Playlist
Show Playlist
Hide Playlist
Drug Metabolism – Biotransformation | Pharmacokinetics (PK)
-
Slides Drug Metabolism Biotransformation PK.pdf
-
Reference List Pharmacology.pdf
-
Download Lecture Overview
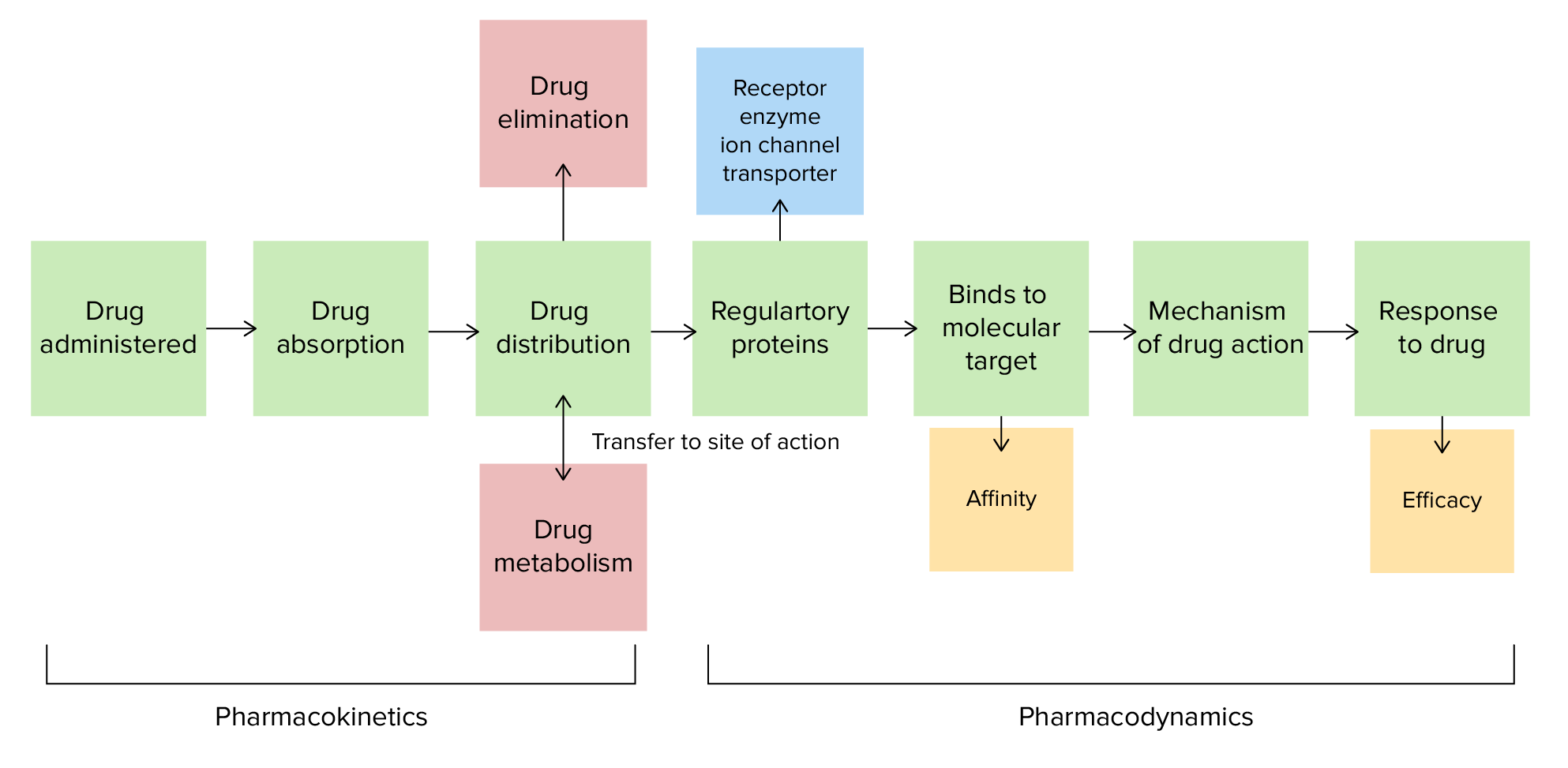
00:01 Welcome to Pharmacology by Lecturio. 00:04 Today's lecture is about biotransformation. 00:07 Biotransformation is how the human body changes drugs into different molecules to either make them more useful or less useful to the human body. 00:16 Now, why do humans metabolize drugs? In order to understand that let's go back in time. 00:21 Let's go back to the age of the cavemen. 00:24 So you can see here I have a bunch of pictures that look remarkably like hospital administrators. 00:30 And you can see that as they are evolving they were eating different types of food. 00:35 It depended on where in the world they were. 00:37 As we continue to evolve, we started to eat different types of foods. 00:41 Some of the foods weren?t originally meant for our biological system and there were toxins in some of these plants. 00:48 As we went through the ages, we adapted to those new plants and as we spread out across the planet, we learn to eat different types of food, different types of toxins entered into our body and our body is adapted to metabolize these toxins. 01:04 These toxins came from plants and when you think about it, plants were the origin of 80% of drugs prior to the development of synthetic drugs. 01:12 Now, how is it that we manage to break down drugs? It's the magic of the cytochrome P450 system. 01:19 So the cytochrome molecule is an incredibly diverse and incredibly complicated molecule. 01:25 It is actually a group of isoenzymes that are clumped together and they're coded by 57 different genes. 01:32 Now, in humans there's widespread cytochrome activity. 01:37 They are found in all of the tissues, the liver, the brain. 01:40 The only places where you don't see cytochrome is in the bones, muscles and in the red blood cells. 01:45 Xenobiotics which are those toxins in the plant that I was talking to you about earlier are present in the diet, the environment and in the drugs that we prescribe. 01:56 Now, cytochrome, are mixed function oxidases. 02:00 75% of all of the drugs that we buy or ingest are metabolized by this system. 02:07 There are many isoenzymes. 02:09 One of the most common is cytochrome P450-3A4 and -3A5. 02:15 We group them together for simplicity sake. 02:18 There's also another one called -2D6. 02:21 There is a whole bunch of them in fact. 02:22 And when we take a look at the different types of isoenzymes we see that they will metabolize different drugs. 02:28 And different people from different regions of the world, will have more active or less active isoenzymes depending on their climate and the biosphere that they inhabited. 02:39 Now, cytochrome is found in two main cellular districts in the liver. 02:45 They're found in the inner membrane of the endoplasmic reticulum and they're found in mitochondria. 02:50 Let's talk about the different types of reactions that occur within the human body. 02:55 We have three phases of metabolism that make a drug become active or inactive in the body. 03:02 Phase 1 reactions are mediated by the cytochrome P450 system. 03:08 So what the cytochrome system does is it takes a parent drug and makes it more polar as a daughter drug. 03:16 It either unmasks a polar group that's already within the molecule or it inserts a polar group on to the molecule. 03:24 The whole purpose behind the cytochrome system is to make the drug more soluble in water so that it can be inside the cell which is of course cytoplasm is water. 03:35 Phase 2 reactions are conjugation reactions that occur in the cytoplasm. 03:40 So they add to the -OH, -SH, and -NH2 sites that were added by the cytochrome system. 03:48 Once again, it makes the drug even more soluble in water so it makes the drug hydrophilic. 03:54 It does so by adding one of these six groups. 03:58 It can be a glutathione group, an acetate group or a methyl group. 04:02 It doesn’t really matter which group it adds. 04:04 The point being that these phase 2 reactions are important to drug metabolism because it makes it more soluble in water. 04:10 Even more so than the cytochrome system did. 04:14 Phase 3 reaction is a further processing of drugs. 04:18 Not all drugs go through phase 3. 04:20 For preparation of drug to excrete, we need phase 3 in many of our drugs. 04:26 So sometimes we're excreting drugs into the bile. 04:29 Sometimes we're excreting it into the urine or other lumens. 04:32 It may even be excreted into the bowel. 04:35 We often use transport proteins to do this. 04:39 These transport proteins are usually P-glycol-protein. 04:43 The gene that codes them are MDR-1. 04:46 So when you start hearing about P-glycol-protein or MDR-1, we're talking about the same thing. 04:53 It just depends on if you are talking about the gene or if you are talking about the actual protein molecule. 04:58 Let's take another look at the cell membrane. 05:00 Here we have the glycoprotein. 05:03 Sometimes those are P-glycoproteins that we mentioned earlier. 05:07 Sometimes they are glycolipids. Sometimes we have protein channels. 05:10 The important thing that we wanna know is that the P-glycoprotein is often sticking out the surface of the cell and it sometimes will help in moving a drug from one cell surface through it to the other cell surface. 05:24 Okay, let's do a question on biotransformation. 05:29 Hepatic metabolism of most drugs results in the following. 05:34 A. The daughter drug is more lipophilic than the parent drug. 05:39 B. The daughter drug is less lipophilic than the parent drug. 05:44 C. The daughter drug is more hydrophobic than the parent drug. 05:49 D. Glucuronidation through a phase 1 process at cytochrome P450 or E. Increased volume of distribution. 06:01 So the answer is B. The daughter drug is less lipophilic than the parent drug. 06:08 So this is less lipophilic, so that means it's more hydrophilic. 06:14 And remember we wanna add polar drugs so that the drug is more dissolvable in water. 06:20 So the right answer is answer B. 06:23 Let's just quickly go down to answer number D. 06:25 That is not correct because glucuronidation is a phase 2 process and it does not occur through Cytochrome P450. 06:33 And let's look at answer number E. That's not correct. 06:37 Increased volume of distribution, that makes no sense at all. 06:40 So we will just discard that answer as one of the possibilities.
About the Lecture
The lecture Drug Metabolism – Biotransformation | Pharmacokinetics (PK) by Pravin Shukle, MD is from the course Pharmacokinetics and Pharmacodynamics. It contains the following chapters:
- Metabolism of Drugs
- Cytochrome P450
- 3 Phases of Metabolism
- Question: Biotransformation
Included Quiz Questions
A parent drug is primarily polarized by the cytochrome P450 system during which phase(s) of drug metabolism?
- Phase 1
- Phase 2
- Phase 3
- Phases 2 and 3
- All phases
Conjugation primarily refers to which phase of drug metabolism?
- Phase 2
- Phase 1
- Phase 3
- Phases 1 and 2
- All phases
Which cells lack cytochrome P450 enzymes?
- Red blood cells
- Liver cells
- Brain cells
- Kidney cells
- White blood cells
In which phase of metabolism do some drugs undergo further processing in preparation for excretion into bile?
- Phase 3
- Phase 1
- Phase 2
- Phases 1 and 2
- Phases 2 and 3
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
4 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
Lecturio is Awesome! I am not a student but all the material I have been through so far is of HIGH quality! My best investment ever!
Best pharmacology classes! Love the questions in between the lecture!
2 customer reviews without text
2 user review without text