Playlist
Show Playlist
Hide Playlist
Colorectal Cancer
-
Slides GIP Colorectal Cancer.pdf
-
Reference List Pathology.pdf
-
Download Lecture Overview
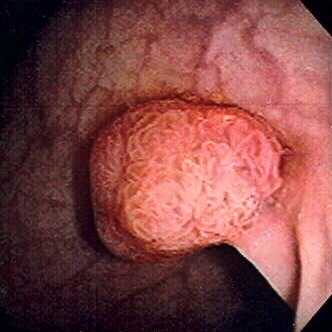
00:01 Welcome. In this talk, we're going to cover colorectal cancer. 00:04 A very important cause of malignancy in the total human population and a major cause of cancer-related deaths. 00:12 So, a little bit first about the epidemiology. It is the third most common cancer among both men and women. 00:19 It is the second most common cause of cancer-associated deaths in the United States. 00:24 The majority of these tumors, the vast majority, are adenocarcinomas. 00:29 And most colorectal cancers arise from preexisting adenomatous polyps. 00:36 This is an important point because if you have routine surveillance, you may be able to find polyps at a stage before they become frankly malignant and that's why we have the colon screening procedures that we do. General risk factors. 00:54 So, most of these cancer occur after the age of 40 or 45. 00:59 There are some hereditary syndromes that are associated with colorectal cancer such as familial adenomatous polyposis, hereditary non-polyposis colorectal cancer also known as Lynch syndrome and Peutz-Jeghers syndrome. 01:15 Clearly, inflammatory bowel disease, most typically, ulcerative colitis is associated with colorectal cancer. 01:21 And then, smoking and diets that are rich in processed meats are also epidemiologically associated and may be of - may be causal in the sense that they are inducing new mutations. 01:36 Let's cover some of the pathophysiology. We're looking here at a normal colon and the epithelium is totally appropriate. 01:47 They're not much in the way of inflammatory cells but it's at this point that we get a first hit. 01:53 The total pictures that you're going to see here, there's something generically called a Vogelgram. 01:59 This is based on the work of Bert Vogelstein who is a pathologist and who described the various genetic changes that can occur in colorectal cancer and other malignancies. 02:12 We will see the accumulation of various mutations as we go along. 02:16 It's important, however, to realize that they don't always happen in the sequence that I'm going to show you. 02:22 Some may happen out of turn. But that the accumulation of various additional mutations is what drives us along the pathway, finally, to malignancy. So, as an example of a first hit, you can have a known or an acquired mutation in adenomatous polyposis coli, the APC gene. 02:44 Mucosa at risk now are - includes the cells that have this mutation. 02:52 And then, we, on top of that, add some additional methylation or other inactivation of normal genes, a so called second hit and this may involve the APC B-catenin gene as well or may involve other genes. 03:09 We will have progression. Now, to the point where we have adenomas. These are not frankly malignant. 03:15 Adenomas are pre-malignant. Left to their own devices, they will eventually turn into malignancy by the acquisition of additional mutations. 03:25 But it's at this stage with screening that we may be able to pick them up. 03:29 At this point, they have developed the epithelial cells that originally had the first and the second hit, have acquired additional mutations. So, this can be KRAS which will be very much involved in epithelial proliferation. 03:46 There may be tumor-suppressor genes that are mutated, so, p53 is mutated and we lose some of that ability to protect the genome. You may have overexpression of COX-2. 04:01 We're moving along. And then, the final step is that we have carcinoma. 04:07 So, at this point, the cells that had acquired in each, in order to be malignant, a single cell has to have all of these acquisitions. Those abnormal cells now acquire things like mutations that will reactivate telomerase, so, that we can immortalize the cell lines. 04:27 There are just a whole host of mutations and I don't want you to memorize the different genes that are indicated in this figure but to be familiar with the concept of first and second hits and additional mutations that are accumulated, eventually, leading to malignancy. 04:48 Clinical presentation. Depending on where the tumor is within the colon, we're talking about colonic adenocarcinoma, depending on where it is, you may or may not be symptomatic. 05:00 Frequently, regardless of location, your patient will be bleeding Frequently, regardless of location, your patient will be bleeding because of erosion of the over lined epithelium and exposure of the underlying submucosal vasculature. 05:14 And so, there will be an iron deficiency anemia associated with chronic, perhaps, not even apparent bleeding into the stools. 05:22 So, we will also want to look for occult blood within the stools. 05:27 In left-sided carcinomas, particularly those more distally within the sigmoid, you may have constipation because the tumor grows so much that it obstructs the lumen. The right-sided carcinomas are rarely obstructed. 