Playlist
Show Playlist
Hide Playlist
Diffusion – Biological Membranes
-
10 Basic BiologicalMembranes.pdf
-
Biochemistry Free and Easy.pdf
-
Reference List Biochemistry.pdf
-
Download Lecture Overview
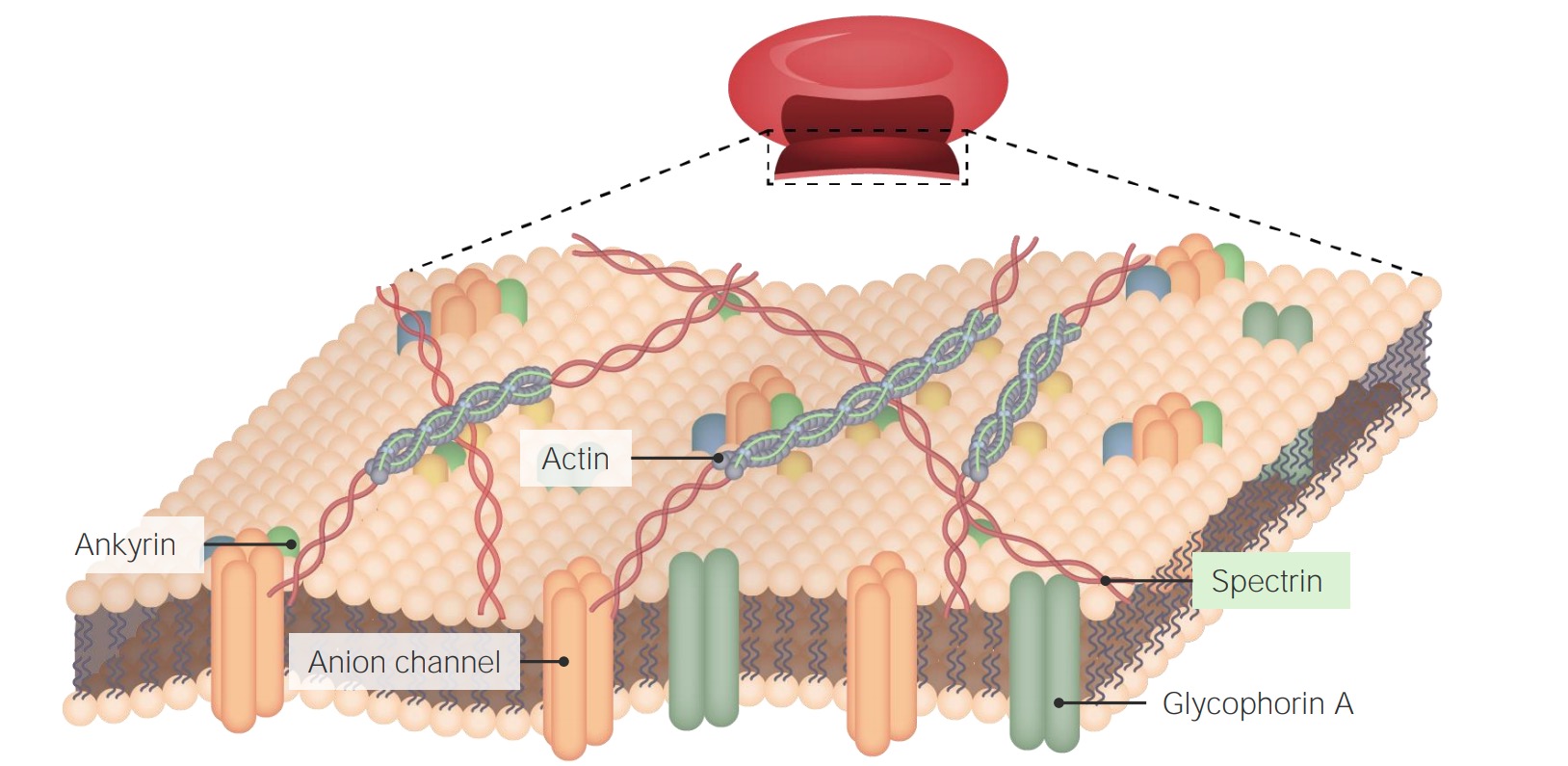
00:01 Eines der Dinge, die man bei einer Lipiddoppelschicht bedenken sollte, ist, wie ich bereits erwähnt habe, dass eine Lipiddoppelschicht ziemlich undurchlässig ist. 00:06 Wir können aber tatsächlich die Permeabilität von Ionen durch eine semipermeable Membran messen und das ist das, was wir hier sehen. Membranen sind nicht absolut undurchlässig, aber sie sind für viele Dinge ziemlich undurchlässig. Wenn wir über Durchlässigkeit sprechen und darüber nachdenken, müssen wir einige Überlegungen über die Tendenzen von Molekülen anstellen. Moleküle in Lösung werden dazu neigen, sich von einer hohen Konzentration zu einer niedrigen Konzentration zu bewegen. 00:31 Wenn ich eine Situation habe, wie ich sie hier dargestellt habe, mit den Kammern auf der linken Seite, kann man sehen, dass die Kammern auf der linken Seite durch eine halbdurchlässige (semipermeable) Membran in zwei Hälften geteilt sind. 00:41 In der linken Kammer des Gefäßes auf der linken Seite sehen wir eine hohe Konzentration von Molekülen und auf der rechten Seite sehen wir eine niedrigere Konzentration. Die Moleküle können sich langsam durch diese Membran bewegen und wenn wir den Dingen genügend Zeit geben, wird es dazu kommen, dass sich die beiden Konzentrationen angleichen, d.h., dass die hohe Konzentration sich zur niedrigen verschieben wird. 01:04 Die Diffusionsgeschwindigkeit oder die Geschwindigkeit der Bewegung eines Moleküls von links nach rechts zum Beispiel hängt mit mehreren Dingen zusammen. Zum einen hängt sie mit den Konzentrations- unterschieden zwischen den beiden zusammen. Je größer der Konzentrationsunterschied, desto wahrscheinlicher und desto einfacher ist es für ein Molekül, sich von links nach rechts zu bewegen. Der zweite Faktor ist die Ladung. 01:26 Die Ladung ist etwas schwieriger vorstellbar, aber man kann sich vorstellen, dass, wenn etwas einen großen Ladungsunterschied hat, sich tatsächlich ein elektrisches Potential über der Membran aufbaut und dieses elektrische Potential einen Prozess in Gang setzen kann. Das ist genau das, was passiert, wenn man einen Ladungsunterschied über der Membran hat. Der dritte Faktor ist natürlich die Durchlässigkeit der der Membran. Je durchlässiger eine Membran ist, desto einfacher und desto schneller kann dieser Prozess ablaufen. Je weniger durchlässig die Membran ist, desto unwahrscheinlicher und desto langsamer wird dieser Prozess ablaufen. 02:01 Nun, solche Membranen, die schlecht durchlässig sind, können, wie gesagt, eine Barriere bilden, werden aber die Bewegung der Moleküle über sie hinweg nicht völlig aufhalten. 02:10 Die erste Bewegung durch Membranen, über die ich sprechen möchte, wird durch dieses Beispiel veranschaulicht. 02:17 Diese Bewegung durch die Membranen findet in einem Prozess statt, den wir erleichterte Diffusion nennen. Wie der Name schon sagt, ist die Diffusion die Bewegung von Molekülen von einer hohen Konzentration zu einer niedrigen Konzentration. Dies gechieht, um sich gegenseitig auszugleichen, wie wir in der vorherigen Abbildung gesehen haben. 02:37 Erleichtert bedeutet, dass etwas dabei hilft diese Bewegung zu vollziehen. Nun, da die Lipid- doppelschicht ziemlich undurchlässig ist, erlaubt es den Moleküle kaum auf eigene Faust durch die Membran zu gelangen. Wenn die Zelle das Bedürfnis hat, diese Moleküle durch die Membran zu befördern, muss die Zelle typischerweise ein Protein haben, das in die Membran eingebettet ist und das dies ermöglicht. 02:58 Nun gibt es zwei typische Arten von Proteinen, die dies in diesem Prozess ermöglichen. 03:04 Wenn ich nun betone, dass ich von Diffusion -und ich sage erleichterte Diffusion- rede, dann meine ich damit einen Prozess, der keine Energiezufuhr erfordert, weil der Prozess natürlich ist und die Moleküle sich normalerweise von einer hohen Konzentration zu einer niedrigen Konzentration bewegen. 03:22 Die Proteine, die ich beschreiben werde, ermöglichen diesen Prozess einfach. 03:28 Wie Sie hier sehen können, gibt es zwei Arten von Proteinen. Das eine ist ein Carrierprotein (Transportprotein), das in der gelblichen Farbe dargestellt wird. Das Transportprotein hat, wie Sie sehen können, eine Kammer, die es ermöglicht, die gelben Moleküle zu binden. Dann verändert der Transporter seine Form, damit sich die Moleküle hinüberbewegen können. Diese Veränderung, die ich mit meinen Händen veranschauliche, diese Veränderung, die auftreten kann, erfordert keine Energiezufuhr und wird einfach durch die Bewegung des Moleküls von hoher Konzentration zu niedriger angetrieben. Wenn dies über längere Zeit geschieht, werden sich die Konzentrationen der beiden ausgleichen. Ein gutes Beispiel dafür ist in unseren Blutzellen. Hier haben wir GLUT. Ich erwähnte die GLUTs bereits vorher. Sie ermöglichen es der Glukose durch die Membran der Blutzelle zu gelangen, entsprechend dem Konzentrationsunterschied. Blutzellen haben also eine niedrigere Konzentration an Glukose als die umgebende Flüssigkeit um sie herum. Mit den GLUTs bewegt sich die Glukose frei in diese Zellen und es erfordert keinen Energieaufwand die Glukose aufzunehmen. 04:31 Ein weiterer Proteintyp, der an diesem Austausch beteiligt ist, ist ein Ionenkanal. Ich werde später in dieser Vorlesung mehr über Ionenkanäle erzählen. Ionenkanäle sind typischerweise spezifisch für Ionen, also geladene Moleküle, wie Natrium oder Kalium. Das Bemerkenswerte an den Ionenkanälen ist, dass sie sehr spezifisch sind, typischerweise für ein Ion und andere Ionen zurückweisen.
About the Lecture
The lecture Diffusion – Biological Membranes by Kevin Ahern, PhD is from the course Biochemistry: Basics.
Included Quiz Questions
Which of the following is NOT true about the movement of materials across a membrane?
- It always requires a transport protein.
- It is affected by the concentration of materials on either side of the membrane.
- It requires an energy source to move from lower to higher concentrations.
- Regulation/control of it is essential for cells.
- The charge difference has an effect on movement across the lipid bilayer.
Which of the following is true of facilitated diffusion?
- It requires only the energy of diffusion.
- It uses ATP or GTP.
- It does not require a protein.
- It requires antiports.
- It moves ions from lower concentrations to higher concentrations.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |