Playlist
Show Playlist
Hide Playlist
Gout Medication: Colchicine, Uric Acid and More
-
Slides Gout Medication.pdf
-
Reference List Pharmacology.pdf
-
Download Lecture Overview
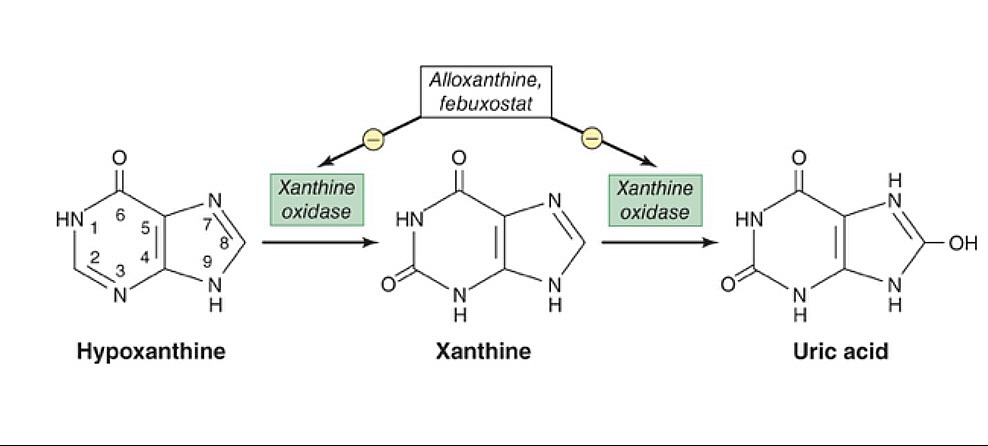
00:02 Let's move on to a completely different topic. 00:05 Let's move on to colchicine. 00:07 Now, colchicine is a selective inhibitor of microtubule assembly. 00:13 Now what happens is is that you're inhibiting tubulin which is necessary for cell division and motality. 00:19 So, what is tubulin? We can think of tubulin as a combination of a girdle or something that holds a building up and a communication tube that moves one thing to another from one part of the cell to another. 00:32 Inhibiting tubulin production actually inhibits a lot of cell function. 00:38 We call this a general mitotic poison. 00:42 So in actual fact, we can use these agents in cancers. 00:46 But in our particular discussion today, we're going to be talking about how it prevents neutrophil migration, activation, and degranulation. 00:54 It reduces the release of inflammatory cytokines and leukotrienes, particularly leukotriene B4. 01:01 This also decreases free radical formation which is also a mediator of pain and inflammation. 01:07 So where do we use colchicine? It's used in acute gout attacks. 01:12 It's often associated with diarrhea and in fact, this is something that patients should be warned about when prescribing this medication. 01:18 it's a very effective agent, it's very very potent, except for the fact that it causes diarrhea, it would be a perfect drug for treating gout. 01:27 Other indications for this medication include chronic gout prevention, and pericarditis treatment. 01:34 So, generally end up treating a patient for 3 months with colchicine which actually reduces the recurrence of pericarditis. 01:42 Interestingly enough, we also use this for disease called familial Mediteranean fever. 01:48 The toxicity of colchicine and overdose can cause liver, kidney, and other organ failure. 01:55 Remember that toxicity can be fatal. 01:58 So if you have a patient show up in your ER who is taking a lot of colchicine, you have to pay close attention to these patients. 02:06 Other drugs used in gout include the uricosurics. 02:10 Probenecid is your prototypical agent. 02:13 It is a weak acid and it competes with uric acid for reabsorption at the proximal tubule inside the kidney. 02:20 When you give a patient probenecid, the uric acid is released in the urine. 02:26 So here's an example of how uric acid is reabsorbed. 02:30 You filter about 100 litres a day of blood into the glomerulus. 02:35 90 % of the uric acid is reabsorbed at the proximal convoluted tubule allowing a small amount of uric acid excretion at the end. 02:45 This weak acid transporter or WAT is what picks up these weak acids and brings them back into the blood. 02:53 By administering uric acid agents such as probenecid, you actually increase the amount of reabsorption of those drugs like probenecid and the uric acid is therefore released into the urine unchanged and not reabsorbed. 03:15 Now at low doses, these uricosuric agents also compete with uric acid for secretion. 03:21 So that causes an increase in uric acid levels as well. 03:25 So, a similar phenomenon also occurs with aspirin. 03:28 This causes an elevation of uric acid. 03:31 So, the bottomline is this. 03:33 Is that these drugs are uricosuric, they do promote release of uric acid in the urine. 