05:40 There's a lot more room for expansion within the cecum and the ascending colon. 05:44 Making a diagnosis, colonoscopy is going to be our most sensitive and specific and it's colonoscopy with biopsy. 05:56 And then, sending that biopsy off to your friendly neighborhood pathologist, me. 06:01 You can also evaluate this by CT but it's much less sensitive and much less specific. 06:07 Increasingly, we have other new techniques. So, swallow a pill that involves a camera and let it go from top to bottom and as it goes through the colon, you can have it take little pictures as it goes all the way through. 06:19 Again, this is more of experimental and I would say first step in a patient that you suspect of having colon carcinoma, do a colonoscopy. Laboratories are not necessarily sensitive or specific. 06:34 So, carcinoembryonic antigen is something that you can use to follow a known diagnosis of colon cancer. 06:40 So, if you treat the colon cancer and you remove it and they had a high CEA and then, it goes to zero once you do the resection, you can follow a CEA to see if you have recurrence in metastatic disease. 06:54 But as a primary diagnostic modality, not all that useful. 06:59 And a complete blood count is obviously useful for documenting anemia but as you already are abundantly aware, there are a bunch of causes for anemia. 07:08 So, the laboratory testing is not going to be making the diagnosis. Again, it's colonoscopy. 07:13 How do we manage this and what's the prognosis? What is being shown there are different stages of malignancy and there are various staging criteria? The Dukes, Kirkland and others. You don't need to memorize those but you do need to remember that stage one disease is superficial, small and localized. 07:36 Stage two is more invasive. And stage three, more invasive with nodal involvement. 07:41 Stage four, tumor is metastatic disease. An outcome for your five-year survival is going to be very much dependent on the stage at the time of the original diagnosis and resection. 07:55 Overall five-year survival for all colon cancers, all-comers is about 65% which is not great but it's not terrible. Five-year survival with curative surgical resection is better than 70%. 08:10 However, if there's distant metastasis or in other words, stage four disease, the five year survival is not very good at all. 08:17 It's about 15%. That speaks to the fact that surgery can be helpful if you have localized disease. 08:23 But if there's metastatic spread, not amenable to surgical intervention. 08:27 Our chemotherapy for colorectal cancer is not very good. So, the clinical staging clearly dictates what you're going to do. 08:36 If there's no distant metastatic disease by imaging preoperatively, then, chemotherapy as an adjuvant, new adjuvant therapy may be indicated or not. 08:49 And then, you can do laparoscopic or more commonly, it's going to be open surgery. 08:54 The open surgery is going to be important because we can take out the portion of the colon that's affected. 09:00 But also, we can do a nodal dissection which may be a bit more difficult to do just by laparoscopic procedures. 09:06 With clearly distant metastatic disease diagnosed by preoperative imaging, you may do palliative surgery so the patient doesn't bleed or the patient doesn't have an obstruction. But that surgery is not going to be curative. 09:20 And then, you will do your very best with the existing chemotherapy strategies. 09:24 With that, we have covered colorectal cancer.
About the Lecture
The lecture Colorectal Cancer by Richard Mitchell, MD, PhD is from the course Small and Large Intestines Disorders.
Included Quiz Questions
Which statement about colorectal cancer in the United States is correct?
- It is the second-most common cause of death from cancer in the United States.
- It is the most common cause of death from cancer in the United States.
- It is the most common cancer among both men and women.
- It is the second-most common cancer among both men and women.
- It is the second-most common cancer among children.
Which conditions are associated with colorectal cancer? Select all that apply.
- Familial adenomatous polyposis
- Hereditary non-polyposis colorectal cancer syndrome
- Peutz-Jeghers syndrome
- Scleroderma
- Neurofibromatosis I
What is a common type of anemia associated with colorectal cancer?
- Iron deficiency anemia
- Autoimmune hemolytic anemia
- Megaloblastic anemia
- Pernicious anemia
- Hemolytic anemia
What is the most sensitive and specific examination for colorectal cancer?
- Colonoscopy with biopsy
- CT scan
- CEA
- PILLCam 2
- MRI
What is the treatment paradigm for colorectal cancer without distant metastatic disease?
- Surgery with or without chemotherapy
- Chemotherapy alone
- Radiation alone
- Immunotherapy and chemotherapy
- Immunotherapy alone
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
Great lecture!! So easy to understand and so easy to memorize the different stages according to the picture.