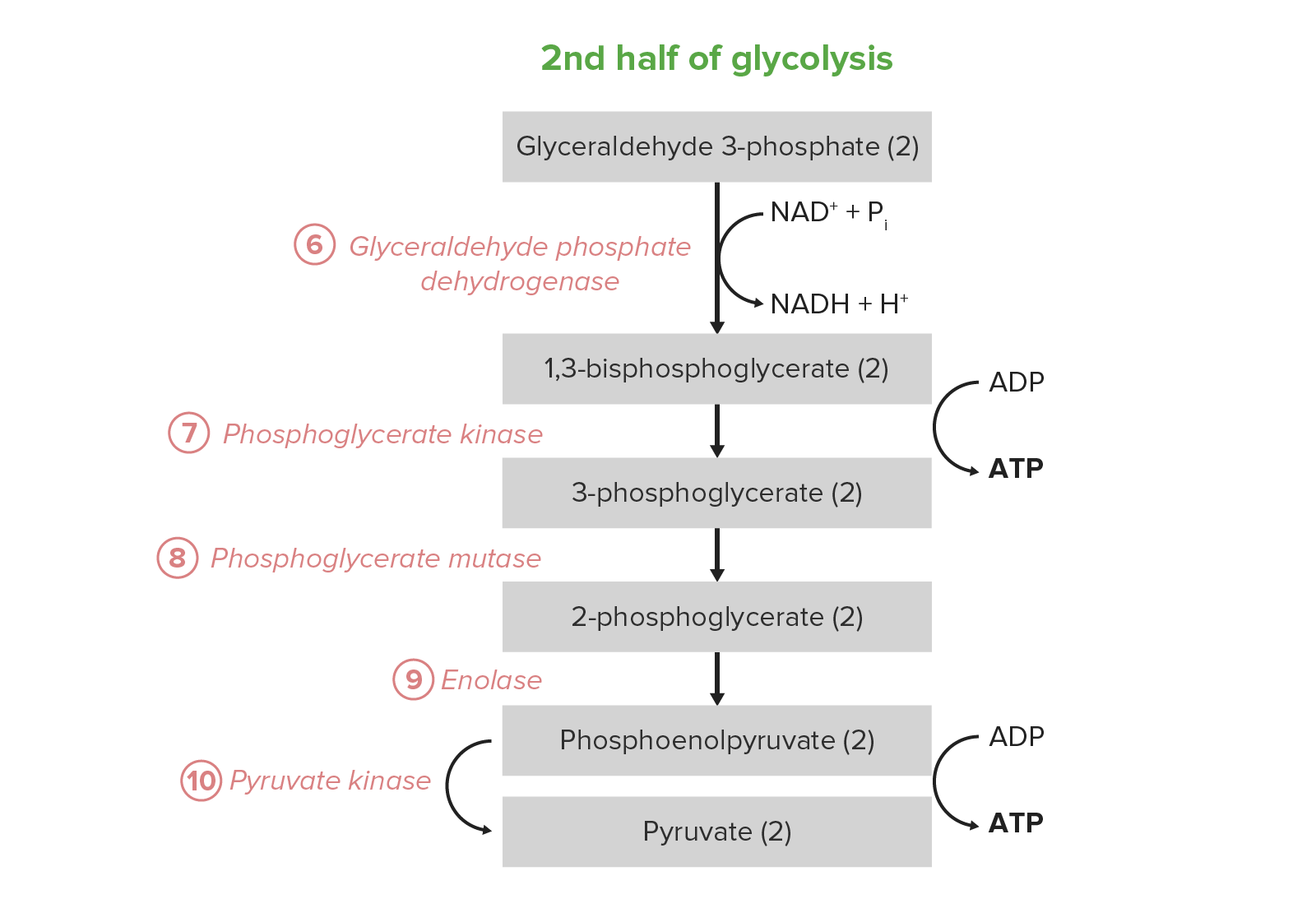
00:00 Das letzte, was ich hier betrachten möchte ist das Schicksal vom Pyruvat. 00:04 Pyruvat ist das Endprodukt dieses Weges, aber wir erinnern uns daran, dass Stoffwechselwege nicht isoliert ablaufen. 00:11 Pyruvat wechselt in andere Wege und kann so zu einer Vielzahl von Produkten weiterverarbeitet werden. 00:14 Wohin es wechselt, hängt von den Begebenheiten in der Zelle ab, in der es hergestellt wurde. 00:21 Schauen wir also ein wenig genauer hin, um zu sehen, wie es mit diesem Pfad weiter geht. 00:25 Wie gesagt, ist es kein Endprodukt und wichtig für eine Vielzahl von Prozessen: Es ist wichtig für die Herstellung von Alanin. 00:30 Es ist wichtig für die Energiegewinnung und eine wichtige Quelle, um die Glykolyse in Gang zu halten, wie wir in Kürze sehen werden. 00:38 Und jetzt? Betrachten wir nun die drei verschiedenen Dinge, die hier zu sehen sind, in die Pyruvat umgewandelt werden kann. 00:45 Pyruvat kann in Bakterien- und Hefezellen in ein Molekül namens Acetaldehyd umgewandelt werden. 00:52 Acetaldehyd kann dann in Ethanol umgewandelt werden. 00:57 Biertrinker mögen diese Reaktion natürlich, denn diese Reaktion ist als alkoholische Gärung bekannt und auf diese Weise wird gebraut. Wir stellen Bier, Wein und weiteres mit dieser Reaktion her. 01:07 Wir fördern diese Reaktion durch den Verschluss des Gefäßes, in dem die Reaktion abläuft, denn die Sauerstoffarmut in diesem Gefäß begünstigt die Produktion von Ethanol. 01:21 Jetzt hättest du wahrscheinlich nicht gedacht, dass deine menschlichen Zellen auch fermentieren. 01:26 Nun funktioniert die Gärung bei uns anders, als in Hefen und Bakterien, sonst müssten ja wir kein Bier kaufen. Richtig? Um Pyruvat in Laktat umwandeln. Es gibt einen Grund, warum wir das tun, wie wir noch sehen werden. 01:39 Wenn unseren Zellen der Sauerstoff ausgeht, bilden sie Laktat, auch bekannt als Milchsäure. 01:44 Sie ist wichtig für die Produktion von NAD, wie wir sehen werden. 01:52 Du siehst, dass bei der Reaktion von Acetaldehyd zu Ethanol auch NAD gebildet wurde. 01:57 Wie sieht es nun unter aeroben Bedingungen aus? Unter aeroben Bedingungen wird Acetyl-CoA dem Citratzyklus für die Oxidation zugeführt. 02:04 Oxidation erfordert Sauerstoff und daher ergibt dies Sinn. 02:09 Bei Sauerstoffmangel haben wir alternative Wege, bei genügend Sauerstoff, nutzen wir ihn, so gut wir können. 02:15 Nun ist der Unterschied enorm, zwischen der Oxidation mit Hilfe des Citratzyklus und der Oxidation über den Fermentationsweg. 02:27 Wenn wir mit Glukose beginnen und den Fermentationsweg durchlaufen, ist das Nettoprodukt dieser Reaktion 2 ATPs. 02:34 Wenn wir mit Glukose beginnen und sie im Citratzyklus vollständig oxidieren, erhalten wir 38 ATPs. 02:42 Das ist 19-facher Energiegewinn unter aeroben Bedingungen als bei Sauerstoffmangel. 02:49 Man sieht also, warum die Zellen gerne Sauerstoff bekommen, und du kannst sehen, warum unser Körper aufgebaut ist, um so viel Sauerstoff wie möglich liefern zu können. 02:56 Das gelingt ihm nicht immer, aber er tut so viel, wie er kann. 03:01 Okay, nun kommen wir dazu, warum diese Gärung überhaupt notwendig ist. 03:05 Es ist dir vielleicht während meiner Vorlesung nicht besonders aufgefallen, aber ich habe angemerkt, dass Glyceraldehyd-3-phosphat in 1,3-Bisphosphoglycerat durch die einzige Oxidationsreaktion der Glykolyse umgewandelt wird. HIER Diese Reaktion ist bedeutend, weil jede Oxidationsreaktion einen Elektronenträger benötigt und der hier verwendete Elektronenträger NAD+ ist, das zu NADH wird. 03:27 Die Zelle hat keine unbegrenzte Menge an NAD. 03:31 Wenn NAD in NADH umwandelt wird, muss es eine Möglichkeit geben, NADH wieder in NAD umzuwandeln. 03:39 Wie geht das? Wie kann eine Zelle das schaffen? Normalerweise gelingt der Zelle das in einem Prozess, der Sauerstoff benötigt. 03:46 Diesen bezeichnet man als die Elektronentransportkette. Und, was benötigt die Elektronentransportkette? Sauerstoff. 03:52 Wenn also kein Sauerstoff vorhanden ist, kann die Elektronentransportkette nicht ablaufen. 03:56 Und was passiert mit der NADH-Konzentration? Sie wird ansteigen und die NAD+-Konzentration sinken. 04:04 Unter diesen Bedingungen kann die Glykolyse nicht weiterlaufen. 04:09 Nun stell dir vor, du bist joggen und dein Blutkreislauf liefert den Sauerstoff nicht so schnell, wie du ihn brauchst. 04:15 Was passiert dann in diesem Fall? In diesem Fall stoppt die Glykolyse, und damit die einzige Reaktion, welche die Muskelzellen mit Energie versorgt. 04:26 Dann hast du ein Problem. Glücklicherweise haben Muskelzellen, Hefezellen, und andere Zellen Möglichkeiten, dieses NAD+ zu regenerieren und zwar die Fermentation. 04:35 Wir können hier sehen, dass das Endprodukt der Glykolyse Pyruvat ist. 04:39 Pyruvat wird also in Prokaryonten in Acetaldehyd umgewandelt, wie ich schon sagte, und Acetaldehyd dann in Ethanol umgewandelt. Deshalb produzieren Hefezellen Ethanol. 04:50 Bei der Ethanolreaktion wird NAD+ produziert und rate mal, wo es verwendet wird: Wieder in der Glykolyse-Reaktion. Durch den Wechsel zwischen diesen beiden Reaktionen, wird NAD+ erzeugt, die Glykolyse wird aufrechterhalten und die Zelle bleibt am Leben. 05:06 Nun wandeln tierische Zellen kein Pyruvat in Acetaldehyd um. 05:11 Stattdessen wandeln sie Pyruvat in Laktat, auch Milchsäure genannt, um. Du kannst hier sehen, dass diese Reaktion durch die Laktatdehydrogenase katalysiert wird und das Gleiche passiert wie in der Hefe. 05:20 Sie nimmt Elektronen von NADH und überträgt sie auf Pyruvat, um Laktat herzustellen. 05:27 So wird NAD+ regeneriert, und geht direkt zurück in die Glykolyse und hält sie am Laufen. 05:32 Wir fermentieren also aus denselben Gründen, wie es Hefen und Bakterien tun, aber wir bilden nicht das gleiche Endprodukt. 05:39 Ich hoffe, dass ich dir in dieser Reihe von Vorträgen einen guten Überblick geben konnte, über den Weg der Glykolyse, den Energiebedarf der Zelle und welchen Einfluss die Energie auf die Art und Weise, in der Reaktionen ablaufen und die Steuerungselemente, die es den Zellen ermöglichen, diesen Prozess effizient ablaufen zu lassen.
About the Lecture
The lecture Fate of Pyruvate – Glycolysis and Pyruvate Metabolism by Kevin Ahern, PhD is from the course Carbohydrate Metabolism.
Included Quiz Questions
Which of the following is true of pyruvate?
- It is made into lactate during fermentation.
- It is converted into acetyl-CoA when oxygen is limited.
- It uses NADH to produce acetaldehyde.
- It is an intermediate in the citric acid cycle.
Which of the following statements about fermentation is incorrect?
- It requires acetaldehyde dehydrogenase.
- It occurs when cells are short of oxygen.
- It occurs when cells are short of NAD.
- It has different products in bacteria and humans.
Under aerobic conditions, what does the pyruvate get converted into?
- Acetyl CoA
- 2,3-bisphosphoglycerate
- Lactate
- Acetaldehyde
- Ethanol
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
A+ teaching style and delivery thank you Dr Ahern for making a difficult topic easier to digest