03:42 However, they can also predict or promote an acute gout attack too, so you have to be very careful using these drugs. 03:49 And you don't want to slam people with these medications just without thinking about them because you can precipitate a gout attack. 03:56 The uses of the uricosurics is treatment of chronic gout. 04:05 Let's talk about the xanthine oxidase inhibitors. 04:08 At one time, these were the mainstay of gout treatment. 04:11 Let's look at the physiology behind allopurinol. 04:15 Allopurinol is converted to alloxanthine by the liver. 04:18 This is a suicide inhibitor of xanthine oxidase. 04:23 So, uric acid production is cut dramatically because xanthine oxidase is also needed to produce uric acid. 04:31 Now, we sometimes use allopurinol in chemotherapy of cancer patients to reduce uric acid production from purines that are released during the cell death of all those cancer cells. 04:43 So, this is an important consideration in cancer chemotherapy. 04:46 When we're killing cells, there's all this stuff being generated as the cells are dying. 04:52 This massive cell lysis releases various intracellular metabolites into the circulatory system. 04:57 Ultimately, impacting multiple organ systems and a process called tumor lysis syndrome. 05:02 One of the cellular components released is nucleic acid, which then causes elevated peering levels. 05:07 Without treatment, these are then converted into uric acid which precipitates in the renal tubules and causes an acute kidney injury. 05:15 Other non-purine inhibitors of xanthine oxidase are out on the market. 05:20 Now they are more selective, but they're quite a bit more expensive, In terms of toxicity of allopurinol, I think the most commonly thing that we see is gastrointestinal upset. 05:32 We often see GI upset in low doses of allopurinol usage. 05:38 Our rash, vasculitis, and peripheral neuritis are often seen. 05:41 I've had many patients who developed rash on allopurinol. 05:45 A rare but very feared complication of allopurinol toxicity is aplastic anemia. 05:51 So if you start to see some abnormalities with your white counts and your red counts, be very aware that your allopurinol may be inducing a serious disease. 06:00 With respect to the newer xanthine oxidase agents, this agent can cause gastrointestinal upset as well as liver function abnormalities, and of course you can get a headache.
About the Lecture
The lecture Gout Medication: Colchicine, Uric Acid and More by Pravin Shukle, MD is from the course Inflammation Pharmacology. It contains the following chapters:
- Drugs in Gout Treatment
- Uricosurics
- Xanthine Oxidase Inhibitors
Included Quiz Questions
What is NOT an effect of colchicine?
- Inhibits uric acid formation
- Inhibits microtubule assembly
- Reduces leukotriene B4 production
- Decreases formation of free radical
- Reduces macrophage migration
Where is 90% of uric acid absorbed in the kidney?
- Proximal convoluted tubule
- Loop of Henle
- Distal convoluted tubule
- Collecting duct
- Bladder
What is the primary mechanism of action for allopurinol?
- Its active metabolite irreversibly inhibits xanthine oxidase.
- Allopurinol prevents the formation of microtubules.
- Allopurinol prevents the release of leukotriene B4.
- Allopurinol is reabsorbed by the collecting tubules of the kidney allowing uric acid to be excreted.
- The byproduct of allopurinol inhibits phagocytosis by macrophages.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |